Abstract
Background: Limited data are available on the relation between left atrial size and outcome among patients referred for clinically indicated echocardiograms. Our aim was to assess the association of left atrial size with all-cause mortality and ischemic stroke in a large cohort of patients referred for echocardiography.
Methods: Left atrial diameter was measured in 52 639 patients aged 18 years or older (mean age 61.8 [standard deviation (SD) 16.3] years; 52.9% men) who underwent a first transthoracic echocardiogram for clinical reasons at our institution between April 1990 and March 2008. The outcomes were all-cause mortality and nonfatal ischemic stroke.
Results: Based on the criteria of the American Society of Echocardiography, 50.4% of the patients had no left atrial enlargement, whereas 24.5% had mild, 13.3% had moderate and 11.7% had severe left atrial enlargement. Over a mean follow-up period of 5.5 (SD 4.1) years, 12 527 patients died, and 2314 patients had a nonfatal ischemic stroke. Cumulative 10-year survival was 73.7% among patients with normal left atrial size, 62.5% among those with mild enlargement, 54.8% among those with moderate enlargement and 45% among those with severe enlargement (p < 0.001). After adjustment in multivariable Cox proportional hazard analysis, left atrial diameter remained a predictor of all-cause mortality in both sexes (hazard ratio [HR] per 1-cm increment in left atrial size 1.17, 95% confidence interval [CI] 1.12–1.22, p < 0.001 in women, and HR 1.09, 95% CI 1.05–1.13, p < 0.001 in men) and of ischemic stroke in women (HR 1.25, 95% CI 1.14–1.37, p < 0.001).
Interpretation: Left atrial diameter has a graded and independent association with all-cause mortality in both sexes and with ischemic stroke in women.
See related commentary by Goldstein at www.cmaj.ca/lookup/doi/10.1503/cmaj.110617
The left atrium plays a major role in cardiac physiology by collecting blood during systole and modulating left ventricular filling during diastole.1 Left ventricular diastolic dysfunction or mitral valve disease may lead to left atrial pressure or volume overload which, if chronically maintained, may result in left atrial remodeling and enlargement.2 As a marker of left ventricular diastolic dysfunction3 or increased filling volumes, left atrial size may provide important prognostic information. In this regard, left atrial enlargement has been related to higher risk of atrial fibrillation4–7 and cardiovascular events.8–13 Our aim was to assess the association of left atrial size with all-cause mortality and ischemic stroke in a large cohort of patients referred for echocardiography.
Methods
Patients
Between Apr. 4, 1990, and Mar. 11, 2008, 116 114 transthoracic echocardiograms were performed in our institution and were entered in a prospectively collected database. For each patient, the first comprehensive transthoracic echocardiogram was selected, leaving a sample of 54 513 patients. We further excluded 863 patients who were younger than 18 years, 128 patients who had a prior history of cardiac transplantation and 883 patients for whom no follow-up data was available. Thus, 52 639 patients were finally included. The study was approved by the local research ethics committee (Comité Ético de Investigación Clínica de Galicia), which waived the requirement for patient consent.
Clinical data
Demographic, clinical and echocardiographic data were entered in our database at the time of the echocardiograms. The study was completed using linked clinical and administrative databases as well as electronic medical records through the Department of Information Technology of our institution. Data sources were linked using unique identifiers.
Diabetes mellitus was defined as a prior fasting serum glucose level of 126 mg/dL or higher or a nonfasting glucose level of 200 mg/dL or higher, abstracted from comprehensive laboratory databases. Hypercholesterolemia was defined as a total cholesterol level of 220 mg/dL or higher in any prior laboratory test. Hypertension was defined as either blood pressure higher than 140/90 or a previously established diagnosis. A history of atrial fibrillation was defined as atrial fibrillation recorded at the time of the echocardiogram or any previously known episode of atrial fibrillation. A history of mitral valve intervention was defined as prior mitral valve replacement or repair or prior percutaneous or surgical mitral commissurotomy. Chronic kidney disease was defined as an estimated glomerular filtration rate less than 60 mL/min/1.73 m2 using the abbreviated Modification of Diet in Renal Disease Study equation in at least two prior observations more than 90 days apart.14
Echocardiographic data
Echocardiograms were performed in the left lateral decubitus position using standard imaging planes, according to the recommendations of the American Society of Echocardiography.15 Left atrial diameter was measured using M-mode or two-dimensional echocardiography, from the posterior aortic wall to the posterior left atrial wall, in the parasternal long-axis view at the end-ventricular systole16 (i.e., just before the mitral valve opening). Left atrial diameter was categorized as a sex-specific variable, such that left atrial enlargement was defined as a left atrial diameter of 4.1 cm or greater in men or 3.9 cm or greater in women; a left atrial diameter below these values defined normal left atrial size. Left atrial enlargement was further classified as mild (left atrial diameter 4.1–4.6 cm in men or 3.9–4.2 cm in women), moderate (4.7–5.1 cm in men or 4.3–4.6 cm in women) or severe (≥ 5.2 cm in men or ≥ 4.7 cm in women) in accordance with the recommendations of the American Society of Echocardiography.16 The intraobserver and interobserver variability for the classification of left atrial size into these four categories was assessed by two independent observers (A.B-M. and E.M.) in a subset of 40 randomly selected patients. The intraobserver agreement was 87.2% (κ = 0.82, standard deviation [SD] 0.07), and the interobserver agreement was 84.6% (κ = 0.79, SD 0.08).
Left ventricular ejection fraction was estimated using the Teichholz formula or the Simpson rule. Left ventricular systolic dysfunction was defined as left ventricular ejection fraction < 55%.16 Ventricular septal thickness, left ventricular posterior wall thickness, and end-diastolic and end-systolic left ventricular diameters were also measured. Left ventricular enlargement was defined as an end-diastolic left ventricular diameter of 60 mm or greater in men or 54 mm or greater in women.16 Left ventricular mass was calculated according to the formula by Deveraux and colleagues.17 The degree of mitral regurgitation was determined semiquantitatively based on parameters such as regurgitant jet area, jet profile in continuous-wave Doppler, proximal isovelocity surface area (flow-convergence), vena contracta or pulsed Doppler quantitative flow methods.18 Significant mitral valve stenosis was defined as an estimated mitral valve area of 2 cm2 or less, assessed using either the pressure half-time method or planimetry of the mitral orifice. Significant mitral valve disease was defined as more than mild mitral regurgitation or significant mitral valve stenosis.
Follow-up and outcome measures
Follow-up data were retrieved from health care databases, electronic medical records and death certificates. The outcomes were all-cause mortality and nonfatal ischemic stroke events. Ischemic stroke was defined as codes 433.x1, 434.x1 and 436 of the International Classification of Diseases, Ninth Revision (ICD-9). Patients who underwent cardiac transplantation during follow-up (n = 316) were censored at the time of the procedure.
Statistical analysis
Categorical variables were reported as percentages and comparison between groups based on the χ2 test. Continuous variables were reported as mean (SD) and differences were assessed using the unpaired t test or by one-way analysis of variance (ANOVA), as appropriate. Cumulative event curves were estimated using the Kaplan–Meier method and compared using the log-rank test. We used Cox proportional hazards regression models to assess the associations between left atrial diameter and the outcomes. Hazard ratios (HRs) with 95% confidence intervals (CIs) were estimated. The assumption of proportionality of hazards was verified using log-minus-log survival plots. Separate analyses were performed using left atrial diameter either as a continuous variable or categorized according to the sex-specific criteria of the American Society of Echocardiography. Multivariable analyses were adjusted for the following covariables: age, sex, hypertension, diabetes, hypercholesterolemia, history of smoking, previous stroke or transient ischemic attack, history of atrial fibrillation, severity of mitral regurgitation, mitral valve stenosis, history of mitral valve intervention, history of congestive heart failure, prior myocardial infarction, history of coronary revascularization, left ventricular ejection fraction, end-diastolic left ventricular diameter, left ventricular mass, history of cancer, chronic kidney disease, chronic obstructive pulmonary disease, anticoagulant therapy, referral setting (i.e., admitted to hospital v. outpatient) and year of evaluation. The independent association of left atrial size with mortality was also verified among different subgroups according to age, sex, hypertension, history of atrial fibrillation, left ventricular ejection fraction, left ventricular enlargement, severity of mitral regurgitation and year of evaluation. For these subgroup analyses, the corresponding variable related to the subgroups was removed from the list of adjusting variables.
Results
Clinical and echocardiographic characteristics
The mean age of patients was 61.8 (SD 16.3) years, and 27 847 of them (52.9%) were men. The demographic and clinical characteristics of the 52 639 patients are summarized in Table 1.
Baseline characteristics of study patients (n = 52 639) by left atrial size
The main reasons for performing the echocardiograms included dyspnea or heart failure in 8382 patients (15.9%), heart murmur in 7609 patients (14.5%), palpitations or arrhythmia (including atrial fibrillation) in 10 743 patients (20.4%), search for a cardiac source of embolism in 3540 patients (6.7%), syncope or presyncope in 1962 patients (3.7%), suspected or confirmed endocarditis in 1477 patients (2.8%), chest pain in 1600 patients (3%), evaluation of known valvular heart disease in 2658 patients (5%), assessment of left ventricular wall thickness in 1858 patients (3.5%), electrocardiographic abnormalities (other than rhythm disorders or left ventricular hypertrophy) in 1933 patients (3.7%), suspected or confirmed pericardial disease in 864 patients (1.6%) and suspected cardiomegaly in 821 patients (1.6%).
The mean left atrial diameter was 4.03 (SD 0.77) cm. According to the criteria of the American Society of Echocardiography, left atrial diameter was normal in 26 540 patients (50.4%), mildly enlarged in 12 918 patients (24.5%), moderately enlarged in 7027 patients (13.3%) and severely enlarged in 6154 patients (11.7%).
Patients with left atrial enlargement were older and were more likely to have a history of hypertension, diabetes and atrial fibrillation. Left atrial enlargement was also associated with lower left ventricular ejection fraction and higher left ventricular dimensions and mass (Table 1).
Left atrial size and outcome
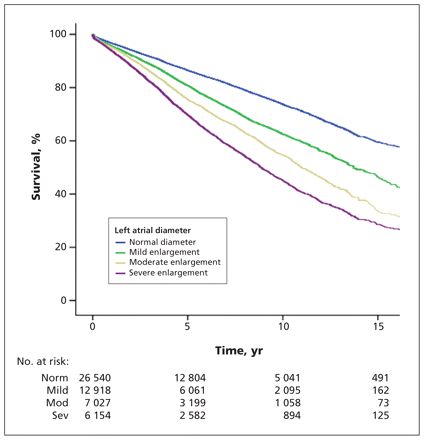
During a mean follow-up of 5.5 (SD 4.1, interquartile range 2.1–8.6) years, a total of 12 527 patients died, and 2314 patients had at least one nonfatal ischemic stroke event. The crude cumulative 10-year survival was 73.7% among patients with normal left atrial size, 62.5% among those with mild left atrial enlargement, 54.8% among those with moderate left atrial enlargement and 45% among those with severe left atrial enlargement (p < 0.001) (Figure 1). This graded association between left atrial size and mortality was consistent in several clinically relevant subgroups, including those stratified according to sex, age, hypertension, history of atrial fibrillation, left ventricular systolic function and left ventricular enlargement (Appendix 1, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.091688/-/DC1).
Kaplan–Meier survival curves stratified according to left atrial diameter (p < 0.001). Normal = left atrial diameter < 4.1 cm in men or < 3.9 cm in women; mild enlargement = 4.1–4.6 cm in men or 3.9–4.2 cm in women; moderate enlargement = 4.7–5.1 cm in men or 4.3–4.6 cm in women; severe enlargement = ≥ 5.2 cm in men or ≥ 4.7 cm in women.
In univariable analysis, left atrial diameter was significantly associated with time to death (HR per 1-cm increment 1.61, 95% CI 1.57–1.66, p < 0.001 in women, and HR 1.43, 95% CI 1.39–1.47, p < 0.001 in men). After covariable adjustment, left atrial diameter remained an independent predictor of mortality either when included as a continuous variable (adjusted HR per 1-cm increment 1.17, 95% CI 1.12–1.22, p < 0.001 in women, and HR 1.09, 95% CI 1.05–1.13, p < 0.001 in men) or as a sex-specific binary variable (HR 1.10, 95% CI 1.06–1.14, p < 0.001). Table 2 shows the association of left atrial size with all-cause mortality in the whole population by severity of left atrial enlargement, based on the criteria the American Society of Echocardiography and stratified according to sex.
Crude and multivariable adjusted hazard ratios for all-cause mortality by the degree of left atrial enlargement in the whole population and stratified according to sex
The association of left atrial enlargement with mortality was also consistent in strata of several subgroups, including age, hypertension, atrial fibrillation, left ventricular ejection fraction, left ventricular enlargement, mitral regurgitation severity and year of evaluation (Table 3). This association remained significant even when patients with a history of atrial fibrillation, significant mitral valve disease or mitral valve intervention were excluded (Table 3). However, there was evidence of some effect modification. In particular, there was evidence of interaction between left atrial size and left ventricular size, such that the adjusted HRs for all-cause mortality were higher among patients with left ventricular enlargement (HR per 1-cm increment 1.22, 95% CI 1.10–1.34, p < 0.001 in women, and HR 1.29, 95% CI 1.19–1.41, p < 0.001 in men) than in those with normal left ventricular size (HR 1.17, 95% CI 1.11–1.22, p < 0.001 in women, and HR 1.04, 95% CI 1.00–1.08, p = 0.07 in men).
Prognostic value of left atrial size* in adjusted multivariable models for the prediction of mortality across a range of baseline characteristics
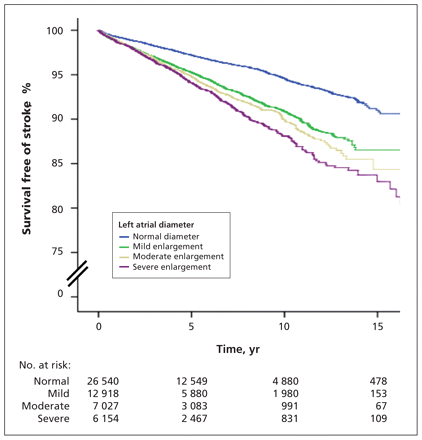
The cumulative 10-year rate of nonfatal ischemic stroke events was 5.4% among patients with normal left atrial size, 9.1% among those with mild enlargement, 10.1% among those with moderate enlargement and 11.9% among those with severe enlargement (p < 0.001, Figure 2). After multivariable adjustment, the association of left atrial diameter with ischemic stroke remained significant among women (HR per 1-cm increment 1.25, 95% CI 1.14–1.37, p < 0.001) but not among men (HR 0.96, 95% CI 0.88–1.06, p = 0.43). When left atrial size was categorized for women according to the criteria of the American Society of Echocardiography and normal left atrial size was used as the reference category, there was also a graded and significant association with ischemic stroke (adjusted HR for mild left atrial enlargement 1.24, 95% CI 1.05–1.46, p = 0.01, HR for moderate left atrial enlargement 1.32, 95% CI 1.10–1.59, p = 0.003, and HR for severe left atrial enlargement 1.59, 95% CI 1.32–1.91, p < 0.001).
Survival free of stroke according to left atrial size (p < 0.001). Normal = left atrial diameter < 4.1 cm in men or < 3.9 cm in women; mild enlargement = 4.1–4.6 cm in men or 3.9–4.2 cm in women; moderate enlargement = 4.7–5.1 cm in men or 4.3–4.6 cm in women; severe enlargement = ≥ 5.2 cm in men or ≥ 4.7 cm in women.
Interpretation
Our study shows that, in a large population of patients referred for transthoracic echocardiography, left atrial size has a graded and independent association with all-cause mortality in both sexes and with ischemic stroke in women.
A number of population-based studies have assessed the value of left atrial size for predicting mortality and cardiovascular events. Benjamin and colleagues9 evaluated 3099 patients age 50 years and older, and found that left atrial diameter was a predictor of death in both sexes. Nagarajarao and coauthors12 reported that left atrial size was a predictor of mortality in African Americans, but the relation of left atrial diameter with stroke was not significant after adjustment for left ventricular hypertrophy and left ventricular ejection fraction. Kizer and coworkers19 found that left atrial diameter was a predictor of fatal and nonfatal cardiovascular events in a population of 2804 American Indians. Laukkanen and colleagues11 found that left atrial diameter was associated with cardiovascular death; however, this association did not remain significant after adjustment for left ventricular mass. The association between left atrial diameter and all-cause mortality in multivariable analysis was not significant either.11 Finally, Benjamin and coauthors,9 and Di Tullio and coworkers,20 reported that left atrial size was a predictor of ischemic stroke in men, but not in women, which contrasts with our results; differences in the characteristics of the patients included might explain at least part of these discrepancies.
We assessed the value of left atrial size for predicting outcome in patients referred for clinical reasons. This allowed us to investigate the association of left atrial size and outcome in a wide range of clinically relevant subgroups. Our results complement and expand previous observations on the value of left atrial size for predicting outcome in patients with left ventricular systolic dysfunction,21–23 patients with myocardial infarction,24,25 patients with known or suspected coronary artery disease referred for stress echocardiography,26,27 and patients with hypertrophic cardiomyopathy.28
The mechanisms accounting for the association of left atrial size with outcome have not been fully unraveled. Left atrial enlargement may predispose to atrial fibrillation,6 which in turn may increase the risk of embolic events, heart failure and death.29 On the other hand, left atrial dilation may be a marker of the severity of other risk factors for cardiovascular events, such as hypertension,30 left ventricular diastolic dysfunction31 or mitral valve disease.32 These conditions may lead to left atrial pressure or volume overload which, if chronically maintained, may result in left atrial remodeling and enlargement.2 In fact, left atrial size may reflect more accurately the duration and severity of chronic elevations in left ventricular filling pressures than Doppler parameters of diastolic function.3,33 While the latter may be readily altered by transient changes in loading conditions, left atrial size may better reflect the cumulative effect of left ventricular filling pressure over time.
Limitations
Our study has several limitations. Even though we controlled for several important covariables, because this was an observational study, residual confounding may account for at least part of the observed differences in outcome. We did not account for a number of variables which might be related to outcome, such as body mass index, antihypertensive medications or Doppler estimators of left ventricular diastolic function. On the other hand, left atrial diameter was evaluated by either M-mode or two-dimensional echocardiography; although the linear distance measured by both methods is theoretically the same, some discrepancies may occur. Finally, left atrial volume is a more reliable estimator of left atrial size than left atrial diameter,34 and also may be a more accurate predictor of cardiovascular events;35 however, left atrial diameter is more readily available and more widely employed in clinical practice.
Conclusion
Left atrial diameter has a graded and independent association with all-cause mortality in both sexes and with ischemic stroke in women. Further research is warranted to refine the prognostic utility of this simple echocardiographic parameter and to determine the degree to which other indices of left atrial size, such as left atrial volume, may improve this prognostic information.
Footnotes
-
This article has been peer reviewed.
-
Competing interests: None declared.
-
Contributors: Alberto Bouzas-Mosquera, Francisco Broullón and Nemesio Álvarez-García were responsible for the conception and design of the study. Alberto Bouzas-Mosquera and Teresa Gándara-Sambade performed the statistical analysis. Alberto Bouzas-Mosquera, Francisco Broullón, Nemesio Álvarez-García, Elizabet Méndez, Jesús Peteiro, Oscar Prada and Victor Mosquera acquired the data. Alberto Bouzas-Mosquera and Francisco Broullón drafted the manuscript. Nemesio Álvarez-García and Alfonso Castro-Beiras supervised the study. All of the authors critically revised the manuscript for important intellectual content and approved the final version submitted for publication.