Abstract
Background: Liver transplantation is now routine therapy for a variety of childhood liver diseases; however, there are no detailed reports of long-term results from a Canadian centre. We reviewed data from the first 16 years of a pediatric liver transplantation program to determine survival, complications and long-term outcomes.
Methods: The outcomes to December 2000 for all children (age less than 18 years) who received a liver transplant at the London Health Sciences Centre between April 1984 and December 1999 were reviewed. The recipients were grouped according to the period in which they received the transplant (period 1, April 1984 to July 1988; period 2, August 1988 to December 1993; or period 3, January 1994 to December 1999). Data were obtained from medical charts; in- person interviews, questionnaires or telephone contact with patients and their families; contact with referring physicians; and school records. Outcome measures included patient survival, retransplantation, complications and long-term outcomes (specifically steroid withdrawal and growth and development).
Results: A total of 116 children (29 in period 1, 46 in period 2 and 41 in period 3) (median age 5.6 years at the time of the procedure) received a total of 140 liver grafts (32 in period 1, 57 in period 2 and 51 in period 3). Of the 116 patients, 23 (20%) were less than 1 year old at the time of transplantation. Biliary atresia was the most common indication for liver transplantation (57 [49%] of the 116 patients). The number of patients surviving to 1 year after transplantation was 20 (69%) of the 29 patients from period 1, 40 (87%) of the 46 patients from period 2 and 38 (93%) of the 41 patients from period 3. The percentage of patients receiving reduced size grafts from adult donors, including live donors, increased from 2/32 (6%) in period 1 to 22/51 (43%) in period 3. Retransplantation was required for 9 (31%) of the 29 patients from period 1, 6 (13%) of the 46 patients from period 2 and 7 (17%) of the 41 patients from period 3. Among these patients, 1-year survival was 33% (3/9) for period 1, 83% (5/6) for period 2 and 100% (7/7) for period 3. Eighteen of the 22 deaths occurred within 4 months after surgery. Only 3 (3%) of the 116 patients experienced post-transplant lymphoproliferative disease. Steroids were discontinued (usually within 2 years after surgery) for the following proportions of surviving transplant recipients: 17 (89%) of the 19 patients from period 1, 29 (78%) of the 37 patients from period 2 and 21 (55%) of the 38 patients from period 3. Most of the surviving patients had normal growth (82/94 or 87%) and development (73/94 or 78%), and these outcomes were consistent across the 3 periods of study.
Interpretation: Survival and long-term outcomes continue to improve for most children who receive liver transplants. These improvements may be due to improved surgical technique, perioperative care and, to a lesser extent, immunosuppressive therapy.
Pediatric liver transplantation is now routine therapy for irreversible acute or chronic liver disease and many inborn errors of metabolism.1,2,3 Because of donor shortages, some Canadian children still die while awaiting transplantation (there were 21 such deaths in the period 1998–2000), but approximately 40 children receive livers each year in Canada, including 8 to 15 annually in London, Ont. As the number of children surviving such surgery increases, it becomes increasingly likely that some of them will be cared for, at least in part, by family physicians. Currently, about 1% of primary care physicians could encounter a child who needs or has received a liver transplant, but there is a paucity of information regarding the long-term outcomes of pediatric liver transplantation in Canada.
Pediatric liver transplantation was first performed at the University of Western Ontario in 1984 as part of a combined adult–pediatric program.4,5 Many of these children are now in their second decade after surgery, and we report here results for more than 100 children who underwent transplantation between 1984 and 1999 with respect to their characteristics, processes of care, survival, complications and long-term outcomes (specifically steroid withdrawal and growth and development), and any effects related to the period in which they underwent surgery.
Methods
Subjects
The London Health Sciences Centre is the principal pediatric liver transplant referral centre for the Atlantic provinces, Manitoba and British Columbia, as well as southwestern Ontario. Children less than 18 years of age who received a liver transplant between initiation of the pediatric program, in April 1984, and December 1999 were included in this review; those who received combined liver and bowel transplants or multiorgan transplants were excluded. Approximately 75% of the patients described here were from outside Ontario. In addition to reviewing the overall data, we examined the results for children less than 1 year of age at transplant and those who received living-related liver transplants.
After obtaining permission from parents or guardians, we gathered long-term data for this report from medical charts; in-person interviews, questionnaires or telephone contact with patients' families; contact with referring physicians; and school records. The results of any formal developmental or psychological testing were also reviewed.
All aspects of the pediatric liver transplant program, including this study, were approved by the ethics review board of the London Health Sciences Centre or the University of Western Ontario (or both).
Immunosuppression
For this review, patients were divided into 3 groups on the basis of the period in which they received their transplant, to allow comparison of the effects of the immunosuppression they received and improvements in surgical techniques and in preoperative and postoperative care.
Patients in all 3 groups received cyclosporine, steroids and azathioprine. In addition, patients who underwent surgery in period 1 (April 1984 to July 1988) received antilymphocyte globulin, a polyclonal preparation against T lymphocytes, and those who underwent surgery in period 2 (August 1988 to December 1993) received muromonab-CD3 (OKT3), a mouse monoclonal antibody against the CD3 complex of T lymphoctyes. Patients who underwent surgery in period 3 (January 1994 to December 1999) did not receive any additional immunosuppression. Since 1997 selected patients have received tacrolimus instead of cyclosporine, usually for recurrent rejection. Both cyclosporine and tacrolimus have potential toxic effects, including renal effects. To minimize nephrotoxicity, cyclosporine was given in 3 divided doses.6,7 Immunosuppression in general has a number of side effects, including increased risk of infection and post-transplant lymphoproliferative disease (PTLD); therefore immunosuppression was decreased as quickly as feasible. Steroids were weaned and discontinued by 1 year after transplantation (if the patient could tolerate the discontinuation), and azathioprine was weaned and discontinued by 2 years.
All patients received antibiotic prophylaxis at the time of transplantation. Since 1990, cytomegalovirus (CMV) prophylaxis, in the form of a gamma globulin product and oral acyclovir or ganciclovir, has been given.
Types of liver transplantation
Children received either a whole graft (usually from a pediatric donor) or a reduced-size graft consisting of the left or left lateral lobe (usually from an adult donor). Reduced-size grafts from cadaveric or live donors were used to increase the number of grafts available for pediatric transplants. In some instances, a single cadaveric graft could be used for 2 recipients, the right lobe being placed in an adult recipient and the left lobe in a child; these grafts are called split-liver grafts.
Outcome measures
For each period, patient survival and short-term and long-term complications were reviewed. The data included causes of death, surgical problems, retransplantation, infection, rejection, renal dysfunction and PTLD. Long-term outcomes included discontinuation of steroids and growth and development (including school performance).
Rejection episodes were confirmed, whenever possible, by liver biopsy and were treated with pulse steroids. Steroid-resistant rejection was treated with an antilymphocyte product, and, from 1997 onward, patients with recurrent rejection episodes were switched from cyclosporine to tacrolimus.
Renal function was assessed, before or after liver transplant (or both), by serum creatinine levels and, since 1995, by glomerular filtration rate as determined by radioactive tracer.8,9
Statistical analysis
Descriptive analyses were used for most of the data. Patient survival was also calculated by the Kaplan–Meier method.
Results
From December 1977 to December 1999, 924 liver transplantations were performed at the London Health Sciences Centre; this total included 140 grafts transplanted in 116 children, beginning in April 1984 (Table 1). Most of these pediatric patients had biliary atresia (57 or 49%), and 29 (75%) of them came from outside of Ontario (Table 1).
Table 1.
Most of the procedures involved whole grafts (Table 2). The proportions of reduced-size, split-liver and living- related grafts increased in period 3 (Table 2).
Table 2.
Survival and deaths
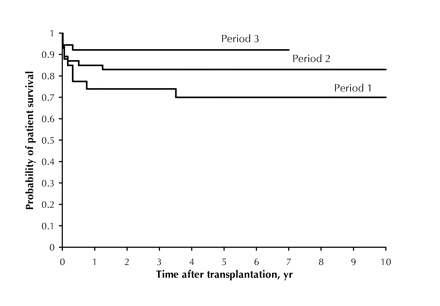
Actuarial patient survival (Fig. 1) was comparable to actual patient survival (Table 2). Patient survival improved continuously, and for period 3 actual 1-year survival was 38/41 (93%). Survival in all groups was stable beyond 6 months. The survival of infants also improved over time (Table 2). For the 23 infants (median age 9 [range 3 to 12] months) who received liver transplants, 1-year actual survival was 19/23 (83%). During 1998 and 1999, 10 infants received liver transplants, and all were alive and well at the time of writing; these results indicate further improvements in outcome for these young children. Similarly, the length of stay in the intensive care unit (ICU) and in hospital declined over time (Table 2).
Fig. 1: Actuarial patient survival (Kaplan–Meier method) according to the period when transplantation was performed. For period 1 there were 29 patients, for period 2 there were 46 patients, and for period 3 there were 41 patients.
Beginning in 1993, 8 children (7 to 12 months of age) received a transplant from a parent donor. The age of donors ranged from 20 to 41 years, and their time in hospital was short (mean 6 days). Of the recipients, 1 child died 15 months after surgery (because of septicemia) and the other 7 children were well at the time of writing.
In total, 22 (19%) of the children died. One child, who was on life support and was in a deep coma, died during the transplant procedure itelf. Eight (36%) of the deaths occurred within the first week after transplantation, and 18 (82%) within 4 months (Table 3). Only 4 deaths occurred more than 1 year after surgery. Two of these, in children who had undergone splenectomy, were due to septicemia and occurred despite appropriate prophylaxis. A child with cystic fibrosis died of pneumonia 8.3 years after the transplant procedure. The fourth death occurred 14 years after initial liver transplantation for autoimmune hepatitis. This patient underwent 2 retransplantations for recurrence of disease, but a third retransplanation was not performed.
Table 3.
Other complications
Complications other than death are summarized in Table 4.
Table 4.
In general, surgical complications were few, and their frequency decreased over time (Table 4). Hepatic artery thrombosis occurred in 11 (8%) of the 140 grafts; 6 of the affected children underwent retransplantation, and 2 died while awaiting this procedure. Portal vein thrombosis occurred less than 1 year after transplantation in 9 (6%) of the 140 grafts; retransplantation was required in 4 cases. Biliary tract problems occurred in 20 (14%) of the 140 grafts. Of these, 8 required reoperation, and retransplantation was performed in 5 cases. Other surgical complications included primary nonfunction of the graft (3/140), bowel perforation (4/140) and abscess (5/140).
Surgical complications occurring more than 1 year after the procedure were infrequent. Portal vein thrombosis developed more than 3 years postoperatively in 6 children. Three of these patients underwent splenectomy and received splenorenal shunts and have subsequently done well. Late biliary stricture developed in 6 children; 4 of these required reoperation. Three other children required surgery for bowel obstruction.
Common respiratory complications included pulmonary edema and pleural effusion (Table 4); others occurred infrequently. Pulmonary edema was more common in patients who underwent transplantation in period 2 and who received the immunosuppressive agent muromonab-CD3.
Infectious episodes were common (Table 4), most occurring early, during the period of maximum immunosuppression. Although most other transplantation programs administer prophylaxis against Pneumocystis carinii, our program does not; nonetheless, there were no cases of P. carinii infection. Seventeen (12%) of the 140 transplants were affected by CMV infections; 5 of these patients died. All CMV-related deaths and 11 of the 17 cases occurred before initiation of routine prophylaxis with gamma globulin and acyclovir or gancyclovir.
Most infections occurring more than 1 year after transplantation did not necessitate admission to hospital (upper respiratory tract infections, otitis media and gastroenteritis), and most of those that did were due to varicella zoster virus. Thirty-one (32%) of the 98 patients who survived more than 1 year were admitted for intravenous treatment with acyclovir or varicella zoster immune globulin therapy (or both). Two other children were admitted for septic shock; one of these had previously undergone splenectomy.
Acute rejection within the first year after surgery was common (Table 4); as previously reported,10 the rates were similar for periods 1 and 3 and higher among patients who received muromonab-CD3 in period 2. Most cases of rejection responded to steroid therapy. Frequently, there was more than one episode of acute rejection per graft. Chronic rejection occurred in 2 patients, 1 of whom underwent retransplantation; at the time of writing, the other patient was awaiting retransplantation.
Acute rejection more than 1 year after surgery was less common (Table 4). Among patients who underwent transplantation in period 1, 4 episodes of late rejection occurred in teenagers, and noncompliance with immunosuppressive therapy was suspected. Among those who underwent surgery in period 2, 4 episodes of acute rejection occurred after weaning from steroids, and 1 episode occurred 3 years after liver transplantation, in a patient for whom all immunosuppression was withheld for 1 week after a presumed drug overdose. Parental noncompliance was suspected in 2 of the 4 episodes that occurred in patients who underwent surgery in period 3. Chronic rejection occurred in 3 patients more than 1 year after transplantation; 2 of these patients underwent retransplantation and the other was stable at the time of writing.
Only one child required dialysis after transplantation (for 1 week), although for 10 (7%) of the 140 transplants, the patients experienced transient renal insufficiency. Long-term renal function was tested in 93 patients; in 81 (87%) of these, serum levels of creatinine were normal, but glomerular filtration rate was reduced in 57 (81%) of the 70 patients in whom it was assessed. No patients experienced renal failure more than 1 year after transplantation.
Retransplantation was required in 22 of the 116 patients (Table 2). The proportion of patients needing retransplantation declined in periods 2 and 3, and their survival increased dramatically: all 11 patients who have undergone retransplantation since 1993 have survived. Two patients originally treated in period 1 underwent a total of 3 transplant procedures; both of these children have died.
Only 3 patients (3%) experienced PTLD, 5, 6 and 8 years respectively after their transplant procedures (Table 4). All 3 had received additional immunosuppression for steroid- resistant rejection. Two were positive for Epstein-Barr virus (EBV), and EBV status was unknown for the third. Therapy for PTLD included reduced immunosuppression in all 3 patients and chemotherapy in 2. One of these children experienced acute liver failure of unknown cause 11/2 years after the chemotherapy and underwent successful retransplantation. At the time of writing, this child had survived for 2 years with no evidence of recurrent PTLD.
Other complications in the early post-transplant period (Table 4) included seizures, which were often associated with metabolic derangements such as hyperkalemia, hypocalcemia and hypomagnesemia; only the hypomagnesemia persisted, and supplementation was often required. These conditions were likely due to a combination of cyclosporine side effects and the transplant itself. Hypertension was observed in most patients immediately after the surgery, but by 1 year most patients were normotensive and did not require antihypertensive therapy. The hypertension was likely due to a combination of high-dose steroid and cyclosporine.
Long-term outcomes
Ninety-eight children survived at least 1 year, and 94 of those were alive in December 2000, including 40 who had reached their second decade after transplantation. The long-term outcome data for these 94 children are summarized in Table 5.
Table 5.
The growth of most children was between the 3rd and 97th percentiles for age (Table 5). Those whose growth was very much less than the 3rd percentile had another problem accounting for their short stature, such as familial short stature, severe neurological impairment or Alagille's syndrome.
Intellectual development was also satisfactory. Up to December 2000, 9 of the 94 of children were of preschool age. For all but one of the preschoolers, parents, child care workers and pediatricians felt that development was appropriate. The child with developmental delay was receiving special help consisting of physical and speech therapy.
Most of the school-age children were considered normal by their parents, school officials and pediatricians. In a number of cases, even though development was felt to be appropriate, the children needed additional instruction, usually in mathematics. Two school-age children were delayed in 1 or 2 or more areas and required special academic help. Three of the children were grossly delayed and had suffered an identifiable neurological insult in the peritransplant period.
Most of the patients who survived more than 1 year after transplantation had their steroid immunosuppression discontinued, although in some cases the drugs were restarted because of rejection (Table 5). Of the 27 patients still receiving steroids as of December 2000, 9 had received their transplant less than 2 years previously while 12 had remained on steroids for more than 5 years since transplantation, usually at the discretion of local physicians.
Interpretation
Survival of children receiving liver transplants has now reliably reached 85% or more at 1 year.1,2,3,11,12,13,14,15,16,17,18,19,20,21,22 Indeed, in our program, survival for patients undergoing surgery from 1994 to 1999 was 93%, which is comparable to the rates in other, larger centres;1,2,3,11,12,13,14,15,16,17,18,19,20,21,22 our survival results for infants are also comparable to those of larger centres.16,23,24,25 The improvements in outcomes are primarily due to improved surgical techniques, along with improved preoperative, intraoperative and postoperative care,1,11,12 and are exemplified by reductions in the frequency of complications and the length of ICU and hospital stay. As in other programs,13,16,17,19,26,27 most deaths in our program occurred in the first 4 months after transplantation; patient survival over the long term was stable, unlike the situation for kidney transplant recipients.28,29 It should also be noted that the percentage of reduced size grafts, including live donors, increased in period 3, reflecting improvements in surgical technique, the decrease in pediatric donors and the increase in potential recipients.
The long-term outcomes of our patients are particularly encouraging, and 40 children are now in their second decade after transplantation and doing well. Most of these patients were able to discontinue steroids by 2 years, and most had normal growth and development. The majority were in the age-appropriate grade, although some did require special help, particularly in the area of mathematics. It is unclear whether this need was due to their preexisting disease, the length of time they waited for their transplant or the procedure itself.30,31,32,33,34,35 Because developmental assessment was not done in a standardized fashion, with some reports coming from parents and teachers, it is possible that our results overestimated developmental achievements. It would be helpful in future studies to undertake standardized assessments before and after transplantation. All of the children with global developmental delay had a known neurological insult before transplantation or during the surgery.
Short-term and long-term complications were similar to those reported by others6,8,36,37,38,39,40 and decreased over time. Some issues will be of particular interest to primary care physicians. First, varicella zoster virus accounted for several hospital admissions. The development of the varicella zoster vaccine may decrease the occurrence of this infection, and preoperative vaccination should be considered. Second, noncompliance with immunosuppression probably played a role in some cases of late rejection, particularly among teenagers.41,42,43,44,45 Finally, PTLD has been a significant concern, particularly in children. The reported incidence of PTLD is 3% to 30%,46,47,48,49,50,51,52,53 and this condition is often associated with EBV infection. Strategies to treat PTLD include initiating antiviral therapies, decreasing immunosuppression and administering chemotherapy. Tacrolimus may be associated with a higher incidence of PTLD than cyclosporine,54 but this suggestion is controversial. Only 3 of the patients in our program experienced PTLD. The reason for the low incidence is unclear. Minimizing immunosuppression as rapidly as possible might have been a factor, and our practice of providing CMV prophylaxis might also have afforded prophylaxis against EBV.
In conclusion, patient and graft survival for pediatric liver transplant recipients improved markedly over the period 1984 to 1999, and increasing numbers of these patients are now reaching their second decade since the procedure. Growth and development in these children has been encouraging. Our results for infants and older children demonstrate that excellent outcomes can be achieved within a combined adult and pediatric program. Finally, complications such as infection, renal dysfunction and PTLD underscore the need to improve immunosuppression protocols.
Footnotes
-
This article has been peer reviewed.
Contributors: Paul R. Atkison was the principal author of the paper. B. Catherine Ross gathered many of the data reported here. She also wrote some early drafts of the manuscript and was involved in editing the paper. Sandy Williams was involved in much of the data acquisition and in editing the manuscript. John Howard was involved in some of the data acquisition and in editing the manuscript. John Sommerauer was involved in data acquisition, particularly for patients treated during the early years of the Paediatric Transplant Program. He also participated in editing the manuscript. Douglas Quan and William Wall contributed to the preparation and editing of the manuscript. All authors gave final approval for the paper.
Competing interests: None declared.