- © 2004 Canadian Medical Association or its licensors
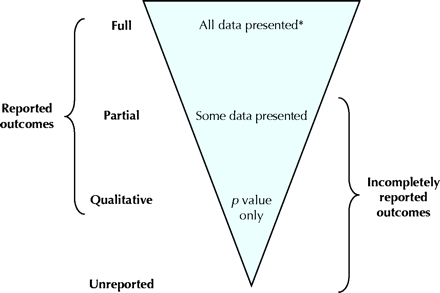
Fig. 1: Hierarchy of the levels of outcome reporting. *See the online appendix (www.cmaj.ca/cgi/content/full/171/7/735/DC1) for descriptions of the amount of data required for meta-analyses of fully reported outcomes
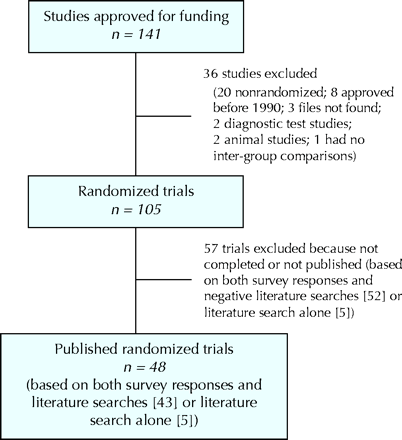
Fig. 2: Identification of published randomized trials approved for funding by the Canadian Institutes of Health Research (formerly the Medical Research Council of Canada) from 1990 to 1998.
- View this table:
Table 1.
Fig. 3: Number of incompletely reported outcomes per trial, displayed according to total number of outcomes per trial, for outcomes measured to assess the efficacy of an intervention (top panel) and those measured to assess the harm of an intervention (bottom panel).
- View this table:
Table 2.
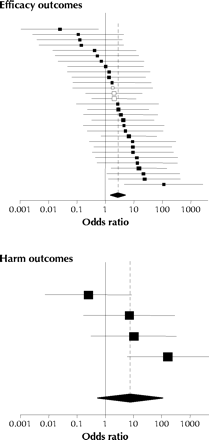
Fig. 4: Odds ratios (squares) and 95% confidence intervals for outcome reporting bias involving efficacy outcomes (30 trials, top panel) and harm outcomes (4 trials, bottom panel). The size of the square reflects the statistical weight of the trial in calculating the pooled odds ratio (represented by the diamond and dotted line).
- View this table:
Table 3.
- © 2004 Canadian Medical Association or its licensors