Figure 1: The serotonin synapse. Serotonin is synthesized from tryptophan by the enzyme tryptophan hydroxylase. Serotonin is then packaged into vesicles for release into the synaptic cleft, which occurs when there is sufficient stimulation of the neuron. Serotonin released from the serotonin neuron into the synaptic cleft has multiple actions. (1) Serotonin binds to its receptors on other neurons. Activation of postsynaptic receptors results in transduction of the signal that initially stimulated the serotonin neuron. (2) Serotonin also binds to presynaptic serotonin receptors on the neuron from which it was released, which provides feedback and regulates plasticity of the neuron. (3) Serotonin is taken up back into the presynaptic serotonin neuron by the serotonin transporter. Serotonin is then recycled for future release or broken down by monoamine oxidase and excreted in urine. Image by: Lianne Friesen and Nicholas Woolridge
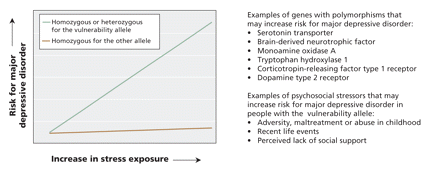
Figure 2: Schematic representation of gene–environment interaction. Genetic polymorphisms may influence a person's risk for major depressive disorder in certain psychosocial environments. Genes may contribute to a person's risk for major depressive disorder through polymorphisms or variations in their DNA sequence that may influence the expression or activity of the gene product. A person may have 2 identical alleles of a gene (homozygous) or 2 different alleles (heterozygous). Although carrying a certain allele may in itself not affect a person's risk for developing major depressive disorder, several genetic polymorphisms have been shown to interact with the environment in terms of their effect on disease vulnerability. Please note that this figure is a schematic representation. Individual polymorphisms and individual studies may differ in terms of how a person's risk for major depressive disorder is affected by carrying 1 or 2 vulnerability alleles. Limitations of studies to date include the method used to assess disease vulnerability (e.g., some studies used a person's score on a depression questionnaire rather than a formal diagnosis of major depressive disorder as the outcome variable) and the retrospective nature of most studies' assessment of psychosocial stress exposure.5–7,9–13
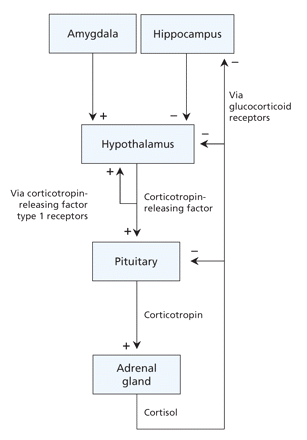
Figure 3: The hypothalamic–pituitary–adrenal axis. This system is activated by stress directly at the level of the hypothalamus or indirectly at the level of the amygdala. The hypothalamus produces and releases corticotropin-releasing factor. Local stimulation of corticotropin-releasing factor type 1 receptors results in additional release of corticotropin-releasing factor. This creates a feed-forward loop, which facilitates a rapid response to the stressor. In the pituitary, stimulation of corticotropin-releasing factor type 1 receptors results in release of corticotropin (also known as adrenocorticotropic hormone). The adrenal glands are stimulated by corticotropin to produce the stress hormone cortisol, which affects many organs, including the brain. The hippocampus is an important target of cortisol. Local activation of glucocorticoid receptors helps the hippocampus control the hypothalamic–pituitary–adrenal axis. Glucocorticoid receptors are also found in the hypothalamus and pituitary. Chronic stress increases the level of corticotropin-releasing factor and cortisol and decreases expression of corticotropin-releasing factor type 1 receptors and glucocorticoid receptors. Similar changes have been found in some patients with major depressive disorder. Stress-and depression-associated changes at the level of the hippocampus in particular are thought to underlie the structural changes seen in this brain region, which in turn may contribute to chronic disinhibition of the hypothalamic–pituitary–adrenal axis.
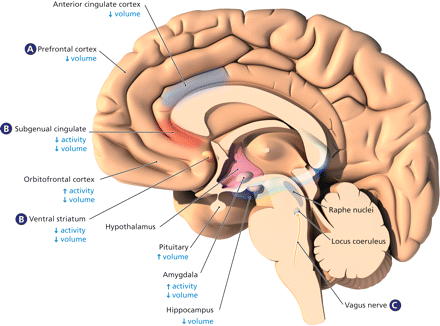
Figure 4: Structural and functional brain abnormalities in patients with major depressive disorder, and the site of action of novel neurostimulation techniques with antidepressant potential. Structural abnormalities in the brains of patients with major depressive disorder have been observed in the cortical and subcortical regions. The anterior cingulate cortex, especially the subgenual cingulate, may show volume reduction. This has also been observed in other subregions of the prefrontal cortex as well as in the orbitofrontal cortex. Subcortical regions in which volume reduction has been observed include the amygdala, hippocampus and ventral striatum. (A) Transcranial magnetic stimulation of the dorsolateral prefrontal cortex and (B) deep-brain stimulation of the subgenual cingulate have been shown to have antidepressant effects in some patients. The rationale for deep-brain stimulation of the subgenual cingulate or the ventral striatum is largely based on neuroimaging findings of functional dysregulation in this region. (C) Vagus nerve stimulation might have antidepressant properties via its effects on the locus coeruleus, an area in the brain stem from which norepinephrine neurons originate. Image by: Lianne Friesen and Nicholas Woolridge