- © 2007 Canadian Medical Association or its licensors
Abstract
Background: Recent reports from North America and Europe have documented an annual increase in the incidence of differentiated thyroid carcinoma. We sought to investigate the relation between rates of detection, tumour size, age and sex.
Methods: Using the Ontario Cancer Registry, we identified 7422 cases of differentiated thyroid carcinoma diagnosed from Jan. 1, 1990, to Dec. 31, 2001. We obtained pathology reports for a random 10% of the 7422 patients for each year of the study period. The sample represented all Cancer Care Ontario regions. We compared the size of the patients' tumours by year, sex and age.
Results: As expected, the incidence of differentiated thyroid carcinoma increased over the 12-year period. A significantly higher number of small (≤ 2 cm), nonpalpable tumours were resected in 2001 than in 1990 (p = 0.001). The incidence of tumours 2–4 cm in diameter remained stable. When we examined differences in tumour detection rates by age and sex, we observed a disproportionate increase in the number of small tumours detected among women and among patients older than 45 years.
Interpretation: Our findings suggest that more frequent use of medical imaging has led to an increased detection rate of small, subclinical tumours, which in turn accounts for the higher incidence of differentiated thyroid carcinoma. This suggests that we need to re-evaluate our understanding of the trends in thyroid cancer incidence.
Cancer of the thyroid is the most common malignant disease of the endocrine system, and it is the seventh most common cancer affecting women.1 It is also one of the few cancers with a documented rising incidence, as reported in studies from Canada,2 the United States3–5 and Europe.6–10 Differentiated thyroid carcinoma is the most common form of thyroid cancer, accounting for about 90% of all cases, and includes both papillary thyroid carcinomas and follicular carcinomas.1
There has been much speculation about why the incidence of differentiated thyroid carcinoma is rising. Some researchers have suggested that potential risk factors include radiation exposure,11–13 iodine deficiency,8,9 family history of thyroid cancer, and personal history of goiter or thyroid nodule.3 It has also been proposed that at least some of the increase may be related to head and neck radiation therapy used to treat benign childhood conditions between 1910 and 1960.14 Studies examining the long-term incidence of thyroid cancer after radiation exposure have suggested that the risk may be as high as 15%.13,15,16 About 74% of newly diagnosed cases of differentiated thyroid carcinoma involve women1 and it has been suggested that certain female hormonal and reproductive factors may play a role.17,18
An alternative explanation is that the rise in incidence may be due to increased detection of subclinical diseases from greater use of medical imaging. A recent retrospective cohort study in the United States examined the change in size of resected thyroid tumours from 1973 to 2002.4 Using data from the National Cancer Institutes' Surveillance Epidemiology and End Results (SEER) Cancer Statistic database, investigators found a significant decrease in tumour size over time and concluded that the apparent increase in disease incidence was due to increased detection of small tumours rather than an actual increase in the number of thyroid cancers.
We sought to assess the incidence of thyroid cancer over a 12-year period in Ontario. We hypothesized that if the rising incidence was due to increased detection of subclinical tumours, small tumours would account for an increasing proportion of tumours detected. On the other hand, if environmental factors caused the increased incidence, the distribution of tumour size would remain stable over time. We tested our hypothesis by comparing differences in detection rates by sex and age within the study population.
Methods
Study population
We searched the Ontario Cancer Registry using the International Classification of Diseases (ninth edition) code 193, which represents malignant neoplasm of the thyroid gland, to identify patients in Ontario who received a diagnosis of thyroid cancer from Jan. 1, 1990, to Dec. 31, 2001. The Ontario Cancer Registry is a population-based registry of all new cases of cancer in the province. Cancer Care Ontario operates the registry and, together with the Princess Margaret Hospital, coordinates and provides comprehensive cancer care for the 11 million people in Ontario's 8 geographic regions. The registry includes pathology reports, electronic patient records (obtained from the Cancer Care Ontario treatment centres), electronic hospital discharge records (from the Canadian Institute for Health Information) and electronic reports of deaths from Ontario's Office of the Registrar General. The Ontario Cancer Registry uses deterministic linkages to reconcile information from all these sources and creates a composite record of incident cases. The completeness of the registry data and success of data linkage has recently been studied for head and neck cancer and was found to be of high quality.19
Once we obtained ethics approval from the Queen's University Research Ethics Board, we identified 8668 cases of thyroid cancer based on the International Classification of Diseases code 193. Of these, 7422 cases of differentiated thyroid carcinoma were identified (92.2% papillary thyroid and 7.8% follicular). An electronic data abstraction form was created to record many variables, including age, sex, size of tumour and pathologic diagnosis. We obtained pathology reports from the Ontario Cancer Registry for a random 10% of the 7422 patients for each year of the study period, representing all Cancer Care Ontario regions. We reviewed the 1227 thyroid-related pathology reports from those patients, some of whom had more than 1 pathology report because they had preoperative fine-needle aspiration biopsies that were diagnostic of cancer. We included in our study the 605 patients who had surgery in Ontario as initial treatment for differentiated thyroid carcinoma. We excluded patients who were under the age of 18 years, and those who had retrosternal tumours, underwent surgery for another condition (thyroglossal duct cyst excision or laryngectomy), had an initial surgical resection outside of Ontario or had no initial definitive surgical treatment during the study period.
Outcomes
We compared the sizes of patients' tumours by year. We grouped patients according to tumour size (small tumours ≤ 2 cm, medium-sized tumours 2–4 cm and large tumours > 4 cm). These size criteria were selected because they correspond to the American Joint Committee on Cancer's tumour–node–metastisis staging system (6th edition),20 and because tumours smaller than 2 cm are rarely palpable and do not cause compressive symptoms, which means this size is representative of subclinical disease.
Statistical analysis
We used analysis of covariance to compare the differences in rates of detection between tumour size groups, first for all patients, and then by sex and age (≤ 45 and > 45 years). First, we fit 3 separate regression lines for the 3 tumour size groups. Then, to compare the rates of detection for the groups, we fit global multiple regression models that included the interaction between year and slope.
Results
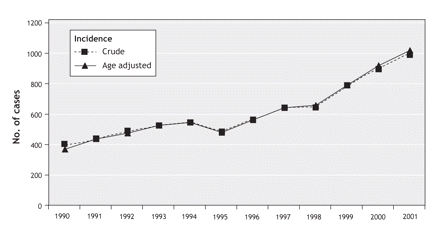
The rising incidence of differentiated thyroid carcinoma across Ontario (crude and age-adjusted) is shown in Figure 1. The number of reported cases increased from 403 in 1990 to 990 in 2001, which translates to an increase of 146% in 12 years or an overall increase of 13% per year (slope = 4.83, 95% confidence interval [CI] 0.71–8.96, p = 0.03).
Figure 1: Overall incidence of differentiated thyroid carcinoma in Ontario from 1990 to 2001, by tumour size.
Table 1 presents the histology, sex, age and tumour size for all 605 patients included in the study. The mean age was 44.6 (standard deviation [SD] 14.7) years. The majority of patients had small tumours.
Table 1.
Table 2 and Figure 2 show the incidence rates each year according to tumour size and demonstrate a statistically significant increase in the detection rate for the small-tumour group, but not for the medium-sized-tumour group (see also Appendix 1, available online at www.cmaj.ca/cgi/content/full/177/11/1357/DC2). The detection rate for the large-tumour group was statistically significant. Large tumours represented a small proportion (8.9%) of the overall number detected in our study population (Table 1). Between-group comparisons showed a statistically significant difference in incidence between the small-tumour group and the medium-sized-tumour group (p = 0.007) and between the small-tumour group and the large-tumour group (p = 0.0002), but not between the medium-sized-tumour group and the large-tumour group (p = 0.173).
Table 2.
Figure 2: Incidence of differentiated thyroid cancer from Jan. 1, 1990, to Dec. 31, 2001, according to tumour size. The regression line of the small-tumour group (< 2 cm) demonstrates a noteworthy increase over time (slope 9.57, 95% CI 5.40–13.74, p = 0.001).
When we compared the tumour sizes among the female patients, there was a significant increase in the detection rates for the small-and the medium-sized-tumour groups (Table 2). Between-group comparisons demonstrated an increase in incidence in the small-tumour group relative to the medium-sized-tumour group (p = 0.004), and between the small-tumour group and the large-tumour group (p = 0.0001) but not between the medium-sized-tumour group and the large-tumour group (p = 0.171). Among the male patients, the overall incidence of differentiated thyroid cancer was much lower than among the female patients (22.1% v. 77.4%) (Table 1), and we did not find statistically significant differences in incidence when we compared tumour sizes.
Among patients 45 years or younger, we found a significant increase in incidence for large tumours over time (Table 2). However, we found no significant difference in incidence between the small-tumour group and the medium-sized-tumour group (p = 0.494), or between the medium-sized-tumour group and the large-tumour group (p = 0.742). On the other hand, among patients over 45 years old, there was a significant increase in both small-and medium-sized tumours over time, but not in large tumours. The between-group comparisons showed a significant increase in the incidence of small tumours compared with medium-sized tumours (p = 0.001) and large tumours (p = 0.0001). There was no significant difference between medium-sized tumours and large tumours (p = 0.057).
We unexpectedly observed a slight, but significant, increase in large tumours among patients 45 years or younger; however, this result may be a spurious finding owing to the small segment of our study population that was affected. A more focused study on this group is needed to determine whether these findings represent a real increase.
Interpretation
Our findings are consistent with our hypothesis that the rising rate of differentiated thyroid cancer in Ontario is due mostly to increasing detection of small tumours from greater use of medical imaging. These data support similar recent findings from the United States,4 but our more detailed analysis identifies patterns according to sex and age.
Two studies examined the use of diagnostic imaging over time. In a US study, the compound annual rate of increase in the use of computed tomography, magnetic resonance imaging and ultrasonography increased 5% to 10% between 1992 and 2001.21 A Canadian study observed a similar pattern. Although data on the use of imaging procedures were not reported, a national survey of medical equipment showed that the number of computed tomography and magnetic resonance imaging scanners in use increased by 55% and 125% respectively from 1991 to 2005.22
In order for increased use of diagnostic imaging to account for the rising incidence of tumours, a large number of subclinical tumours must also exist in the population. Several studies have attempted to assess the prevalence of these types of tumours. Of these, autopsy studies provide the best assessment. They found clinically occult tumours in 2.7%–36% of patients.23–26 Although these findings varied widely, they suggest that a large proportion of the population lives with previously undetected thyroid tumours.
We observed that small differentiated tumours were found predominately in women older than 45 years, which may be explained by the higher incidence of benign thyroid disease in this segment of the population.23 Greater use of medical imaging in this population is an alternative explanation. Research has shown that female sex and higher age are factors that predict increased use of diagnostic medical imaging in this segment of the population.27
The main limitation of our study is that it is a retrospective review of pathology reports from a sample population representing only 10% of patients who received a diagnosis of differentiated thyroid carcinoma during our study period. However, we present compelling evidence of an increasing incidence of small, subclinical differentiated thyroid carcinomas. Previous studies that examined the natural history of these tumours have primarily focused on microcarcinomas (< 1 cm) or occult carcinomas found incidentally. Their findings suggest that these lesions, even when multifocal, seem to have an indolent natural history distinct from that of larger tumours.24,26,28,29
Both our findings and findings from these studies contradict traditional clinical practice and training, which promotes total thyroidectomy as the optimal surgical procedure for the management of all differentiated thyroid carcinomas.30,31 Although the percentage may be less in Canada, several US studies suggest that as many as 85% of patients with papillary carcinomas undergo total thyroidectomy regardless of the size of the tumour.31–34 With a rate of postoperative hypoparathyroidism between 5% and 30% and of recurrent laryngeal nerve injury between 1% and 2%,34,35 total thyroidectomy has an important risk of complications. For a disease that has an excellent prognosis for many patients, management of small thyroid tumours may be based on an outdated clinical model and result in unnecessary investigations and overly aggressive surgery.
@ See related article page 1383
Footnotes
-
Une version française de ce résumé est disponible à l'adresse www.cmaj.ca/cgi/content/full/177/11/1357/DC1
This article has been peer reviewed.
Contributors: Dr. Hall and Dr. Kent played the principal role in the conception and design of the study, data analysis, and the writing and revision of the manuscript. Dr. Isotalo, Dr. Houlden, Dr. George and Dr. Groome all had significant input in the design, and data acquisition and interpretation. They assisted in the writing and revision of the manuscript. All authors approved the final version of the manuscript.
Acknowledgements: We thank Tina Dyer, research associate.
Stephen Hall is supported by the Canadian Institutes of Health Research New Investigator Program. Patti Groome is the Canada Research Chair in Cancer Care Evaluation. This project was funded by the Clinical Teachers of Queen's University.
Competing interests: None declared.