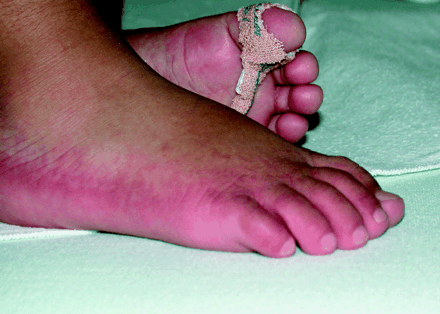
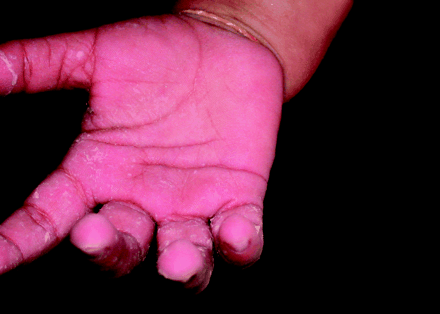
Previously well, developmentally normal 20-month-old twin girls presented with weakness, anorexia, a papular rash and increasingly swollen, red and painful hands and feet of 1 month's duration. They had no history of fever, conjunctivitis, lymphadenopathy or oral changes characteristic of Kawasaki disease. The children appeared irritable and unwell and were diaphoretic but afebrile. Both had tachycardia, and one had an elevated blood pressure of 130/90 mm Hg (95th percentile for age 108/62 mm Hg). Both children had reduced muscle power and diminished reflexes. Their palms and soles were erythematous and indurated with desquamation, judged to be acrodynia (Figs. 1 and 2).
Figure 2. Photo: Images courtesy Dr. Michael Weinstein
Figure 1. Photo: Images courtesy Dr. Michael Weinstein
Mercury toxicity was suspected, and further questionning revealed that the infants had been given a mercury-containing “teething powder” from India once or twice a week over the 4 preceding months. The girls' blood mercury levels were 176 and 209 (normally < 18) μmol/L. Chelation therapy with 2,3-dimercaptosuccinic acid was administered through nasogastric tubes. Before admission the twins had regressed developmentally and were unable to feed orally, sit or walk. Over the 8 weeks in hospital they showed some minor neurocognitive improvements, but their long-term prognosis is uncertain.
Mercury exists in inorganic and organic forms. Organic mercury has recently received attention because of the accumulation of methylmercury in some predatory fish1 and the use of thimerosal as a preservative in some vaccines.2 A type of inorganic mercury known as calomel (“sweet mercury”) was once commonly used to treat many ailments, including yellow fever, typhus and syphilis.3 Until the recognition of their toxicity in the 1940s, calomel-based teething powders caused a scourge of mercury poisoning called “pink disease” or acrodynia among infants and children.4
Acrodynia is characterized by a dusky pink discolouration, swelling, paresthesia and desquamation of the hands and feet. Symptoms of catecholamine excess such as sweating and hypertension occur because mercury blocks the degradation pathway of catecholamines. Other manifestations of mercury toxicity include renal dysfunction, peripheral neuropathy and neuropsychiatric symptoms (e.g., emotional lability, memory impairment and insomnia). Although calomel-containing compounds are banned in North America, they are still used in other parts of the world such as Southeast Asia, and calomel can also be found in various alternative medicine products.5
Clinical presentations suggestive of pheochromocytoma (e.g., excessive sweating, tachycardia and hypertension) or of Kawasaki disease but not meeting the full criteria6 should also prompt consideration of mercury toxicity. Although rash, oral mucosa and extremity changes are features of both Kawasaki disease and mercury toxicity, patients with the latter do not have a fever. Rash and extremity changes are not features of pheochromocytoma. The peeling of the skin on the extremities seen in cases of Kawasaki disease often occurs 1–3 weeks after presentation, as opposed to occurring concurrently with the rash and other findings in cases of mercury poisoning. Mercury poisoning is confirmed by measuring levels in blood, urine or hair samples.7,8
The most important step in the management of mercury poisoning is eliminating the source of exposure. The effectiveness of chelation therapy in reversing symptoms is not entirely clear.9
Our case stresses the potential harm of mercury. It reminds us to think of a toxic exposure when family members present with the same unusual constellation of symptoms. It also highlights the common misconception that all alternative medicines are safe and benign.10
Michael Weinstein Stacey Bernstein Department of Pediatrics Hospital for Sick Children Toronto, Ont.