Abstract
Background: Although warfarin has been extensively studied in clinical trials, little is known about rates of hemorrhage attributable to its use in routine clinical practice. Our objective was to examine incident hemorrhagic events in a large population-based cohort of patients with atrial fibrillation who were starting treatment with warfarin.
Methods: We conducted a population-based cohort study involving residents of Ontario (age ≥ 66 yr) with atrial fibrillation who started taking warfarin between Apr. 1, 1997, and Mar. 31, 2008. We defined a major hemorrhage as any visit to hospital for hemorrage. We determined crude rates of hemorrhage during warfarin treatment, overall and stratified by CHADS2 score (congestive heart failure, hypertension, age ≥ 75 yr, diabetes mellitus and prior stroke, transient ischemic attack or thromboembolism).
Results: We included 125 195 patients with atrial fibrillation who started treatment with warfarin during the study period. Overall, the rate of hemorrhage was 3.8% (95% confidence interval [CI] 3.8%–3.9%) per person-year. The risk of major hemorrhage was highest during the first 30 days of treatment. During this period, rates of hemorrhage were 11.8% (95% CI 11.1%–12.5%) per person-year in all patients and 16.7% (95% CI 14.3%–19.4%) per person-year among patients with a CHADS2 scores of 4 or greater. Over the 5-year follow-up, 10 840 patients (8.7%) visited the hospital for hemorrhage; of these patients, 1963 (18.1%) died in hospital or within 7 days of being discharged.
Interpretation: In this large cohort of older patients with atrial fibrillation, we found that rates of hemorrhage are highest within the first 30 days of warfarin therapy. These rates are considerably higher than the rates of 1%–3% reported in randomized controlled trials of warfarin therapy. Our study provides timely estimates of warfarin-related adverse events that may be useful to clinicians, patients and policy-makers as new options for treatment become available.
Atrial fibrillation is a major risk factor for stroke and systemic embolism, and strong evidence supports the use of the anticoagulant warfarin to reduce this risk.1–3 However, warfarin has a narrow therapeutic range and requires regular monitoring of the international normalized ratio to optimize its effectiveness and minimize the risk of hemorrhage.4,5 Although rates of major hemorrhage reported in trials of warfarin therapy typically range between 1% and 3% per person-year,6–11 observational studies suggest that rates may be considerably higher when warfarin is prescribed outside of a clinical trial setting,12–15 approaching 7% per person-year in some studies.13–15 The different safety profiles derived from clinical trials and observational data may reflect the careful selection of patients, precise definitions of bleeding and close monitoring in the trial setting. Furthermore, although a few observational studies suggest that hemorrhage rates are higher than generally appreciated, these studies involve small numbers of patients who received care in specialized settings.14–16 Consequently, the generalizability of their results to general practice may be limited.
More information regarding hemorrhage rates during warfarin therapy is particularly important in light of the recent introduction of new oral anticoagulant agents such as dabigatran, rivaroxaban and apixaban, which may be associated with different outcome profiles.17–19 There are currently no large studies offering real-world, population-based estimates of hemorrhage rates among patients taking warfarin, which are needed for future comparisons with new anticoagulant agents once they are widely used in routine clinical practice.20
We sought to describe the risk of incident hemorrhage in a large population-based cohort of patients with atrial fibrillation who had recently started warfarin therapy.
Methods
Setting
We performed a population-based cohort study among Ontario residents aged 66 years and older who started warfarin therapy between Apr. 1, 1997, and Mar. 31, 2008. Eligible residents had access to prescription drug coverage, physician care and hospital services through the province’s universal health care system, the Ontario Health Insurance Plan (OHIP). The study was approved by the Research Ethics Board of the Sunnybrook Health Sciences Centre, Toronto, Ontario.
Data sources
We linked multiple administrative datasets from Ontario, Canada’s most populous province. Outpatient prescription records were identified from the Ontario Public Drug Benefit Program Database, which contains comprehensive information on prescription drugs dispensed to about 1.8 million residents of Ontario aged 65 years and older, annually. We identified hospital visits using the Canadian Institute for Health Information’s Discharge Abstract Database and emergency department visits using the National Ambulatory Care Reporting System. These databases contain detailed diagnostic and procedural information regarding all inpatient hospital admissions and visits to emergency departments. We used the OHIP database to identify claims for inpatient and outpatient physician services, and the Registered Persons Database to obtain demographic information for all members of the cohort. We used the Ontario Diabetes Database,21 the Congestive Heart Failure Database and the Hypertension Database22 to calculate the CHADS2 score (determined using criteria validated by Gage and colleagues,23 with 1 point each for congestive heart failure, hypertension, age = 75 yr, and diabetes mellitus, and 2 points for previous ischemic stroke) for each patient in the cohort. The sensitivity and specificity of each of these databases are high.21,22 They are held securely in a linked, deidentified form and analyzed at the Institute for Clinical Evaluative Sciences, and are routinely used for the purposes of studying drug safety.24–27
Identification of the cohort
We identified all patients to whom warfarin was dispensed between Apr. 1, 1997, and Mar. 31, 2008. For each participant, we identified a period of continuous warfarin use beginning with the first prescription dispensed after his or her 66th birthday. To create an inception cohort, we excluded patients with any prescription for warfarin dispensed in the preceding year, and we did not include participants during their first year of eligibility for prescription drug coverage (age 65 yr) to avoid incomplete medication records. To restrict the analysis to patients with atrial fibrillation receiving warfarin therapy, we excluded patients who had not visited a physician, been assessed in an emergency department or been admitted to hospital for atrial fibrillation or flutter (International Classification of Diseases, 9th Revision (Clinical Modification) code 427.3; International Statistical Classification of Diseases and Related Health Problems, 10th revision codes I48.0 or I48.1; OHIP diagnosis code 427) in the 100 days preceding their first prescription for warfarin being dispensed. For all analyses, we defined the date of entry into the cohort as the date on which the first prescription for warfarin meeting these criteria was dispensed.
Warfarin exposure
Because the maximum duration of a prescription eligible for funding by the Ontario Public Drug Program is 100 days, we defined ongoing warfarin therapy by successive refills of a prescription within 180 days to allow for periodic adjustments to doses, lapses in adherence and variable timing of refills. If more than 180 days elapsed between successive prescriptions, patients were deemed to have stopped treatment; such patients were followed for 100 days from the date of the last prescription to identify instances of hemorrhage that may have precipitated the end of treatment.
Risk of hemorrhage
A major hemorrhage was defined as a visit to an emergency department or an admission to hospital for hemorrhage during warfarin therapy. We followed patients until 1 of the following events occurred: a visit to hospital for hemorrhage, the end of warfarin therapy, death, 5 years of follow-up or the end of the study period (Mar. 31, 2010). We stratified hemorrhage anatomically as upper gastrointestinal, lower gastrointestinal, intracranial or other (principally consisting of hemorrhage involving the genitourinary tract or respiratory system). Detailed ICD-9 and ICD-10 codes for these definitions are provided in Appendix 1 (available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.121218/-/DC1), and are based on a validation study that found these codes to have 94% sensitivity and 83% specificity in defining major hemorrhage events.28 If a patient had multiple admissions for hemorrhage during warfarin therapy, we included only the first such event.
Patient characteristics
We determined the following baseline characteristics for members of the study cohort: age, sex, estimated socioeconomic status (based on neighbourhood income quintile),29 residence in a long-term care facility, rural residence and receipt of prescriptions for antiplatelet drugs (acetylsalicylic acid [ASA], clopidogrel, dipyridamole or ticlopidine) or nonsteroidal anti-inflammatory drugs (NSAIDs) in the 120 days preceding entry to the cohort. We defined several indicators of comorbidity in the preceding 3 years, including the Charlson comorbidity index,30,31 the total number of drugs for which patients had received a prescription in the previous year, admission to hospital for renal disease, liver disease, alcoholism or dementia, and previous admissions to hospital involving hemorrhage.
Although we did not have access to laboratory data to assess the quality of anticoagulation, we performed 2 analyses to explore the frequency of international normalized ratio testing in our population. In the first analysis, we restricted our cohort to those patients receiving warfarin for at least 365 days, and measured the average number of prothrombin time tests per person during the first year of follow-up. The second analysis assessed the prevalence of a prothrombin time test in the 7 and 14 days before hemorrhage as a surrogate measure of the adequacy of monitoring the international normalized ratio. To create a comparator group, we randomly assigned index dates to patients without a hemorrhage during the 5-year follow-up that followed the same temporal distribution as the hemorrhage dates. For each patient with a hemorrhage, a patient who did not have a hemorrhage was randomly selected, and the prevalence of a prothrombin time test was assessed for the period before his or her index date.
Statistical analysis
We calculated the crude rate of hemorrhage in each 30-day interval after the start of warfarin therapy. The rate was calculated as the total number of people with a visit to hospital for a hemorrhage divided by the number of people taking warfarin, alive and event-free, at the beginning of the interval. All rates were expressed as a percentage per person-year of follow-up. We stratified analyses by CHADS2 score. We used the Cochrane–Armitage test to identify differences in hemorrhage rates among groups. In a sensitivity analysis, we stratified hemorrhage rates by age (< 75 yr and ≥ 75 yr) to align with typical inclusion criteria in randomized controlled trials. This stratification is important, because patients aged 75 years and older typically have a higher burden of atrial fibrillation, greater frailty and greater propensity for hemorrhagic outcomes than younger patients.
Results
Over the 13-year study period, we identified 266 460 new users of warfarin in Ontario aged 66 years or older. Of these patients, 125 195 (47.0%) had a visit to a physician or an admission to hospital for atrial fibrillation in the preceding 100 days. The median age for starting warfarin therapy was 77 years, and about one-half of the participants were men (Table 1). Overall, 69.0% (n = 86 432) of patients with atrial fibrillation newly receiving warfarin therapy had an estimated CHADS2 score of 2 or more at the start of therapy. The 5-year all-cause mortality for the cohort was 35.7% (n = 44 692).
Baseline characteristics of people starting warfarin therapy after atrial fibrillation was diagnosed
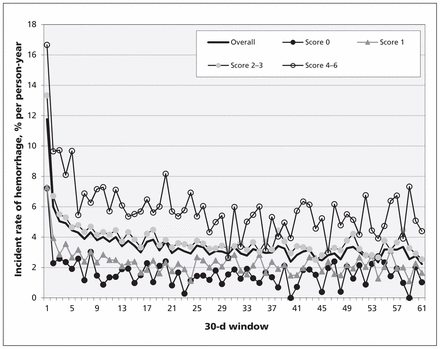
Among patients starting warfarin for atrial fibrillation, the cumulative incidence of hemorrhage was 1.0% (n = 1196) at 30 days, 4.1% (n = 5095) at 1 year and 8.7% (n = 10 840) at 5 years. The overall risk of hemorrhage for the duration of the study was 3.8% (95% confidence interval [CI] 3.8%–3.9%) per person-year (Table 2). The risk was highest during the first 30 days of therapy (11.8% [95% CI 11.1–12.4] per person-year), falling to 3.4% (95% CI 3.4–3.5) per person-year during the remaining follow-up period (Table 2; Figure 1). Crude 5-year rates of hemorrhage varied significantly according to patients’ CHADS2 scores at the start of treatment (Table 2; p < 0.001). Those with a CHADS2 score of 4 or higher had higher than average rates of hemorrhage (16.7% [95% CI 14.3%–19.4%] per person-year in the first 30 d, 6.0% [95% CI 5.7%–6.3%] per person-year thereafter), and patients with a CHADS2 score of less than 2 had lower than average rates of hemorrhage (1.8% [95% CI 1.6%–2.0%] per person-year for a score of 0, and 2.5% [95% CI 2.4%–2.6%] per person-year for a score of 1; Figure 1). Similarly, rates of hemorrhage varied by patient age at the start of treatment. The risk of hemorrhage among patients older than 75 years was 4.6% (95% CI 4.5%–4.7%) per person-year, compared with 2.9% (95% CI 2.8%–3.0%) per person-year among patients aged 75 years or younger (Table 2).
Rates of hemorrhage by length of time since starting warfarin therapy
Incident rate of visits to hospital with hemorrhages in 30-day increments after the start of warfarin therapy among older patients (≥ 66 yr) with atrial fibrillation. Rates are stratified by CHADS2 score at the start of treatment.
Most admissions to hospital involved gastrointestinal hemorrhages (62.6%, n = 6785); more than one-third involved hemorrhages at other sites (38.7%, n = 4190) (Table 2). In total, 1963 (18.1%) patients admitted to hospital with hemorrhages died in hospital or within 7 days of being discharged (data not shown). As expected, mortality was highest among patients admitted to hospital for intracranial hemorrhage (n = 229, 41.7%) as compared with upper gastrointestinal hemorrhages (n = 408, 14.4%), lower gastrointestinal hemorrhages (n = 595, 15.0%) and other hemorrhages (n = 527, 12.6%) (data not shown).
In an analysis of the frequency of monitoring the international normalized ratio among the 77 267 patients receiving warfarin therapy for at least 1 year, we found that the average number of prothrombin time tests per person was 24.1 (standard deviation [SD] 14.9). Furthermore, in an analysis of the quality of such monitoring among the 10 840 patients admitted to hospital with a hemorrhage over 5 years and a randomly selected group of patients who did not have hemorrhages, we found similar rates of testing in the preceding 7 (35.9% v. 31.7%) and 14 days (51.9% and 51.2%) (Appendix 1).
Interpretation
In this population-based study spanning 13 years, we found that the rate of hemorrhage among older patients with atrial fibrillation who were receiving warfarin therapy is roughly half that reported in other observational studies (3.8% v. 6.8%–7.2% per person-year).13–15 This is expected, because previous studies have considered shorter periods of warfarin use (when the risk of hemorrhage is highest) and patients whose condition is managed at anticoagulation clinics, where closer monitoring may lead to more complete identification of major hemorrhagic events. Conversely, the rate of hemorrhage in our study is considerably higher than those reported in randomized controlled trials of warfarin therapy, which have ranged between 1% and 3% per person-year.6,7,9,11 This difference is likely due to the strict inclusion criteria and close monitoring of patients in clinical trials and the average age of our participants being older than that of patients included in trials. This latter factor is emphasized by our sensitivity analysis stratified by age, in which the rate of hemorrhage for the younger group of patients (aged ≤ 75 yr) newly receiving warfarin therapy more closely aligns with results from randomized clinical trials.
Our study shows that the risk of hemorrhage is particularly high during the first 30 days of warfarin therapy, with almost 1% of all new users admitted to hospital for hemorrhage during this period. Furthermore, this risk is amplified among patients with CHADS2 scores higher than 4. The clinical importance of this finding is underscored by the number of patients admitted to hospital for hemorrhage who died either in hospital or shortly after discharge.
Strengths and limitations
Strengths of our study include its large sample, extended follow-up and inclusiveness. Furthermore, our cohort members had comprehensive health insurance, including coverage for prescription drugs, thereby minimizing selection bias. Although the rates of hemorrhage we describe are crude estimates, characterizing these risks in such a large population receiving routine clinical care is an important contribution to the literature.
Our findings derive from Ontario residents aged 66 years and older. Thus, the generalizability of our results to younger patients and people in other jurisdictions is unknown. However, because atrial fibrillation is diagnosed at a mean age of 71 years,32 most people receiving warfarin therapy for new onset of the condition would be eligible for inclusion in our study.
We do not have information regarding medications obtained without a prescription, and thus the true prevalence of the use of ASA and NSAIDs by our cohort is underestimated.
We do not have access to international normalized ratio values during bleeding episodes, which would provide interesting data on risk of hemorrhage for individual patients. However, a population-based study in Ontario recently found the time in therapeutic range among residents of Ontario receiving warfarin therapy to be about 59%.33 In addition, we found similar rates of monitoring the international normalized ratio among patients with and without hemorrhage.
We limited our analyses to patients who started warfarin therapy after a clinical encounter for atrial fibrillation or flutter. Although the sensitivity (77.8%–87.7%) and specificity (99%–100%) of this definition has been shown to be high,34 our approach will undoubtedly exclude some patients with atrial fibrillation, particularly those with paroxysmal atrial fibrillation or flutter.
We are unable to determine which patients received treatment in the community as opposed to in hospital-based anticoagulation clinics, which could help guide future research.
Finally, our study examined the incidence of hemorrhage resulting in a visit to hospital, and therefore excludes minor events not resulting in a visit to hospital and severe events leading to pre-hospital death. Consequently, our rates of hemorrhage underestimate the true overall rates in this population. Furthermore, although the codes used to identify major hemorrhage are highly sensitive and specific, care must be taken when comparing hemorrhage rates between studies given the varying definitions of major bleeding events.
Conclusion
In this large cohort of older patients with atrial fibrillation, we found that rates of hemorrhage are highest during the first 30 days after the start of warfarin therapy, and that almost 1 in 5 such patients who have a hemorrhage requiring admission to hospital died either in hospital or shortly after discharge. Our study provides timely estimates of warfarin-related adverse events that may be useful to clinicians, patients and policy-makers in light of newly approved and emerging anticoagulant therapies.
Acknowledgement
The authors thank Brogan Inc., Ottawa, Ont., for the use of their Drug Product and Therapeutic Class Database.
Footnotes
Competing interests: Tara Gomes, Chelsea Hellings and David Juurlink have received grant funding from the Ontario Drug Policy Research Network. Muhammad Mamdani is a consultant for AstraZeneca, Bristol-Myers Squibb, Eli Lilly, GlaxoSmithKline, Hoffmann-La Roche, Novartis, Novo Nordisk and Pfizer. No other competing interests were declared.
This article has been peer reviewed.
Contributors: Tara Gomes was involved in the conception and design of the study, the acquisition, analysis and interpretation of data and the drafting of the manuscript. Muhammad Mamdani, Anne Holbrook, J. Michael Paterson and David Juurlink were involved in the conception and design of the study, the interpretation of data and critical revision of the manuscript. Chelsea Hellings was involved in the conception and design of the study and the critical revision of the manuscript. All of the authors approved the final version of the manuscript submitted for publication.
Funding: This study was supported by a grant from the Ontario Ministry of Health and Long-Term Care (MOHLTC) Drug Innovation Fund and the Institute for Clinical Evaluative Sciences (ICES), a nonprofit research institute sponsored by the Ontario MOHLTC. The opinions, results and conclusions reported in this paper are those of the authors and are independent from the funding sources. No endorsement by ICES or the Ontario MOHLTC is intended or should be inferred.