Persistent polycythemia, even if mild, should prompt investigations for secondary causes and, if none are found, referral to hematology and JAK2 mutation testing for polycythemia vera.
Pregnancy-specific hemoglobin and hematocrit normal ranges are lower than normal because of physiologic hemodilution.
Targeted treatment of polycythemia vera during pregnancy can prevent both fetal and maternal complications.
Thrombosis is a common complication of polycythemia vera; treatment during pregnancy is individualized based on acuity of thrombus and the patient’s thrombotic and bleeding risks.
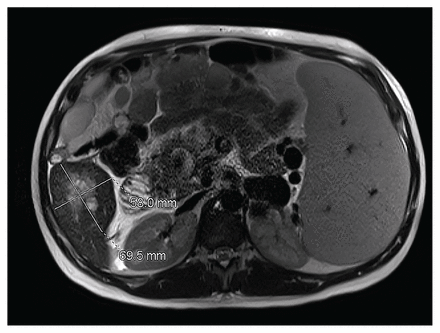
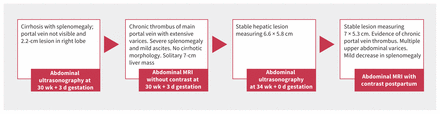
A 35-year-old woman (gravida 2, para 0, miscarriage 1) at 30 weeks and 1 day gestation via in vitro fertilization was seen in consultation in our obstetric medicine clinic for liver cirrhosis and splenomegaly without ascites. These were found incidentally on abdominal ultrasonography arranged to investigate pelvic pain. The patient was a nonsmoker with a history of polycystic ovarian syndrome and diet-controlled gestational diabetes. She was taking acetylsalicylic acid (ASA), calcium, vitamin D, and a prenatal vitamin. Physical examination revealed a palpable spleen, mild palmar erythema, and symmetric pitting edema of the lower extremities without signs of heart failure. We initiated a cirrhosis workup (Table 1) including repeat abdominal ultrasonography with Doppler, which revealed possible portal vein occlusion as well as a 2.2-cm liver lesion. The differential diagnosis of this lesion included hepatocellular carcinoma and estrogensensitive adenoma. Further imaging with abdominal magnetic resonance imaging (MRI) showed a 5.8- by 7.0-cm liver mass (Figure 1), mild ascites, nonvisualization of the main portal vein suggesting chronic thrombosis, extensive portosystemic varices, and severe splenomegaly with normal liver architecture. No findings to suggest liver fibrosis were present. The radiologist was unable to further characterize the mass given that contrast was not used in the study. The discrepant liver-lesion size between imaging procedures was considered to be due to intermodality variability, without concern for rapid growth. A multidisciplinary team was assembled to investigate and manage each issue.
Investigations for workup of cirrhosis and chronic portal vein thrombus in a 35-year-old pregnant woman
Magnetic resonance imaging of the abdomen of a 35-year-old pregnant woman with polycythemia vera and noncirrhotic portal hypertension showing a solid oval mass in the inferior right lobe of the liver (segment 6), splenomegaly, and ascites.
After discussion and review of the above investigations, particularly the MRI showing normal liver architecture, the working diagnosis shifted from cirrhosis to noncirrhotic portal hypertension with portal vein thrombus suspected as the underlying etiology. We began a thrombophilia workup, which included investigations for myeloproliferative neoplasms (Table 1) to explain the presence of an atypically located thrombus in a young woman. Given our high suspicion of esophageal varices, we initiated propranolol for variceal bleed prophylaxis. Esophagogastroduodenoscopy was deferred as our patient had no symptoms suggestive of an active gastrointestinal bleed, and it was considered to be not immediately relevant for her management. We started low-molecular-weight heparin (LMWH) at a prophylactic dose for the patient’s portal vein thrombosis. We chose prophylactic rather than therapeutic dosing owing to uncertainty around the timing of development of the portal vein thrombosis, the presence of varices, and the expected hemostatic challenges around delivery. Finally, we arranged serial imaging to monitor the hepatic lesion, rather than proceeding immediately to biopsy (Figure 2). Given incomplete characterization without contrast, the differential was broad and included benign etiologies. Further, as the mass remained stable on repeat imaging, nonurgent biopsy was recommended. A multidisciplinary meeting was held with maternal–fetal medicine, obstetric medicine, and anesthesia to plan for safe delivery considering the patient’s anticoagulation, risks of variceal bleeding, and potential trauma to a liver lesion of unclear etiology. Plans were made for delivery at 37 weeks’ gestation via cesarean delivery.
Summary and key findings from abdominal imaging. Note: MRI = magnetic resonance imaging.
At 34 weeks and 2 days’ gestation, the patient was admitted for reduced fetal movement. She was found to have elevated blood pressure (130–150/80–95 mm Hg), and rising liver enzyme and creatinine levels (Table 2), consistent with preeclampsia. She underwent an uncomplicated cesarean delivery the following day, delivering a newborn girl (Apgar score 9, 9). On postpartum day 2, the patient developed delayed postpartum hemorrhage requiring Bakri balloon insertion, 1 dose of fibrinogen, and 2 units of packed red blood cells for mild disseminated intravascular coagulation. Five days after delivery, testing for the JAK2 V617F mutation returned positive, highly suspicious for polycythemia vera.
Comparison of investigations before and during pregnancy with normal-range values
On retrospective chart review, we noted mild polycythemia dating back 2 years. A postpartum bone marrow biopsy confirmed the diagnosis of polycythemia vera. Postpartum, liver MRI with gadolinium confirmed the presence of a portal vein thrombosis and redemonstrated a stable lesion within the right hepatic lobe, later revealed to be focal nodular hyperplasia after tissue biopsy. The patient was discharged home 7 days postpartum on therapeutic LMWH owing to her ongoing risk of thrombosis from polycythemia vera, in her postoperative and postpartum state. She has ongoing follow-up with the hematology service for phlebotomy and cytoreduction.
Discussion
Erythrocytosis, or polycythemia, refers to an elevated erythrocyte count that exceeds the sex-specific normal range.2 Secondary causes of polycythemia include hypoxemia (commonly from smoking, lung disease, or sleep apnea), erythropoietin (EPO)-secreting tumours, and drugs including exogenous testosterone or EPO. Primary polycythemia due to polycythemia vera, a type of myeloproliferative neoplasm, is uncommon, with an overall prevalence of 22 per 100 000,2,3 a male predominance, and an average age at onset of 60 years.4 The incidence in women younger than 40 years is low at 0.3 per 100 000.2,3 Patients with polycythemia vera may be asymptomatic or may report pruritis, erythromelalgia, headaches, dizziness, or a history of thrombosis (including splanchnic vein thrombosis). Polycythemia should be investigated, even when mild, as it may be the only clue to the diagnosis of polycythemia vera. When a secondary cause is not evident or there is clinical suspicion for polycythemia vera, referral to a hematologist and testing for JAK2 V617F exon 14 and, if negative, the JAK2 exon 12 mutation, is recommended.2,5 As many as 95%–97% of patients with polycythemia vera test positive for the JAK2 V617F mutation, and a further 2% harbour an exon 12 mutation.6
Early recognition and treatment of polycythemia vera in pregnancy can prevent both fetal and maternal complications.4 As our case illustrates, polycythemia and polycythemia vera may go unrecognized when the hemoglobin only marginally exceeds the upper limit of the reference range. Given that hemoglobin and hematocrit levels drop with the physiologic hemodilution of pregnancy, appropriate interpretation requires awareness of trimester-specific values. Other challenges to the diagnosis include the nonspecific symptoms, the rarity of this condition in females of childbearing age, and the fluctuating hemoglobin levels in menstruating patients, especially if they have menorrhagia or, in our patient’s case, substantial bleeding during her miscarriage (Table 2, hemoglobin level in July 2020). Specific polycythemia vera therapy during pregnancy includes low-dose ASA, phlebotomy to lower hematocrit (the hematocrit target is lower in pregnancy), and cytoreduction therapy as needed, usually with interferon since other therapies are contraindicated in pregnancy. 5 A small study involving women with polycythemia vera showed significantly improved live birth rates among women who received polycythemia vera–specific therapy, including ASA or interferon, compared with those who did not.3 In addition to an increased risk of thrombosis, women with polycythemia vera have been found to have a higher risk for miscarriages, hypertensive disorders of pregnancy, preterm birth, intrauterine growth restriction, and placental insufficiency and bleeding.3,5,7
Thromboembolic events are the major complication of polycythemia vera, with a reported incidence rate of 1.7% venous thromboses per patient per year. These can occur as deep vein thromboses of extremities, as pulmonary emboli, or in atypical sites such as splanchnic or cerebral veins.8 Pregnant people are at higher risk for thrombosis, and this risk is further increased among those who also have an underlying myeloproliferative disorder such as polycythemia vera.4 Clinical manifestations of portal vein thromboses are nonspecific and include abdominal pain, nausea and vomiting, and decreased appetite, making diagnosis challenging. Patients with risk factors for portal vein thrombosis include those with a known diagnosis of cirrhosis, malignancy, intra-abdominal infection, inherited thrombophilia, myeloproliferative neoplasm, antiphospholipid antibody syndrome, inflammatory bowel disease, and autoimmune disease. 9,10 The diagnosis of portal vein thrombosis often relies on several imaging modalities such as Doppler ultrasonography, computed tomography (CT), or MRI. However, radiation exposure to the fetus from abdominal CT is concerning, and MRI with gadolinium is contraindicated in pregnancy. Anticoagulation is recommended for the management of symptomatic, acute portal vein thrombosis in patients without active bleeding. In chronic portal vein thrombosis, the choice to treat with anticoagulation therapy is individualized, based on the balance of estimated thrombotic and bleeding risk.11 Both LMWH and unfractionated heparin are safe in pregnancy, although LMWH is preferred because of the need for fewer daily injections, lower risk of osteoporosis, and lower risk of heparin-induced thrombocytopenia. When therapeutic-dose LMWH is given in the antepartum period, scheduled delivery can occur with discontinuation of the anticoagulation 24 hours prior. With prophylactic dosing, planned delivery is not necessary and patients may safely proceed to delivery (including placement of an epidural catheter) 12 hours after the last dose.12
Our patient had evidence of portal hypertension as a complication of the portal vein thrombosis, with the ascites, splenomegaly, and varices seen on imaging. The major complication of portal hypertension in pregnancy is variceal bleeding, occurring in as many as 25% of patients with noncirrhotic portal hypertension in a small prospective study of 108 cases.7 The risk of worsening portal hypertension is increased during pregnancy owing to the physiologic increase in plasma volume. In females with known portal hypertension, surveillance endoscopy can be considered before conception, or during the second trimester of pregnancy. This allows planning for appropriate management, including initiation of β-blockers such as propranolol or nadolol, and endoscopic variceal ligation.13 Acute variceal bleeding is managed similarly as outside of pregnancy with hemodynamic stabilization, intravenous octreotide, and endoscopy.
Our young patient in her third trimester, who presented with mild polycythemia and unexplained portal hypertension, was ultimately diagnosed with polycythemia vera. Her workup, diagnosis, treatment, and successful delivery of a healthy newborn underscore the importance of effective multidisciplinary collaboration in the management of complex cases.
The section Cases presents brief case reports that convey clear, practical lessons. Preference is given to common presentations of important rare conditions, and important unusual presentations of common problems. Articles start with a case presentation (500 words maximum), and a discussion of the underlying condition follows (1000 words maximum). Visual elements (e.g., tables of the differential diagnosis, clinical features or diagnostic approach) are encouraged. Consent from patients for publication of their story is a necessity. See information for authors at www.cmaj.ca.
Footnotes
Competing interests: None declared.
This article has been peer reviewed.
The authors have obtained patient consent.
Contributors: Cairina Frank and Paloma O’Meara were the primary authors and were responsible for the acquisition of the data and initial drafting. Noor Amily, Erin Kelly, Miriam Kimpton, and Pierre Villeneuve provided substantial contributions to the design of the work within their respective fields. They assisted with interpretation of data and clinical findings, and assisted with writing the manuscript by contributing opinions and framework around each of the topics, including obstetrics, gastroenterology, thrombosis, and hematology. All of the authors reviewed the article, gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/