Abstract
BACKGROUND: Trazodone is increasingly prescribed for behavioural and psychological symptoms of dementia, but little is known about its risk of harm. Our objective was to describe the comparative risk of falls and fractures among older adults with dementia dispensed trazodone or atypical antipsychotics.
METHODS: The study cohort included adults with dementia (excluding patients with chronic psychotic illnesses) living in long-term care and aged 66 years and older. Data were obtained from routinely collected, linked health administrative databases in Ontario, Canada. We compared new users of trazodone with new users of atypical antipsychotics (quetiapine, olanzapine or risperidone) between Dec. 1, 2009, and Dec. 31, 2015. The primary outcome was a composite of fall or major osteoporotic fracture within 90 days of first prescription. Secondary outcomes were falls, major osteoporotic fractures, hip fractures and all-cause mortality.
RESULTS: We included 6588 older adults dispensed trazodone and 2875 dispensed an atypical antipsychotic, of whom 95.2% received a low dose of these medications. Compared with use of atypical antipsychotics, use of trazodone was associated with similar rates of falls or major osteoporotic fractures (weighted hazard ratio [HR] 0.89, 95% confidence interval [CI] 0.73 to 1.07), major osteoporotic fracture (weighted HR 1.03, 95% CI 0.73 to 1.47), falls (weighted HR 0.91, 95% CI 0.75 to 1.11) and hip fractures (weighted HR 0.92, 95% CI 0.59 to 1.43). Use of trazodone was associated with a lower rate of mortality (weighted HR 0.75, 95% CI 0.66 to 0.85).
INTERPRETATION: Trazodone is not a uniformly safer alternative to atypical antipsychotics, given the similar risk of falls and fractures among older adults with dementia.
The prevalence of dementia in Canada is 7.1%, but the rate approaches 25% among Canadians aged 85 years and older.1 Importantly, 61.9% of residents in continuing care facilities have dementia, and 48% have demonstrated aggressive behaviours.2 The behavioural and psychological symptoms of dementia (e.g., aggression) can lead to caregiver burden and difficulty in providing safe and timely care for patients with dementia.3–6 Despite the limited evidence of treatment efficacy, both antipsychotics and trazodone (an antidepressant medication) are commonly used to alleviate the behavioural and psychological symptoms of dementia.7 In 2013, 34% of older adults with dementia living in a long-term care facility in Ontario, Canada, were dispensed an atypical antipsychotic, and 21.3% were dispensed trazodone.3,8 Similarly high rates of antidepressant and atypical antipsychotic use were reported among patients with dementia in the United States and Europe.5,9,10
Antipsychotics are associated with substantial harm among older adults with dementia, including myocardial infarction, aspiration pneumonia and death.11–13 There is growing concern about the use of antipsychotics for indications other than the treatment of chronic psychotic illnesses.14–21 Clinical practice guidelines and quality-improvement initiatives have encouraged clinicians to decrease antipsychotic use in older adults with dementia.15,17–22 Comparatively, little is known about the risk of harm from trazodone in older adults with dementia — despite its increasing use.3,8,9 In Ontario, the prevalence of trazodone prescriptions has risen sharply, from 7.7% in 2002 to 21.3% in 2013.3 Randomized controlled trials (RCTs) of trazodone in patients with dementia described adverse effects, including parkinsonism, drowsiness, dizziness and hypotension, which could contribute to an increased risk of falls or fractures; only one study reported a patient was lost to follow-up in its placebo arm for having a fracture.23–27 Understanding how potential harms associated with trazodone compare with the harms associated with antipsychotics is important because we know there are risks associated with antipsychotic use in older adults.28 Clinicians might use trazodone to treat the behavioural and psychological symptoms of dementia instead of antipsychotics to avoid these risks.
We examined the comparative risk of the composite outcome of falls and major osteoporotic fractures, falls, major osteoporotic fractures, hip fractures and all-cause mortality among older adults with dementia dispensed trazodone or atypical antipsychotics.
Methods
This manuscript is reported in accordance with the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) and RECORD (Reporting of Studies Conducted Using Observational Routinely-collected Data) statements for the reporting of observational studies.29,30
Setting and data sources
We created our cohort using the linked health administrative databases at ICES in Ontario, Canada. Ontario has a largely publicly funded health care system, in which individuals aged 65 years or older have guaranteed housing in long-term care facilities when necessary, and universal coverage for physician services and most prescription medications. Patient-level information was linked using an encoded version of each patient’s health insurance program number.31 We linked data from the following databases: Ontario Drug Benefit, ICES Physician Database, Ontario Mental Health Reporting System, National Ambulatory Care Reporting System, Ontario Health Insurance Plan Database, Registered Persons Database, Discharge Abstract Database and Continuing Care Reporting System.32 A description of these databases is found in Appendix 1a, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.180551/-/DC1. These databases are accurate and reliable.31,33–37
Study design
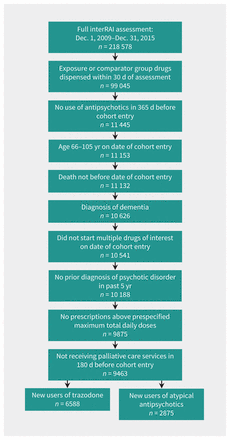
We implemented a retrospective cohort study design. Our index date was the date of first prescription of an exposure or comparator drug. We identified patients who were dispensed a study drug and had a full interRAI (International Resident Assessment Instrument) assessment within 30 days before cohort entry. The Resident Assessment Instrument–Minimum Data Set is a validated assessment that contains information about aspects of patients’ health, including independence in activities of daily living and severity of cognitive impairment.38,39 We required assessments within 30 days to ensure a close temporal relation between drug exposure and data on patients’ state of health. This cohort included adults aged 66 years or older with dementia who were living in long-term care facilities and newly dispensed oral trazodone or atypical antipsychotics (quetiapine, olanzapine or risperidone) between Dec. 1, 2009, and Dec. 31, 2015. We identified patients with dementia using the validated algorithm of Jaakkimainen and colleagues, and diagnostic codes from the interRAI assessment.33,40 The algorithm of Jaakkimainen and colleagues has a sensitivity of 79.3% and specificity of 99.1% for identifying dementia. Our observation window was 90 days, which was chosen to balance the need for sufficient time for accrual of events with the need to lessen the chance of residual confounding. The maximum follow-up date was Mar. 31, 2016.
We excluded participants from our cohort if they did not have a complete interRAI assessment within 30 days before cohort entry, received any antipsychotics or trazodone within the year before cohort entry, did not have a history of dementia, were dispensed 2 or more of our exposure drugs on the date of cohort entry, had a diagnosis of a chronic psychotic illness within 2 years of cohort entry, received palliative care services within 180 days of cohort entry, received the study drugs above a prespecified maximum total daily dose at cohort entry, or were younger than 66 or older than 105 years.40–42
Patients in our exposure group received oral trazodone (maximum total daily dose: 300 mg) and those in our comparator group received oral quetiapine (maximum total daily dose: 300 mg), risperidone (maximum total daily dose: 3 mg) or olanzapine (maximum total daily dose: 10 mg). Equivalency ratios were calculated as the mean of equally efficacious doses of drugs across RCTs (Appendix 1b, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.180551/-/DC1.).43,44
We derived stabilized inverse probability of treatment weights from a propensity score model in which exposure status was regressed on a set of measured baseline covariates. Covariates were selected for inclusion in the propensity score model based on the existing literature and clinical judgment (see Appendix 2, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.180551/-/DC1, for the variables included in the propensity score model).45 As a sensitivity analysis, inverse probability of treatment weights were also derived from a high-dimensional propensity score model.46 The following variables were forced into the high-dimensional propensity score: age, sex, Cognitive Performance Scale score, dependency in activities of daily living, year of cohort entry, presence of delusions, Aggressive Behaviours Scale score, Depression Rating Scale score, weight, wheelchair ambulation, number of geriatrician visits, and history of major osteoporotic fracture or falls leading to hospital presentation.47–49
We defined our outcomes with input from 12 care partners of older adults with dementia. Outcomes were ranked in descending order of importance from among commonly reported safety outcomes (e.g., mortality and stroke) in studies of pharmacologic treatments for the behavioural and psychologic symptoms of dementia.11,50,51 We defined our primary outcome as a composite of a fall resulting in an emergency department visit or major osteoporotic fracture. A major osteoporotic fracture was defined as a fracture of the hip, pelvis, humerus or forearm.52 These outcomes have been identified in administrative databases with a high positive predictive value and level of agreement during medical chart re-abstraction.31,52–55 Our secondary outcomes were all-cause mortality and the components of our primary outcome: falls, major osteoporotic fracture and hip fracture. We included a tracer outcome (cataract surgery) to assess the sensitivity of our findings to unmeasured confounding.56–58 All International Classification of Diseases, 10th Revision (ICD-10) codes and algorithms used to define patients’ baseline characteristics and study outcomes are found in Appendix 3, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.180551/-/DC1.31,33,40–42,52,59–68
Statistical analysis
We compared baseline characteristics of patients in our exposure and comparator groups with standardized differences.69 Groups were considered similar if the standardized difference was less than 0.1.70,71 We reported the proportion of patients missing data for individual baseline characteristics. Stabilized inverse probability of treatment weights were derived from the estimated propensity score. Treatment weights were inspected for outlying values (> 50).72 Unweighted and weighted cause-specific hazard ratios (HRs) were derived from cause-specific hazards models that accounted for the competing risk of death.73,74 We chose the cause-specific hazard model because we wanted to understand the association between our exposure and the rate of outcome in participants who were event-free and thus at risk of the event. This addresses a primarily etiologic question.75 Robust standard errors were used to account for within-subject homogeneity in outcomes induced by weighting.76 We verified that the HR did not vary over time. We based our primary analyses on an intention-to-treat principle whereby patients in the cohort were followed until the first of the following: outcome of interest, death or 90 days after index date. In secondary analyses, we censored patients in the cohort if they were dispensed a drug from the other exposure group during the 90-day follow-up period. Weighted incidence rates are reported as the number of events per 100 person-years. Risk differences were calculated as the weight-adjusted difference in absolute risk among patients dispensed trazodone minus the absolute risk in patients dispensed atypical antipsychotics at 90 days. Where numbers permitted, we planned to conduct subgroup analyses of outcomes based on age, sex and dementia severity. We also planned to describe the effect of drug dose on outcomes using dose as a time-varying covariate in an unweighted Cox proportional hazards model incorporating all of the characteristics described in Appendix 2.
As a sensitivity analysis, we derived stabilized inverse probability of treatment weights from the estimated high-dimensional propensity score. Re-weighted cause-specific HRs were derived for our primary and secondary outcomes. Lastly, we repeated our weighted regression analyses using a subdistribution hazard model that accounted for the competing risk of death.77 With the subdistribution hazard model, we are estimating the association of exposure with the cumulative incidence function.78 Formally, the subdistribution HR compares the instantaneous rate of the outcome in the risk set of participants who are event-free or who have experienced a competing event, while the cause-specific hazard model focuses on comparing the rates of the outcome in participants who are currently event-free. All analyses were conducted using SAS, version 9.4.
Ethics approval
This study was approved by the University of Toronto and Sunnybrook Health Sciences Centre research ethics boards.
Results
Our cohort consisted of 9463 patients: 6588 were newly dispensed trazodone and 2875 were newly dispensed atypical antipsychotics (Figure 1). There were no outlying stabilized inverse probability of treatment weights. Among patients dispensed atypical antipsychotics, 275 (9.6%) were dispensed olanzapine, 1511 (52.6%) were dispensed quetiapine and 1089 (37.9%) were dispensed risperidone. Almost all patients were dispensed a low dose of the exposure or comparator drug: 95.2% of patients were dispensed a dose that was less than or equivalent to 2.5 mg of olanzapine per day, 4.1% were dispensed a moderate dose and 0.7% were dispensed a high dose. After applying inverse probability of treatment weights, exposure and comparator groups were similar at baseline (Table 1). The mean age of patients on the date of cohort entry was 85.3 (standard deviation 7.2) years, and 68.7% were female.
Flow diagram of study cohort creation. Note: interRAI = International Resident Assessment Instrument.
Selected baseline characteristics* of older adults dispensed trazodone or atypical antipsychotics
In our primary analysis, patients newly dispensed trazodone experienced a similar rate of falls and major osteoporotic fractures after inverse probability of treatment weighting (cause-specific HR 0.89, 95% confidence interval [CI] 0.73 to 1.07; Table 2) compared with those initiating atypical antipsychotics. We also found similar rates of our secondary outcomes of falls (cause-specific HR 0.91, 95% CI 0.75 to 1.11), major osteoporotic fractures (cause-specific HR 1.03, 95% CI 0.73 to 1.47), and hip fractures (cause-specific HR 0.92, 95% CI 0.59 to 1.43). However, patients dispensed trazodone had a lower rate of all-cause mortality (HR 0.75, 95% CI 0.66 to 0.85). In our tracer analysis, there was no difference in the rate of cataract surgery between new users of trazodone or atypical antipsychotics (HR 0.67, 95% CI 0.33 to 1.34).
Primary and secondary analyses of the comparative risk of primary and secondary outcomes for new users of trazodone versus atypical antipsychotics within 90 days
The results of our secondary analyses were consistent with those of our primary analyses (Table 2). The baseline characteristics of our cohort and our conclusions were unchanged using the high-dimensional propensity score to derive our inverse probability of treatment weights (Appendix 4, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.180551/-/DC1). The subdistribution hazard analysis showed that the cumulative incidence function for falls or major osteoporotic fracture, falls, major osteoporotic fractures, and hip fractures were similar between the 2 drug groups (Appendix 5, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.180551/-/DC1). We did not do our planned analysis of time-varying dose because almost all patients in our cohort were dispensed a low-dose equivalent. We also did not conduct subgroup analyses because our sample was too small to derive meaningful effect estimates.
Interpretation
In this population-based study of older adults with dementia, we found that patients dispensed trazodone had a rate of falls, major osteoporotic fractures and hip fractures comparable to those of patients dispensed atypical antipsychotics; however, their rate of all-cause mortality within 90 days was significantly lower, as discussed below. In Ontario, falls and fractures are not uncommon among residents of long-term care facilities: 2.8% had a fall resulting in an emergency department visit or hospital admission within 90 days of entering into a study cohort and 3.3% had a fracture of the hip, pelvis, wrist or humerus over a 1-year period.79,80 Among residents of Ontario long-term care facilities who were not prescribed an antipsychotic, 2.7% of residents died within 30 days and 15.1% of residents died within 180 days of cohort entry.81
In the broader literature concerning trazodone, its association with a patient’s risk of fall or fracture is unclear.80,82–84 In a cross-sectional analysis of older men living in the community, trazodone use was not associated with an increased risk of fracture; however, these men had less burden of illness than our study cohort.84 A study of older adults in long-term care facilities found an increased risk of fall and fracture among patients dispensed trazodone.80 The association between trazodone use and death among older adults with dementia was a novel finding. Could the greater risk of death associated with atypical antipsychotic use be related to an altered cardiometabolic profile? Although both trazodone and atypical antipsychotics have been associated with an increased risk of falls and fractures, antipsychotic use has also been associated with an increased risk of myocardial infarction and stroke in patients with dementia.12,50,80
Limitations
Despite our use of a clinically derived propensity score using detailed clinical data, the nonrandomized nature of our study means that it is possible for unmeasured confounding to influence our findings. However, our results did not change in our sensitivity analysis, in which we implemented a high-dimensional propensity score model, and the results of our primary and secondary analyses were consistent. Because of software limitations, we were unable to estimate the cumulative incidence functions in the weighted samples. To limit confounding from frailty and medication noncompliance, we chose a moderately to severely frail population of patients with dementia living in long-term care facilities. This might limit the generalizability of our findings to less frail older adults outside of a long-term care setting, but frailty has prognostic importance in older adults.85,86 Our outcome of falls was limited to patients presenting to hospital.52,87 Therefore, the number of events we observed is likely an underestimate of the number of falls associated with trazodone and atypical antipsychotics; however, there should not be any differential misclassification between our exposure and comparator groups (assuming that patients in each group experienced falls of similar severity).
Conclusion
As clinicians move to decrease antipsychotic use, we should not consider trazodone as a uniformly safer alternative to atypical antipsychotics, because trazodone use was associated with a comparable risk of falls and major osteoporotic fractures to atypical antipsychotics — drugs associated with these adverse outcomes in our patient population.11,28 As appropriate prescribing campaigns target antipsychotic use in older adults with dementia, we will need to consider how these campaigns could lead to collateral changes in clinical practice.14,17
Acknowledgements
The authors thank Dr. Zahra Goodarzi for her help in conceptualizing our stakeholder engagement survey. The authors also thank the following people for completing our survey: Dr. Camilla Wong, Ms. Mary-Anne Lee, Ms. Joanna Stanley, Ms. Denise Watt, Ms. Hazel Sebastian, Dr. Marie Patton, Ms. Loralee Fox, Ms. Junyan Shi, Dr. Jayna Holroyd-Leduc and Dr. David Hogan. The authors thank Dr. Andrea Tricco and Dr. Areti-Angeliki Veroniki for their ongoing contributions to the completion of Dr. Jennifer Watt’s doctoral thesis, of which this manuscript is a component. The authors thank Brogan Inc., Ottawa, for use of its Drug Product and Therapeutic Class Database.
Footnotes
See related article at www.cmaj.ca/lookup/doi/10.1503/cmaj.181486
Competing interests: Tara Gomes has received grant funding from the Ontario Ministry of Health and Long-Term Care. All other authors have no conflicts of interest to declare.
This article has been peer reviewed.
Contributors: Jennifer Watt, Tara Gomes, Susan Bronskill, Peter Austin, Joanne Ho and Sharon Straus designed the study. Anjie Huang and Jennifer Watt completed all data analysis. Jennifer Watt drafted the manuscript, which all of the authors revised. All of the authors gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Funding: This study was funded by the breaKThrough (knowledge translation) Program of St. Michael’s Hospital. Jennifer Watt is supported by a Canadian Institutes of Health Research (CIHR) doctoral research award and the University of Toronto Department of Medicine Eliot Phillipson Clinician–Scientist Training program. Sharon Straus is funded by a Tier 1 Canada Research Chair in Knowledge Translation. Peter Austin is funded by a Mid-Career Investigator Award from the Heart and Stroke Foundation. This study was also supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health and Long-Term Care (MOHLTC). Data sets provided by ICES were linked using unique encoded identifiers and analyzed at ICES.
The funders had no role in the design and conduct of the study; collection, management, analysis and interpretation of the data; preparation, review, or approval of the manuscript; or decision to submit the manuscript for publication. Parts of this material are based on data and information compiled and provided by the Canadian Institute for Health Information (CIHI). However, the analyses, conclusions, opinions and statements expressed herein are those of the authors, and not necessarily those of CIHI.
Data sharing: The data set from this study is held securely in coded form at ICES. Although data-sharing agreements prohibit ICES from making the data set publicly available, access may be granted to those who meet prespecified criteria for confidential access, available at www.ices.on.ca/DAS.
Disclaimer: This study was supported by ICES, which is funded by an annual grant from the Ontario MOHLTC. The opinions, results and conclusions reported in this paper are those of the authors and are independent from the funding sources. No endorsement by ICES or the Ontario MOHLTC is intended or should be inferred.
- Accepted October 5, 2018.