Abstract
Background: Few large, systematic, prospective studies have documented the characteristics and clinical outcomes of patients awaiting cardiac catheterization and the delays that they experience. The primary objective of this study was to quantify the waiting times, morbidity and mortality of patients waiting for catheterization. A secondary objective was to identify predictors of cardiac events that occur while patients are waiting.
Methods: A computerized, prospective, central waiting list registry was developed at a regional centre in Hamilton, Ont., serving 2.2 million people in southern Ontario. Between Apr. 1, 1998, and Mar. 31, 2000, 8030 consecutive patients (4725 outpatients and 3305 inpatients) were referred for cardiac catheterization. Major cardiac outcomes while on the waiting list (death, myocardial infarction and congestive heart failure) were documented prospectively and related to requested versus actual waiting time.
Results: Most of the referrals (7345 [91.5%]) were for a primary diagnosis of suspected coronary artery disease. The median waiting time was 6 (interquartile range [IQR] 4) days for inpatients and 60 (IQR 68) days for outpatients. Actual waiting times correlated with the waiting times requested by the referring physicians. However, only 37% of the procedures overall were completed within the requested waiting time. Of the 8030 patients, 50 (0.6%) died, 32 (0.4%) had a myocardial infarction and 41 (0.5%) experienced congestive heart failure. Overall, 109 patients (1.4%) had a major cardiac event, namely, death, myocardial infarction or congestive heart failure. These events occurred over a median wait of 27 days (2 days for inpatients and 35 days for outpatients), and over half (57%) occurred within the waiting time requested by the referring physician. In the multivariate analysis, predictors of the composite of death, myocardial infarction or congestive heart failure were increasing age (relative risk [RR] 2.39, 95% confidence interval [CI] 1.52–3.75) and New York Heart Association class III/IV symptoms (RR 2.86, 95% CI 1.11–7.33) in inpatients, and increasing age (RR 1.36, 95% CI 1.12–1.66), aortic stenosis (RR 3.70, 95% CI 1.93–7.08) and left ventricular ejection fraction less than 35% (RR 4.35, 95% CI 2.48–7.61) in outpatients.
Interpretation: Patients awaiting cardiac catheterization may experience major adverse events, such as death, myocardial infarction and congestive heart failure, which may be preventable. Our findings provide a benchmark by which to measure the effect of increased capacity and prioritization schemes that allow earlier access for patients at higher risk, such as those with aortic stenosis and reduced left ventricular function.
Cardiac catheterization is a key diagnostic procedure, providing information about coronary anatomy and structural cardiac abnormalities that is critical to making appropriate choices in the management of patients with heart conditions.1 It is one of the most frequently performed hospital-based procedures in adults. Owing to the combination of aging of the population and recent trends toward more aggressive investigation and treatment of heart disease, the demand for cardiac catheterization is expected to increase significantly over the next decade.2,3 The risks of waiting for other specialized cardiac procedures, such as bypass surgery, have been reported,4,5,6,7 but there is very little systematic information on the nature of waiting lists for cardiac catheterization and the rates of clinical events during the wait. The few previous reports are limited to physician-reported estimates or studies limited by small numbers and retrospective chart review.8,9,10,11,12,13
In this report, we describe the characteristics of a prospective waiting list registry of consecutive patients referred for cardiac catheterization over a 2-year period to a tertiary care regional centre serving 2.2 million people in southern Ontario. The specific aims of the study were to (1) document the mortality and morbidity associated with waiting for cardiac catheterization and (2) identify predictors of cardiac events that occur while patients are awaiting the procedure.
Methods
The Hamilton Health Sciences Corporation — General Site, Hamilton, Ont., is the sole provider of cardiac catheterization for most patients residing in the Central-South Region of Ontario. As of Apr. 1, 1998, all referrals for cardiac catheterization to our institution were done with a standardized referral form completed by the referring physician and sent by facsimile to the booking office. The information was edited with the Datafax system and included demographic information (including postal code), clinical variables that could relate to prognosis and the referring physician's requested waiting time for the patient (< 48 hours, < 7 days, < 14 days, < 42 days or < 90 days). Recent myocardial infarction (MI) was defined as any MI occurring within the 3 months before referral. The priority rating of patients in hospital at the time of referral (inpatients) who needed an urgent in-hospital procedure was determined by the booking office based on clinical urgency with advice from one of the angiographers. Patients at home (outpatients) were allocated to individual angiographers' offices if a specific angiographer was requested by the referring physician; if no such request was made, patients were assigned to the angiographers on a rotational basis. These patients were then scheduled for the procedure by the individual angiographers' offices.
All patients were followed until they underwent the procedure or it was cancelled or until the patient died. Additional events during the waiting period were captured in a standardized outcome form completed by the referring physician. These events included cancellation of the procedure, death, MI, congestive heart failure (CHF) and other hospital admission for a primary cardiac diagnosis. MI was defined as symptoms consistent with cardiac ischemia with either new electrocardiographic changes (Q waves, ST segment elevation, persistent ST segment depression) or elevation of cardiac enzyme levels.14,15 CHF was defined as symptoms and signs consistent with pulmonary edema (requiring admission to hospital for outpatients), with supporting radiologic findings if available. We assessed all reported major cardiac events (death, MI and CHF) through review of the medical chart or discussion with the referring physician where appropriate. The protocol was approved by the Hamilton Health Sciences Research Ethics Board.
Baseline data are expressed as means and standard deviations or proportions where appropriate. Given their highly skewed distribution, the data related to waiting times are presented as box plots with medians with 25th and 75th percentiles (interquartile range [IQR]). We compared groups using 𝛘2 tests for categorical variables. We used Spearman's coefficient of rank to compare differences between actual and requested waiting times, and we used the Cox proportional hazards model to identify independent predictors of major cardiac events (death, MI and CHF). Variables included in the univariate analyses were chosen for clinical importance and relevance. The multivariate analyses included all the variables to provide magnitude of effect adjusting for each variable. Symptoms classified according to the Canadian Cardiovascular Society functional classification16 and the New York Heart Association functional classification17 were handled as class III/IV versus class I/II based on a previous report that patients with higher grades of symptoms may be at higher risk for adverse events.18
Results
Between Apr. 1, 1998, and Mar. 31, 2000, 8030 patients (4725 outpatients with a mean age of 63.3 years and 3305 inpatients with a mean age of 64.4 years) were referred for cardiac catheterization. Most of the referrals (7345 [91.5%]) were for a primary diagnosis of suspected coronary artery disease (Table 1). Valvular disorders were suspected in 1177 cases (14.7%). Compared with outpatients, inpatients had higher rates of severe angina (Canadian Cardiovascular Society class III/IV) (87.9% v. 14.3%), New York Heart Association class III/IV symptoms (16.7% v. 8.9%), recent MI (48.8% v. 14.9%) and previous CHF (13.3% v. 8.4%). Noninvasive stress testing (exercise stress test or nuclear perfusion study) was performed in 3801 patients (47.3%), of whom 2379 (62.6%) were reported to be at high risk. Details of medications were collected after September 1998 and were available for 5981 consecutive patients. ASA was used in 4574 patients (76.5%), β-blockers in 3856 (64.5%), calcium channel blockers in 2295 (38.4%), nitrates in 3781 (63.2%) and 2 or more antianginal medications in 3531 (59.0%).
Table 1.
Angiography showed that 183 patients (2.5%) had more than 50% left main artery stenosis, 1939 (26.0%) had 3-vessel disease, 1347 (18.1%) had 2-vessel disease, 1480 (19.9%) had 1-vessel disease, 760 (10.2%) had mild disease and 1735 (23.3%) had normal coronary arteries (Table 2). Angiograms were coded as completely normal studies (i.e., no cardiac problem identified) for 1195 patients (16.0%).
Table 2.
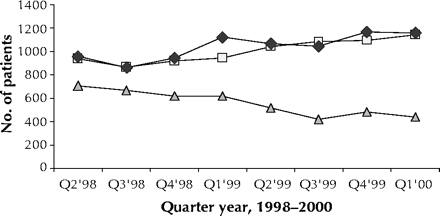
The number of referrals and the number of procedures performed increased over the study period (Fig. 1). During this period, the median waiting time was 20 (IQR 16) days for the overall population, 6 (IQR 4) days for inpatients and 60 (IQR 68) days for outpatients. There was a decrease in both the waiting times and the number of patients on the waiting list over the 2 years. These changes were coincident with an increase in the number of procedures because of the opening of a third catheterization suite, on Apr. 1, 1998, which became fully operational in September 1998.
Fig. 1: Trends in numbers of patients referred for cardiac catheterization (squares), numbers who underwent the procedure (diamonds) and numbers on the waiting list (triangles) in quarter-year intervals, Apr. 1, 1998, to Mar. 31, 2000.
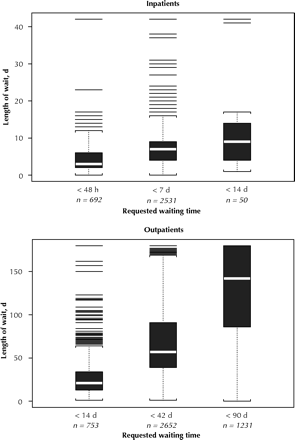
Actual waiting times correlated with requested waiting times (r = 0.87), with shorter waiting times for more urgent requests (Fig. 2). However, only 37% of the procedures overall were completed within the requested waiting time. Table 3 shows the median waiting times and proportion of patients who waited longer than the requested waiting time by baseline characteristics. Among outpatients, those with Canadian Cardiovascular Society class III/IV angina, those with recent MI and those with aortic stenosis had the shortest median waits; among inpatients there were no major differences among the various baseline variables. The median wait for patients referred by a physician who also performed cardiac catheterization was shorter for both inpatients (4 v. 6 days) and outpatients (38 v. 64 days). However, waiting times were similar for patients living within the city limits of Hamilton versus in surrounding communities (as determined through the postal code) (6 v. 6 days for inpatients and 64 v. 57 days for outpatients). Among inpatients, waiting times were similar at all hospitals whether comparisons were done by hospital size (median wait 6 days for hospitals with fewer than 100 acute care beds and those with 100–250 acute care beds, and 5 days for hospitals with more than 250 acute care beds) or number of patients referred per hospital (median wait 5 days for hospitals with fewer than 100 referrals, and 6 days for those with 100–250 referrals and those with more than 250 referrals).
Table 3.
Fig. 2: Median number of days from referral to cardiac catheterization or cancellation of procedure, by requested waiting time, for inpatients and outpatients. The bottom of each box represents the 75th percentile; thus, the height of the box is the interquartile range (IQR). The white line represents the median. Values that lie more than 1.5 IQRs beyond the top or bottom of the box (outliers) are shown as individual lines. The square brackets connected by dotted lines to the top and bottom of the box represent the most extreme values not more than 1.5 IQRs beyond the box. For inpatients, the median waiting time and proportion who underwent catheterization within the requested waiting time were as follows: < 48 hours: 3 (IQR 4) days, 48%; < 7 days: 6 (IQR 4) days, 50%; < 14 days: 7 (IQR 7) days, 82%. The corresponding values for outpatients were: < 14 days: 21 (IQR 18) days, 28%; < 42 days: 56 (IQR 44) days, 29%; < 90 days: 133 (IQR 88) days, 29%.
Cancellation requests were made for 586 patients (7.3%), of whom 47 underwent the procedure elsewhere (Table 4). Of the 8030 patients, 50 (0.6%) died, 32 (0.4%) had an MI and 41 (0.5%) experienced CHF. Overall, 109 patients (1.4%) had a major cardiac event, that is, death, MI or CHF (33 inpatients [1.0%] and 76 outpatients [1.6%]). Two deaths were reported as due to noncardiac causes. Most of the remaining 48 deaths were sudden unexpected events and were considered most likely to have been secondary to cardiac causes. The median time from referral to a major adverse cardiac event was 2 (IQR 3) days for inpatients and 35 (IQR 46) days for outpatients (Fig. 3). In the multivariate analysis, predictors of the composite of death, MI or CHF were increasing age (relative risk [RR] 2.39, 95% confidence interval [CI] 1.52–3.75) and New York Heart Association class III/IV symptoms (RR 2.86, 95% CI 1.11–7.33) for inpatients, and increasing age (RR 1.36, 95% CI 1.12–1.66), aortic stenosis (RR 3.70, 95% CI 1.93–7.08) and left ventricular ejection fraction less than 35% (RR 4.35, 95% CI 2.48–7.61) for outpatients (Table 5).
Table 5.
Table 4.
Interpretation
In this prospective registry of over 8000 patients referred for cardiac catheterization in 1998–2000 in the Central-South Region of Ontario, the median waiting time was 6 days for inpatients and 60 days for outpatients. Only 37% of patients received the procedure within the time requested by the referring physician. The overall incidence of major cardiac events was 1.4%, with 50 deaths during a median wait of 27 days.
In a retrospective study of 871 patients in Manitoba referred for cardiac catheterization in 1981–1982, the incidence rates of cardiac arrest, acute MI, death and emergency admission during a mean waiting time of 4.2 weeks were 0.5%, 0.9%, 0.4%, and 3.7% respectively.10 In an observational cohort study of 381 adult outpatients placed on a waiting list at a public hospital in Galveston, Tex., in 1993–1994, 36 patients (9.4%) experienced adverse events, including 6 deaths (1.6%), 4 MIs (1.0%) and 26 admissions to hospital (6.8%) for angina or CHF, over a mean follow-up period of 8.4 months.13 In a single-centre prospective cohort study of 357 adult patients referred for cardiac catheterization in 1997, 22 patients (6.2%) experienced adverse events: 4 patients (1.1%) died, 2 (0.6%) had an MI, and 16 (4.5%) required a more urgent procedure or hospital admission.11 Finally, in a retrospective study of 696 inpatients referred in the greater Toronto area in 1997–1998, the adverse event rate was 1.6%, with 6 deaths (0.9%) and 3 nonfatal MIs (0.4%) during a mean wait of 5.7 days.12 Our finding of a major cardiac event rate of 1.4% is consistent with these data.
Most of the deaths reported in our study were sudden unexpected events that were probably secondary to cardiac causes. The death rate observed in our study was on average 6 times higher than the expected procedural death rate of less than 0.1%.1 Furthermore, about one-third of patients undergoing cardiac catheterization will need to join the waiting list for bypass surgery and face a waiting list mortality risk of 0.4% in Ontario.6
We found left ventricular ejection fraction less than 35% and aortic valve stenosis to be strong predictors of adverse outcomes in outpatients. These findings are consistent with previous analyses of mortality risk in patients waiting for cardiac surgery.6 They are also consistent with the data from randomized studies of cardiac surgery versus medical therapy, which showed that left ventricular dysfunction was a predictor of adverse events in patients who received medical therapy.19 It is therefore possible that some of the deaths in our study could have been prevented with earlier catheterization and expedited referral for surgical revascularization.
In our study, half of the adverse events occurred within 2 days of referral for inpatients and within 35 days of referral for outpatients, with a substantial number of these events occurring within the waiting time requested by the referring physician. These findings are consistent with a previous report demonstrating that a substantial number of adverse events occurred within 3 to 13 weeks after referral.13 The categories that we adopted for requested waiting time relate closely to previously published urgency rating categories for coronary revascularization in Ontario and other jurisdictions.20,21,22,23 In the absence of formal queuing criteria and standards for maximal acceptable waiting times, the requested waiting time may serve as an estimate of urgency.
Addition of catheterization suites alone may not reduce the number of adverse events during the wait for this procedure.24,25,26,27,28 Other important goals would be to develop booking systems that (1) specify minimal acceptable clinical criteria by which patients are placed on a waiting list for cardiac catheterization,22 (2) can reliably identify and prioritize patients at high risk, ensuring timely access to the procedure for them, (3) can effectively monitor and adapt to the changes in patients' status and (4) specify maximal waiting times for patients at low risk to avoid undue suffering.
In our study, 16.1% of patients had normal angiograms. Clarification of coronary anatomy in a timely manner may offer benefits related to patient management, such as selection of medical therapy, issues surrounding return to work and improved quality of life. In addition, the rate of normal studies may serve as a measure of appropriateness of case selection, referral patterns and resource use.29,30,31,32
Possible limitations to our study include the following. First, our study documented only patients referred to our centre for diagnostic catheterization. During the study period, patients from our region were also referred to other centres outside our region. However, the consecutive nature of referrals and the longitudinal cohort design in which each patient was followed until a prespecified end point (i.e., procedure, cancellation or death) provide sufficient validity for interpreting the results.33,34 Second, it is possible that the clinical severity of patients' conditions was overestimated by the referring physicians in the hope of shortening the wait. To reduce bias we limited our analyses of outcomes to major end points, such as death, MI and CHF. Third, it is possible that the true number of events during the wait for cardiac catheterization was underestimated. We relied on reporting of events by the referring physicians. We increased the accuracy of determination of events by interviewing the patients on the day of the procedure. Fourth, factors other than angiographic capability may have influenced waiting times. Significant differences between angiographers, such as amount of time available for procedures, size of clinical practice, length of waiting list and productivity, were not systematically documented. Finally, medical consequences are only part of the overall burden of the wait. Other consequences of waiting, such as effect on quality of life and potentially wasteful use of resources (e.g., emergency departments visits, unscheduled family doctor visits and unnecessary noninvasive testing) during the wait were not systematically measured.
Our findings provide a benchmark by which to assess ongoing initiatives to increase capacity and reduce maximal waiting times for all patients. They can also guide the development of formalized systems for managing waiting lists for cardiac catheterization that offer more rapid access for patients at higher risk, as well as flexibility to accommodate periodic shifts in caseload and urgency.
𝛃 See related article page 1247
Footnotes
-
This article has been peer reviewed.
Contributors: Dr. Natarajan was the principal investigator and was involved in study conception, data acquisition, analysis and interpretation, and drafting the manuscript. Dr. Mehta contributed to data analysis and interpretation and critical revision of drafts of the manuscript. Dr. Holder contributed to study conception and design and critical revision of drafts of the manuscript. Dr. Goodhart contributed to the design of the case report forms, data acquisition and revision of drafts of the manuscript. Dr. Gafni contributed to study conception, data interpretation and revision of drafts of the manuscript. Mr. Shilton contributed to data acquisition and interpretation and revision of drafts of the manuscript. Mr. Afzal contributed to data analysis and interpretation and drafted portions of the manuscript. Dr. Teo contributed to data analysis and interpretation and revision of drafts of the manuscript. Dr. Yusuf contributed to study conception and design, and data analysis and interpretation, and drafted portions of the manuscript.
Acknowledgements: We thank Cheryl Christmas for her assistance with data management, Dr. Tef Kalamparambath and Dr. Jeffrey Pang for reviewing the coronary angiograms, and Dr. John B. Gill and Dr. Charanjit S. Rihal for reviewing the manuscript.
This study was supported by grant RCI-0240-06 from the Canadian Health Services Research Foundation in partnership with the Ontario Ministry of Health and the Hamilton Health Sciences Cardiac and Vascular Program.
Competing interests: None declared.