Abstract
Background: Practice guidelines recommend that imaging to detect metastatic disease not be performed in the majority of patients with early-stage breast cancer who are asymptomatic. We aimed to determine whether practice patterns in Ontario conform with these recommendations.
Methods: We used provincial registry data to identify a population-based cohort of Ontario women in whom early-stage, operable breast cancer was diagnosed between 2007 and 2012. We then determined whether imaging of the skeleton, thorax, and abdomen or pelvis had been performed within 3 months of tissue diagnosis. We calculated rates of confirmatory imaging of the same body site.
Results: Of 26 547 patients with early-stage disease, 22 811 (85.9%) had at least one imaging test, and a total of 83 249 imaging tests were performed (mean of 3.7 imaging tests per patient imaged). Among patients with pathologic stage I and II disease, imaging was performed in 79.6% (10 921/13 724) and 92.7% (11 882/12 823) of cases, respectively. Of all imaging tests, 19 784 (23.8%) were classified as confirmatory investigations. Imaging was more likely for patients who were younger, had greater comorbidity, had tumours of higher grade or stage or had undergone preoperative breast ultrasonography, mastectomy or surgery in the community setting.
Interpretation: Despite recommendations from multiple international guidelines, most Ontario women with early-stage breast cancer underwent imaging to detect distant metastases. Inappropriate imaging in asymptomatic patients with early-stage disease is costly and may lead to harm. The use of population datasets will allow investigators to evaluate whether or not strategies to implement practice guidelines lead to meaningful and sustained change in physician practice.
Most women with newly diagnosed breast cancer present with early-stage, potentially curable disease.1 Among patients whose disease is restricted to the breast and axillary lymph nodes, without signs or symptoms of metastatic disease, the likelihood of having radiologically evident metastases in pathologic stage I and II disease is about 0.2% and 1.2%, respectively.2 This low frequency has not changed significantly, even with the increasing use of magnetic resonance imaging (MRI) and positron emission tomography.2,3 For this reason, most provincial, national and international guidelines do not recommend imaging for all patients with early-stage breast cancer who are asymptomatic.4–8
Despite these evidence-based guidelines, imaging for distant metastases in patients with a new diagnosis of breast cancer remains common.2,9–12 In response to the Choosing Wisely campaign of the American Board of Internal Medicine Foundation,13 the American Society of Clinical Oncology (ASCO) published its inaugural “top 5” list for choosing wisely in oncology.14 It recommended against routine imaging for staging purposes in women with early breast cancer, because “such imaging adds little benefit to patient care and has the potential to cause harm.”14 In 2014, Choosing Wisely Canada was launched in an effort to encourage physicians and patients to engage in conversations about unnecessary tests, treatments and procedures, to help ensure that patients receive the highest-quality care.15
The ASCO Choosing Wisely recommendation14 is similar to the Cancer Care Ontario guideline,4 which has been in existence for over a decade. Whereas ASCO in its Choosing Wisely campaign recommends no imaging for patients with stage I or II disease, the Cancer Care Ontario guideline recommends no imaging for patients with stage I disease and a bone scan for those with stage II disease. A recent study at a large Canadian academic cancer centre showed that, despite publication of both a provincial guideline and the ASCO recommendations, most patients with primary operable (early-stage) breast cancer undergo imaging for distant metastases.10 We hypothesized that despite the provincial guideline, this practice may be more widespread. We undertook this population-based study to determine whether physician practice patterns in Ontario regarding imaging of patients with early-stage breast cancer are in keeping with the published Cancer Care Ontario guideline.
Methods
We aimed to determine the extent and type of radiologic imaging that is being performed for assessment of distant metastases in patients with asymptomatic stage I or II breast cancer and to identify regional differences and factors that may be associated with a higher likelihood of imaging.
Study population
This retrospective, population-based cohort study was approved by the Sunnybrook Health Sciences Centre Research Ethics Board. We linked data from population-based datasets available through the Institute for Clinical Evaluative Sciences to capture perioperative (i.e., before and after definitive breast cancer surgery) imaging studies for patients with primary operable (early-stage) breast cancer in Ontario. Complete study methods are provided in Appendix 1 (available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.150003/-/DC1), and health codes are available in Appendix 2 (available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.150003/-/DC1).
We used the Ontario Cancer Registry to identify all women with a diagnosis of stage I (I, IA and IB) or stage II (II, IIA and IIB) breast cancer (Table 1)16 between 2007 and 2012, with 2012 being the last year that data from the Ontario Cancer Registry were available at the Institute for Clinical Evaluative Sciences. The cancer stage recorded in the registry is determined from electronic medical records after all of the patient’s results have been received; as such, staging as recorded in the registry is considered the “best available” stage.
Description of breast cancer staging16
We excluded patients who had a prior breast cancer diagnosis and those who had stage 0, III or IV disease, or null or unknown stage disease. We also excluded cases of ductal carcinoma in situ because the Institute for Clinical Evaluative Sciences does not receive information about such cases from the Ontario Cancer Registry.
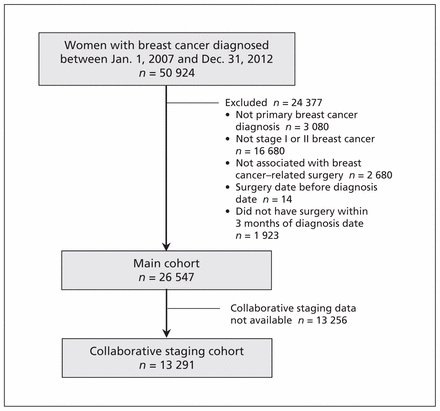
The registry cohort was linked with the Discharge Abstract Database of the Canadian Institute for Health Information to identify patients who underwent breast-related surgery. To identify patients with primary operable disease, we restricted the study population to patients with a first diagnosis of breast cancer who underwent definitive surgery within 3 months of the tissue diagnosis date. This window was selected to exclude patients who might have received preoperative systemic therapy for initially inoperable, locally advanced disease, since contemporary preoperative chemotherapy regimens are a minimum of 4–5 months in duration.17 The plan for creating the cohort dataset is shown in Figure 1.
Creation of a population-based cohort to investigate the frequency of imaging for distant metastases among women with early-stage breast cancer.
Factors associated with imaging
To identify factors associated with utilization of imaging, patient-level data were collected for age at diagnosis, disease stage, histologic findings (characterized as ductal, lobular or other), comorbidity (calculated using the Deyo modification of the Charlson comorbidity index18 and characterized as 0, 1–2 or ≥ 3), surgery type, whether additional preoperative locoregional imaging beyond mammography was performed (either breast ultrasonography or breast MRI), and neighbourhood income quintile based on Postal Code Conversion File Plus.19 Information about institution type, rurality of the patient’s residence (based on the Rurality Index for Ontario 2008)20 and the Local Health Integration Network (LHIN) of the hospital performing the surgery was also collected. Ontario is divided geographically into 14 LHINs, which administer and coordinate local health systems.21 For additional disease characteristics not available in the Ontario Cancer Registry (i.e., tumour grade [1, 2 or 3], human epidermal receptor-2 [HER2] status [positive or negative], presence of triple-negative disease [i.e., negative for estrogen receptors, progesterone receptors and HER2 status; yes or no], involvement of axillary lymph nodes [yes or no] and presence of lymphovascular invasion [yes or no]), we linked to the Ontario Cancer Registry’s Collaborative Staging dataset.
Imaging quantification and classification
To quantify all perioperative imaging performed for the purpose of staging, we linked the cohort of patients with primary operable breast cancer with the Ontario Health Insurance Plan (OHIP) database to obtain all imaging fee codes (see Appendix 2, Part 2) associated with these patients. Imaging was considered preoperative if it occurred between the tissue diagnosis date and the day of surgery; imaging was considered postoperative if it occurred within the 3-month window after the definitive breast surgical procedure. All imaging tests were classified by site (skeleton, thorax, abdomen and/or pelvis, other) and modality.
We further classified imaging tests as initial imaging or confirmatory imaging. Initial imaging was defined as the first imaging test performed on a body site (i.e., skeleton, thorax, abdomen and/or pelvis, other) in the prespecified time period. Confirmatory imaging was defined as any additional imaging test performed on a body site that had already been imaged. We did not count any imaging of the same anatomic body area beyond the first confirmatory scan in the total imaging counts. We adopted this conservative approach in an effort to reduce the chance of including further imaging unrelated to breast cancer staging. To determine who ordered the imaging test, we extracted the referring physician’s self-reported main specialty associated with the OHIP imaging fee code.
Statistical analysis
We calculated the total numbers of initial and confirmatory imaging tests for patients with stage I or II breast cancer, considering both the number of patients imaged and the number of imaging tests performed. Initial and confirmatory imaging was further classified into pre- and postoperative imaging. All initial and confirmatory imaging tests were quantified by body site and imaging modality. To assess whether the use of imaging modalities changed over time, the proportion of patients imaged each year was calculated by body site and across all body sites, considering initial and confirmatory imaging separately. Trends were evaluated using the Cochran–Armitage test.
To identify characteristics potentially associated with imaging for metastatic disease, we used multivariable logistic regression to examine the association between the covariables and imaging at any site (skeleton, thorax, abdomen and/or pelvis, other) and at each site individually For the purpose of this analysis, imaging for metastases did not include any imaging performed on the breast, such as ultrasonography, MRI or mammography. All statistical analyses were performed using SAS software, version 9.3 (SAS Institute).
Results
Patient characteristics
Between 2007 and 2012, a total of 26 547 women in Ontario were identified with stage I (51.7%) or stage II (48.3%) breast cancer (Table 2). Eighty percent of the patients were at least 50 years old. Locoregional staging with breast MRI was performed in 23.0% of the patients, and 74.6% underwent breast-conserving surgery.
Characteristics of women with early-stage breast cancer treated with surgery in Ontario, 2007 to 2012
Imaging performed
Of the 26 547 patients, 22 811 (85.9%) had at least one imaging test for distant metastatic disease (Table 3 and Appendix 3, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.150003/-/DC1). Imaging was performed in 10 921 (79.6%) and 11 882 (92.7%) of women with stage I and II disease, respectively. The total number of imaging tests was 83 249. Overall, 76.2% of these (n = 63 465) were classified as initial imaging, and 23.8% (n = 19 784) were classified as confirmatory imaging. On average, 3.6 (standard deviation 1.9) imaging tests were performed per patient who underwent imaging. Nearly half of the initial imaging was performed in the preoperative period (48.7%, 30 907/63 465), whereas most of the confirmatory imaging was performed postoperatively (79.1%, 15 647/19 784).
Initial and confirmatory imaging for women with early-stage breast cancer treated with surgery in Ontario, 2007 to 2012
Provider characteristics
In the preoperative setting, initial imaging was ordered mainly by the surgeon (73.9% of imaging tests), the primary care physician (19.9%), the medical oncologist (1.1%) or the radiation oncologist (0.3%) (Table 4). Postoperatively, the relative proportions of initial imaging ordered by surgeons (42.6%) and primary care physicians (11.4%) declined, and the proportions ordered by medical oncologists (35.0%) and radiation oncologists (6.2%) increased.
Main specialty of referring physician
Imaging trends over time
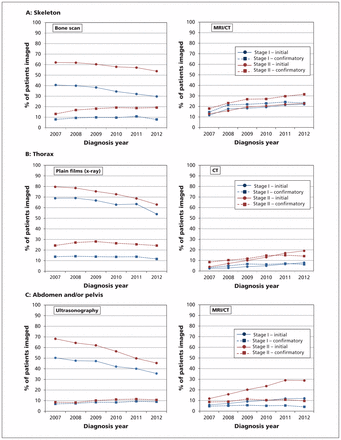
The overall total proportion of patients with breast cancer who underwent conventional and advanced imaging remained relatively stable over the study period; however, there was a trend toward the use of more advanced imaging (i.e., computed tomography [CT] and MRI) and a decline in the use of conventional imaging over time. Figure 2 contrasts conventional and advanced imaging modalities for each body site. For example, the use of thoracic radiography for initial imaging of patients with stage II cancer (Figure 2B) declined from 80% in 2007 to 63% in 2012, whereas the use of thoracic CT increased steadily, from 3% to 19% over the same period (trend for all stages, p < 0.001). A similar pattern was observed for abdominal and pelvic imaging (Figure 2C), where the use of ultrasonography for initial imaging declined, while use of CT increased (trend for all stages, p < 0.001) (see Appendix 4, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.150003/-/DC1). Overall, use of advanced imaging (i.e., isotope bone scans, CT, MRI, positron emission tomography) to look at potential sites of metastasis represented 40.6% (25 776) of initial imaging tests; see Appendix 3 for details.
Yearly imaging trends for skeleton (A), thorax (B) and abdomen and/or pelvis (C) by conventional imaging (at left) and more advanced imaging (at right). CT = computed tomography, MRI = magnetic resonance imaging.
Factors associated with imaging
Multivariable analysis was performed on the subset of 13 291 patients for whom collaborative staging data were available (50.1% of full study cohort). Associated odds ratios (OR) and 95% confidence intervals are presented in Table 5. A number of patient-related (younger age, greater comorbidities), disease-related (higher-stage tumour, negative HER2 status, involvement of axillary lymph nodes, lymph or vascular invasion, locoregional breast staging, imaging beyond mammography [i.e., ultrasonography], non–breast-conserving surgery) and system-related (surgery performed in the community) factors were associated with greater use of imaging. We also found significant associations between geographic status (by LHIN) and use of imaging, both overall and by body site.
Estimated odds of imaging at specific body sites or all sites, for women with collaborative staging data
To verify that there were no significant differences between cohorts, we performed multivariable analysis for both cohorts, using only those variables available for the full cohort. The results were not significantly different (data not shown).
Interpretation
Despite guidelines against imaging to detect radiologically evident distant metastases, our results show that this practice is very common among patients with early-stage breast cancer in Ontario.4–6,8 The reasons for this disconnect between evidence and practice are likely attributable to a combination of patient-related11 and physician-related22 factors. It is not surprising, given the low pretest probability of detecting disease, that ASCO highlighted this practice disparity in its inaugural “top 5” list for choosing wisely in oncology.14 The ASCO document stresses that the routine use of imaging for staging in asymptomatic patients with early-stage disease does not extend survival, is costly and may lead to harm (since false-positive results may lead to invasive procedures and overtreatment), all of which can impair quality of life.
A recent single-centre study showed that the ASCO publication had no impact on the overuse of imaging for staging purposes.10 Our current study confirms that nonadherence with evidence-based guidelines is common across Ontario. According to guidelines, asymptomatic patients with stage I disease should not undergo imaging, but such imaging was performed for 79.6% of the patients in our cohort. In stage II disease, where the ASCO document recommends no imaging, such imaging was performed in 92.7% of patients. An additional concern is that 23.8% of all imaging performed was confirmatory radiography of a body site that had already been imaged.
The current dataset was large enough to allow us to evaluate factors associated with increased imaging. Some of these factors may reflect characteristics associated with more aggressive tumours and therefore a higher pretest probability of detecting asymptomatic metastatic disease (e.g., younger patients, higher-stage disease, mastectomy). However, for other variables associated with increased use of imaging, such as more comorbidities, negative HER2 status, increased locoregional breast imaging and surgery in a community setting, there is no biological rationale for increased imaging.
Our findings, which are in keeping with those of other studies, raise the question of how to enhance adherence to evidence-based practice guidelines. Similar to another study, performed in the United States,23 we have shown that a range of patient, tumour and treatment characteristics are associated with increased imaging use, which may be useful in improving adherence. For example, certain adverse tumour characteristics such as stage, involvement of the axillary lymph nodes, lymphovascular invasion, negative HER2 status and tumour grade were all associated with increased utilization of imaging. This information is potentially useful because subgroups of patients with early-stage breast cancer who have these risk factors may have a higher rate of metastatic disease. However, at present, none of the commonly used guidelines include these characteristics, other than cancer stage, in their recommendations.4–8 The finding of significant variability in utilization across geographic regions and between community and academic centres may also represent gaps in guideline awareness, as well as opportunities for homogenization of practice across Ontario.
In general, physicians tend to be receptive to guidelines. Most believe that guidelines are a helpful source of advice, a valuable educational tool, a means for improving the quality of care and sufficiently practical to apply to their patients; however, guidelines do not reflect the potential legal and ethical implications of variable (nonadherent) practices.22,24,25 Additional work is needed to identify reasons why guidelines pertaining to perioperative imaging are not being followed. The factors evaluated should include factors related to patients,22 physicians26 and funding agencies.27
The strengths of the current study included use of a large cohort representing all women with primary operable breast cancer in Ontario. The dataset is likely to be representative, since all costs of imaging are covered by the provincial health care plan, and Ontario has a comprehensive collection of linked health administrative databases allowing linkage of patient data from diagnosis to postsurgical care.28–31 Furthermore, the Ontario Cancer Registry includes all breast cancer cases in Ontario.
Limitations
This study had several limitations. First, the data did not allow for determination of whether patients were symptomatic or whether symptoms developed within the 3-month period after surgery. However, the literature and clinical experience suggest that most patients undergoing surgery for newly diagnosed breast cancer are asymptomatic.10 Second, we could not determine the specific indication for imaging, and some tests may have been ordered for reasons other than breast cancer staging. Third, tumour characteristics for the full cohort were not available, and regression modelling was performed for the collaborative staging cohort only (about half of the total study cohort). To verify that there were no significant differences between cohorts, we performed multivariable analysis for both cohorts, using only those variables available for the full cohort, but the results were not significantly different. Fourth, because the data were contemporary, we were unable to compare longer-term outcomes such as survival between patients who underwent imaging and those who did not. Such a comparison represents an opportunity for future studies, along with an evaluation of the cost-effectiveness of imaging.
Another limitation of the current study is that while the 2001 Cancer Care Ontario guideline recommends isotopic bone scanning for patients with stage II disease, we have continued to consider this type of scanning as inappropriate, partly because more recent guidelines and the ASCO Choosing Wisely campaign do not recommend bone scans for patients with stage II disease. In addition, the methodology of the current study means that if the data for bone scans for patients with stage II cancer were removed from the analysis, then the number of initial skeletal radiography tests performed for patients with stage II disease would increase from 688 to 2021 and the number of initial skeletal MRI tests would increase from 357 to 905.
Conclusion
Most women in Ontario with primary operable (early-stage) breast cancer undergo imaging to look for distant metastases, despite guidelines recommending against this practice.
The use of this imaging for women with stage I and II breast cancer, for whom evidence suggests that it is unnecessary, may be due in part to the lack of consensus among clinicians about the guidelines themselves. Further discussions about the appropriateness of guidelines in light of modern imaging techniques may be needed, especially as the data for the current study were collected from Jan. 1 2007, to Dec. 31, 2012. However, if guideline recommendations are to be implemented in practice, additional knowledge translation strategies are needed, as dissemination of clinical practice guidelines alone is not an effective method of changing physician practice behaviours.32 For example, identification of factors associated with imaging could allow targeted strategies to improve adherence. These strategies will also require patient engagement. The use of provincial datasets allows a unique opportunity to evaluate whether such strategies are effective at the broader population level.
Footnotes
CMAJ Podcasts: editor’s summary at soundcloud.com/cmajpodcasts/150003-res
Competing interests: None declared.
This article has been peer reviewed.
Contributors: All authors contributed to the conception and design of the study. Demetrios Simos and Christina Catley contributed to acquisition of the data, and all authors contributed to analysis and interpretation of the data. All authors reviewed and approved the final draft of the manuscript and agree to act as guarantors of the work.
Funding: This project was funded by a grant from the Patient Quality and Safety Committee, Department of Medicine and Division of Medical Oncology, University of Ottawa, Ottawa, Ont.
Disclaimer: This study was supported in part by the Institute for Clinical Evaluative Sciences (ICES), which is funded by an annual grant from the Ontario Ministry of Health and Long-Term Care (MOHLTC). The opinions, results and conclusions reported in this paper are those of the authors and are independent from the funding sources. No endorsement by ICES or the Ontario MOHLTC is intended or should be inferred.
- Received June 22, 2015.
- Accepted May 7, 2015.











Podcast