Recent data suggest that almost 10% of patients who undergo coronary artery bypass grafting (CABG) are readmitted to hospital within 30 days of surgery.
Patients who lack primary care follow-up have been shown to have worse long-term outcomes.
Many complications after CABG surgery are related to the systemic inflammatory response associated with cardiopulmonary bypass.
Primary care practitioners play an integral role in recognizing complications of CABG and completing postoperative care.
About 15 000 people in Canada undergo coronary artery bypass grafting (CABG) surgery each year.1 According to the 2019 Canadian Institute for Health Information Cardiac Care Quality Indicators Report, 9.4% of these patients are urgently readmitted to hospital within 30 days of CABG surgery.1 As the Canadian population continues to age, cardiac surgery is being offered to older, sicker and more frail patients; such patients have higher rates of readmission to hospital after surgery.2 A retrospective study showed 30-day readmission rates of 15%–20% in patients older than 80 years undergoing cardiac surgery at a single centre.2 Furthermore, a lack of outpatient primary care practitioner visits within 30 days after discharge was associated with a fivefold increased risk of poor 1-year functional survival (hazard ratio 5.18, p < 0.01).2 These findings highlight the important role that primary care practitioners play in the management of patients following CABG surgery.
Most CABG surgeries are performed using cardiopulmonary bypass, which uniquely affects patients’ physiology and leads to specific postoperative complications in some patients. Cardiopulmonary bypass facilitates a motionless and bloodless surgical field, while maintaining blood oxygenation and tissue perfusion by circulating the patient’s venous blood through a mechanical oxygenator. However, contact of the blood with the nonendothelialized surfaces of the bypass circuit triggers an intense systemic inflammatory response syndrome (SIRS), and leads to activation of the coagulation system. Although heparin is routinely given to mitigate clotting, SIRS also leads to massive fluid shifts, dysregulation of capillary beds and, consequently, tissue hypoperfusion despite fluid retention.3 Hypoperfusion related to SIRS, combined with micro-emboli, can lead to organ system damage and postoperative morbidity. Patients may develop chest pain, shortness of breath or dysregulation of blood pressure in the weeks after surgery secondary to SIRS.
We discuss the management of postoperative clinical syndromes and potential early and late complications that primary care practitioners may encounter. Important medication and activity considerations, based on best practice guidelines and current evidence (Box 1), are also highlighted. It is important to note that CABG performed without cardiopulmonary bypass (“off-pump”) is still a major surgery and patients may have many of the postoperative complications discussed in this review.
Evidence used in this review
Literature and guideline searches relating to cardiac surgery and postoperative management were performed using Google Scholar, PubMed, the Canadian Cardiovascular Society, the American College of Cardiology/American Heart Association, and the European Society of Cardiology/European Association for Cardio-Thoracic Surgery. Search terms included “cardiac surgery,” “postoperative management,” “coronary artery bypass grafting” and complications (e.g., “atrial fibrillation,” “chest pain”). The most recent and robust guidelines, systematic reviews, randomized controlled trials, cohort studies and case reports were used to inform the review. To describe some common practices and pathophysiology, relevant cardiac surgery textbooks were referenced as well.
What causes chest pain after cardiac surgery?
It is helpful to consider the 3 “I”s (infectious, inflammatory and ischemic) when considering important causes of chest pain after cardiac surgery. Pain is often related to mechanical trauma from the sternotomy, in which case it is localized and exacerbated by movement. A palpable click suggests mediastinal instability, which should prompt referral back to the cardiac surgeon. Pain may be related to wound infection, which also leads to localized pain, along with characteristic signs of infection. Nerve damage while harvesting the internal mammary artery for grafting purposes may result in neuropathic parasternal hypo- or hyperesthesia. Pericarditis (inflammatory pain) and myocardial ischemia are considered in more detail below.
Pericarditis
Patients with pericarditis often report vague anterior chest pain. There may be an associated audible pericardial rub on physical examination with diffuse ST elevation or PR depression on electrocardiogram (ECG).
Postpericardiotomy syndrome is an immune pericarditis that can develop weeks to months after cardiac surgery.4,5 According to the 2015 European Society of Cardiology Guidelines for the Diagnosis and Management of Pericardial Diseases, its clinical features include unexplained fever, pericardial or pleural rubs, new or worsening pleural or pericardial effusion (although this is not present in all), and pericarditic or pleuritic chest pain.6 This guideline recommends ordering an echocardiogram early after diagnosis to determine the presence of a pericardial effusion, which would require serial follow-up imaging.6 Treatments include nonsteroidal anti-inflammatory drugs (NSAIDs), colchicine and steroids as single agents or combination therapy, as the clinical scenario demands.6,7 The primary cardiac surgeon should be informed, as drainage of effusions may be required.
Postoperative cardiac tamponade is a rare complication of postpericardiotomy syndrome that most commonly presents early (< 7 d after surgery), but can also occur late (> 7 d) in 1%–12.6% of patients.8 Cardiac tamponade presents with Beck triad (muffled heart sounds, jugular venous distention and hypotension) as well as sinus tachycardia, pulsus paradoxus, electrical alternans on ECG, and enlargement of the cardiac silhouette on chest radiograph.9 Late cardiac tamponade is associated with high mortality rates and requires transfer of the patient to the emergency department and emergent intervention.10
Myocardial infarction after CABG
Occlusion or stenosis of bypass grafts or nonbypassed coronary arteries (caused by disease progression or because severely diseased coronary arteries were nongraftable or too small) can lead to ischemic chest pain after CABG surgery. This chest pain can be typical or atypical. Ischemia can also be painless, which is commonly seen preoperatively in patients with diabetes,11 but sometimes in patients who do not have diabetes, owing to disruption of the autonomic cardiac plexus during surgery.
Early perioperative myocardial infarction occurs in 0.6% to 19% of cases after CABG, depending on the serological criteria used.12 The Synergy Between Percutaneous Coronary Intervention with Taxus and Cardiac Surgery (SYNTAX) trial, which compared percutaneous coronary intervention and CABG for the treatment of patients with left main coronary artery disease, reported rates of late myocardial infarction of 3.8% (syntax score ≤ 32) and 6.1% (syntax score ≥ 33) at 5 years after CABG.13 A 2016 Cochrane systematic review estimated that the graft occlusion rate after CABG is about 2%–5% per year.14 To prevent graft occlusion, the Canadian Cardiovascular Society 2012 focused update of the guideline for the use of antiplatelet therapy recommends dual antiplatelet therapy with acetylsalicylic acid and clopidogrel or ticagrelor, for 1 year, after CABG for acute coronary syndrome. This recommendation was unchanged in the 2018 update.15
Because there are no specific guidelines describing the management of stable angina or acute coronary syndrome after CABG, recurrent ischemic chest pain after CABG is diagnosed and managed similarly to the first presentation of ischemic heart disease before CABG. Any patient presenting with symptoms and signs of acute coronary syndrome should be managed promptly in the nearest emergency department. Stable angina with medical management should include aggressive control of cardiac risk factors such as hypertension, diabetes mellitus, obesity, dyslipidemia and smoking, as proposed by the 2015 American Heart Association Scientific Statement on secondary prevention after CABG.16 Patients with stable angina should be referred back to their cardiologist.
What causes shortness of breath after cardiac surgery?
In the immediate postoperative period, most patients have dyspnea, with or without hypoxia, owing to pain-induced splinted breathing or atelectasis or both. This is managed aggressively with analgesia, early mobilization, incentive spirometry and physiotherapy.17 New dyspnea may occur after patients are discharged, and important causes that should be excluded are (from most to least likely) pleural effusion, atelectasis, congestive heart failure, pneumonia and pulmonary embolism.
Pleural effusion
Owing to intraoperative factors outlined above, patients usually require substantial fluid administration and vasopressors to maintain adequate systemic perfusion postoperatively, which results in movement of fluid from the intravascular to the interstitial compartment (third spacing). As patients stabilize, fluid mobilizes from the interstitial to the intravascular space and diuretic therapy aids the return to euvolemia, often continuing after discharge. Not uncommonly, patients are discharged with small pleural effusions that improve spontaneously because of resolving SIRS and ongoing diuresis. However, new or re-accumulation of pleural effusions may occur, most commonly due to ongoing fluid overload (e.g., from inadequate diuresis or congestive heart failure). Less common causes include serositis, bleeding (usually in patients on anticoagulation) and, rarely, chylothorax. The underlying cause should be established and treated; e.g., restriction of oral intake and increasing diuresis in the case of fluid retention and treatment with anti-inflammatories for serositis. Discussion with the patient’s cardiac surgeon will facilitate appropriate management in case invasive treatment is required.
Atelectasis
If patients remain mobile after discharge, recurrence of atelectasis is rare. However, if patients are sedentary after discharge, or have ongoing chest pain that results in hypoventilation, atelectasis may redevelop after discharge and will be apparent on chest radiograph. Patients should be given appropriate analgesia with investigation of underlying causes of pain, advised to mobilize and referred to physiotherapy.
Congestive heart failure
According to large retrospective studies, about 13% of readmissions to hospital after CABG are a result of heart failure, with higher rates seen in patients with low ejection fraction before surgery.18,19 Preoperative irreversible myocardial damage, intraoperative myocardial injury, residual lesions in coronary arteries leading to ongoing ischemia, diastolic dysfunction or hibernating myocardium after CABG are possible causes. Patients should be referred to cardiology services for medical management in accordance with the 2017 Comprehensive Update of the Canadian Cardiovascular Society Guidelines for the Management of Heart Failure20 — the mainstay of management is diuretic titration.
Pneumonia
A large, 10-centre National Institutes of Health and Canadian Institutes of Health Research prospective cohort study found the incidence of pneumonia after cardiac surgery to be 2.4%,21 with 86% of cases developing within 30 days after cardiac surgery, but only one-third occurred after discharge. Postoperative pneumonia is usually ventilator acquired, but sedentary patients who do not adequately clear pulmonary secretions are at risk for community-acquired pneumonia after discharge from hospital. If pneumonia develops shortly after hospital discharge, it is important to consider empiric treatment that provides coverage of nosocomially acquired organisms as well.
Pulmonary embolism
Pulmonary embolism is uncommon after cardiac surgery, owing to routine aggressive thromboprophylaxis, dual antiplatelet therapy and early postoperative mobilization in hospital. Continued physical mobilization at home is also preventive. Pulmonary embolism is usually a sudden emergent event and patients present with substantial hypoxia. In patients with severe dyspnea, physicians should investigate for common causes while maintaining a high index of suspicion for pulmonary embolism.
What cognitive or neurologic problems may patients experience after surgery?
Neurologic complications after cardiac surgery have been classically divided into type 1 and type 2 deficits. Type 1 deficits — stroke or transient ischemic attack related to perioperative hypoperfusion, embolization or hemorrhage22,23 — occur in about 2% of patients undergoing cardiac surgery,24 usually early in the perioperative period. Such events are managed according to standard guidance for the management of stroke.25 Type 2 deficits (previously termed “pump-brain”) include more subtle neurocognitive complications such as postoperative cognitive dysfunction, delirium and mood disorders.
Postoperative cognitive dysfunction is generally described as a reduction in any cognitive domain after surgery, in the absence of delirium and dementia.26 It occurs in about 50%–70% of patients in the first week postoperatively. Most patients recover, but some continue to experience cognitive dysfunction (20%–40% 1 year later27), and some even decline further. It is debated whether further decline may be caused or exacerbated by underlying undiagnosed preoperative cognitive decline in older adults. Similar cognitive decline has been shown in patients with medically managed coronary artery disease.28
Delirium occurs in up to 73% of patients after cardiac surgery29 and anxiety, depression or psychosis can occur in up to one-third of patients postoperatively.30 The pathophysiology of type 2 complications is not entirely understood, but microemboli, hypoperfusion, cerebral hyperthermia, anesthetic gases and SIRS are recognized as potential mechanisms.
Risk factors for type 2 dysfunction include older age, preoperative cognitive dysfunction and premorbid psychiatric or mood disorders.31 Obvious type 2 deficits are often addressed in hospital, but subtle cognitive deficiencies might be noticed only later, by caretakers at home. Primary care practitioners should be aware of the important impacts that persistent cognitive impairment may have on social function, independence and lack of awareness of changes in physical health. To our knowledge, there is no evidence or guidelines on how to manage these subtle neurocognitive deficiencies after cardiac surgery specifically, and thus we suggest primary care practitioners adhere to their usual practice in managing and following patients with these issues.
What other complications may arise after cardiac surgery?
Complications associated with the surgical site
Primary care physicians who see patients in the 30 days after CABG surgery should routinely inspect all the surgical sites, as shown in Appendix 1 (available at www.cmaj.ca/lookup/doi/10.1503/cmaj.191108/tab-related-content), during follow-up appointments to check for superficial or deep infection or possible nonunion of the mediastinum (nontender palpable sternal click). Early identification and involvement of the cardiac surgeon can potentially prevent very serious complications, as sternal wound complications can increase morbidity and mortality by up to 40%.32 Characteristics and important considerations of surgical wounds after cardiac surgery are summarized with more detail in Appendix 2, Table 1 (available at www.cmaj.ca/lookup/doi/10.1503/cmaj.191108/tab-related-content).
Kidney injury
As many as 30% of patients develop varying degrees of acute kidney injury after cardiac surgery.33 This is usually a result of renal hypoperfusion or embolic complications perioperatively. About 1% of patients require dialysis after cardiac surgery.33 Most patients with postoperative acute kidney injury have preoperative risk factors (e.g., older age, atherosclerotic disease, diabetes, preoperative renal dysfunction or heart failure) that predispose them to acute kidney injury. Acute kidney injury is often transient, but those patients with pre-existing renal dysfunction may have long-term worsening of their renal impairment that will require careful ongoing monitoring and management.
Challenges in control of diabetes mellitus
The 2018 Diabetes Canada Clinical Practice Guidelines highlight that poor perioperative glycemic control is associated with increased complications after cardiac surgery, including sternal wound infection, delirium, renal impairment, respiratory dysfunction and prolonged length of stay in hospital.34 To avoid these complications, physicians caring for patients with diabetes perioperatively often adjust diabetic treatment to ensure tighter glycemic control. Postoperative follow-up with a primary care practitioner is critical to evaluate ongoing diabetic control and treatment, preferably targeting a glycosylated hemoglobin level of ≤ 7%, as per the Diabetes Canada Clinical Practice Guidelines.35
Gastrointestinal bleeding
Gastrointestinal bleeding is an uncommon but important consideration in patients who have undergone cardiac surgery, especially if they have pre-existing gastrointestinal pathology. Intraoperative trauma from transesophageal echocardiography, stress ulceration and NSAIDs prescribed in hospital contribute to gastrointestinal bleeding. Those patients receiving anticoagulant therapy in addition are at risk for major gastrointestinal bleeds. In the setting of gastrointestinal bleeding, the decision about whether to continue anticoagulation is best made in conjunction with the cardiac surgeon. Management should follow best practice guidelines; life-threatening bleeding should always prompt discontinuation of anticoagulation, redirection to an emergency department, and contacting the cardiac surgeon on call.
How should anticoagulation therapy after cardiac surgery be managed?
Anticoagulation for atrial fibrillation
Atrial fibrillation occurs in 30% of patients after isolated CABG.36 The 2020 Canadian Cardiovascular Society Comprehensive Guidelines for the Management of Atrial Fibrillation recommends that treatment for atrial fibrillation after cardiac surgery be individualized with either rate or rhythm control.36 Patients may therefore be discharged with a β-blocker or amiodarone or both, which can be titrated in primary care as necessary. Patients may also be discharged on anticoagulation, specifically warfarin if their atrial fibrillation persists for more than 48 hours, to decrease their risk of stroke.37 New-onset atrial fibrillation after CABG is self-limiting, and almost all patients return to a normal sinus rhythm by 6–12 weeks after cardiac surgery.36,38,39 Therefore, primary care practitioners following such patients should always re-evaluate whether anticoagulant treatment is still necessary by 3 months postoperatively.36,39 Discontinuation of anticoagulation should be considered at this time if the patient is arrhythmia free (normal sinus rhythm, no palpitations) and the only instance of atrial fibrillation was after cardiac surgery.36,39
How should hypertension be managed after CABG surgery?
Many patients with preoperative hypertension are discharged on a lower dose of their antihypertensive medications.16 This is in part because of SIRS associated with cardiopulmonary bypass and volume status changes postoperatively.40 The inflammatory process resolves with time (days) but can have lasting effects for weeks after surgery. This, combined with postoperative anemia, transient decreased cardiac function and ongoing diuresis, means that the patient’s blood pressure may transiently be lower than before surgery. Worsening hypertension may become evident as recovery progresses and require adjustment of antihypertensive medication during the following months, as per the 2020 Canadian Hypertensive Guidelines.41
How should patients approach activity after surgery?
Rehabilitation
Patients are usually actively referred by their surgical centre to cardiac rehabilitation to start 6–12 weeks postoperatively. Some Canadian provinces provide outreach rehabilitation programs by community physiotherapists.42 A comprehensive review found that cardiac rehabilitation is associated with reduced mortality after surgery.43 If recommended programs are not available, patients are encouraged to serially increase activity as tolerated to achieve the Canadian Society for Exercise Physiology activity recommendations of 150 minutes per week of moderate- to vigorous-intensity aerobic exercise.44 In practice, this is as simple as increasing daily walking distance and speed until this goal is achieved. As described previously, it is important that patients do not become sedentary at home after discharge.
Sternal precautions
Most surgical centres will discharge patients with advice on how to avoid sternal injury. Traditional advice includes instructions to avoid pulling, pushing or lifting more than 10 pounds (5 kg), avoid weight-bearing with the arms, and refrain from twisting the upper body for the first 6–12 weeks postoperatively.17,45 Newer strategies have been developed, including the “Keep Your Move in the Tube” technique, which focuses on encouraging mobility after cardiac surgery by moving away from load-bearing and time restrictions and focusing on kinesiology principles.46 The latter technique encourages patients to keep their upper arms close to their body (as if they are in a tube) to avoid excessive stress to the sternum during load-bearing movements. Resumption of a patient’s normal activities is determined at their own pace, ensuring no chest or incisional pain.46
Driving
The 2003 Canadian Cardiovascular Society executive summary on fitness to drive after cardiac surgery permits private driving 4 weeks after CABG surgery.47
Conclusion
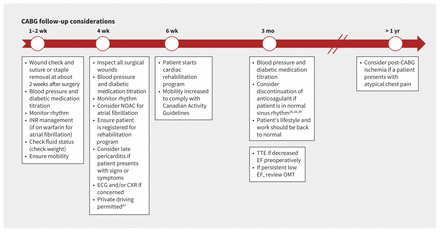
Primary care practitioners play a vital role in the postdischarge care of patients who undergo CABG surgery. This includes, but is not limited to, monitoring patients for important potential complications during their recovery, reviewing and optimizing medications, appropriate referral if complications arise, and overseeing safe return to activity and functionality. A summary of follow-up considerations in timeline form (Figure 1) is provided for quick reference.
Timeline of suggested postoperative follow-up with primary care providers. Note: CABG = coronary artery bypass grafting, CXR = chest radiography, ECG = electrocardiography, EF = ejection fraction, INR = international normalized ratio, NOAC = novel oral anticoagulant, OMT = optimal medical therapy, TTE = transthoracic echocardiography.
Footnotes
Competing interests: None declared.
This article has been peer reviewed.
Contributors: Dominique de Waard, Andrew Fagan and David Horne contributed to the conception and design of the work. All of the authors drafted the manuscript, revised it critically for important intellectual content, gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/