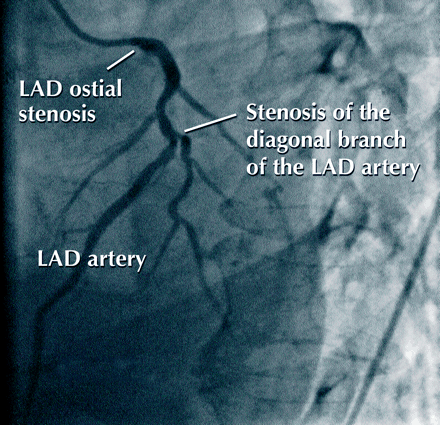
A 63-year-old man with a long-standing history of nephrotic syndrome due to membranous glomerulonephropathy, an 85% stenosis of his right carotid artery and class IV angina was referred for urgent cardiac surgery. He had a previous positive treadmill test, and a coronary angiogram revealed triple-vessel disease, including an 80% stenosis at the ostium of the left anterior descending (LAD) artery (Fig. 1) with disease of its diagonal branch, 70% stenosis of the circumflex artery with occlusion of its obtuse marginal branch, and an occluded right coronary artery that filled via collaterals from the circumflex system. Coronary artery bypass grafting (CABG) was performed and included a saphenous vein graft to the diagonal and obtuse marginal arteries as well as anastomosis of the left internal thoracic artery (LITA) to the LAD artery.
One challenge of CABG is that good methods of assessing graft patency have not been established. With “off-pump” CABG, or “beating heart surgery,” the aorta is not cannulated or clamped as in conventional CABG,1 and thus the risk of stroke and other complications is theoretically reduced. Delicate arterial anastomoses are performed on a beating heart, and in order to assess the patency of these anastomoses many surgeons rely solely on the absence of myocardial ischemia when the patient is taken off bypass. Two widely used but unproven methods of assessing anastomoses rely on non-imaging, pulsed-wave Doppler ultrasonography2 or transit time flow measurement.3
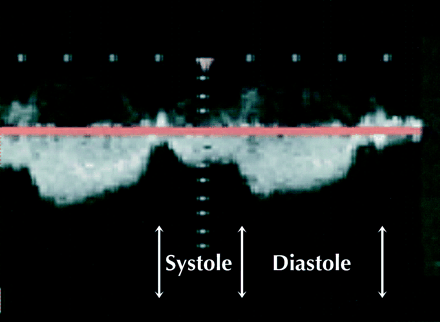
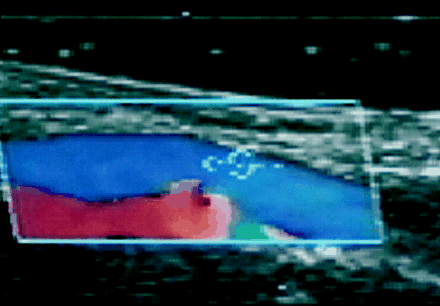
For our patient, a conventional CABG technique was used. The same type of ultrasound probe that was used to detect a fragile atheromatous plaque in the patient's aorta (which can be avoided during cross-clamping and cannulation of the aorta, thus reducing the risk of intraoperative cerebral ischemia) was applied to the LITA–LAD anastomosis after the heart was restarted. Fig. 2 shows this anastomosis: the blue section represents laminar flow in the LITA and in the LAD distal to the anastomosis, and the red section represents the turbulent flow in the diseased and stenosed segment of the LAD proximal to the anastomosis. The continuity of laminar flow from the LITA to the LAD suggests that the graft has bypassed the diseased LAD and is functioning as a single conduit. The image gives a glimpse of the frailty of human life — occlusion of this 2-mm–wide channel is enough to make the difference between life and death. Fig. 3, obtained using the same ultrasound probe, shows a pattern of blood flow at the site of anastomosis that is also consistent with a patent graft. The Doppler spectrum shows that flow increases in systole (a downward Doppler shift) and continues to increase, with longer duration, during diastole4 (the red baseline indicates zero flow). An occluded vessel may show only a systolic Doppler signal.
The patient recovered from the CABG procedure without complications. His exercise tolerance improved, and at the time of writing he was free from angina. As the efficacy of this technology is reinforced in future clinical trials, intraoperative ultrasonography may become standard practice for many indications in cardiothoracic operating rooms.
Trevor Bardell G. Andrew Y. Hamilton Douglas H. Bergstrome Mohamed J. Ali Departments of Anesthesiology and Surgery Queen's University Kingston, Ont.
Footnotes
-
Mr. Bardell is a medical student at Queen's University.