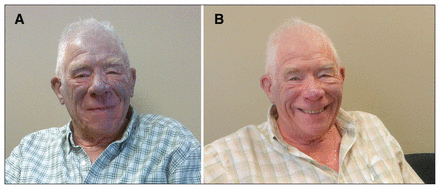
A73-year-old man noticed progressive bluish skin discolouration, predominantly in sun-exposed areas, about six years after he had started taking amiodarone (Figure 1A). He had been taking 400 mg per day, with a total cumulative dose of about 876 g at six years and 2900 g by the time of his assessment (about 18 years since the initiation of amiodarone). The patient had a history of sustained monomorphic ventricular tachycardia after having had an inferior myocardial infarction. We recommended a reduction in the dose of amiodarone, followed by cessation of the drug, in addition to the use of sunscreen and protective clothing. One year after the patient stopped taking amiodarone, his skin showed marked improvement (Figure 1B), and he had no recurrence of ventricular tachycardia.
(A) Photograph of a 73-year-old man presenting with a bluish-grey skin discolouration, predominantly in sun-exposed areas. (B) Near-complete resolution of the discolouration was seen one year after amiodarone was stopped.
Although photosensitivity reactions due to amiodarone are common, bluish-grey skin discolouration is an uncommon adverse effect of amiodarone, occurring in less than 3% of patients undergoing chronic therapy.1 Cutaneous biopsy of discoloured skin has shown lysosomal membrane-bound dense bodies similar to lipofuscin.2 Amiodarone may accelerate normal cellular autophagocytosis, leading to increased production of lipofuscin, which accumulates in lysosomes. Phototoxic damage explains the distribution of the discolouration in sun-exposed areas.3 Alternatively, discolouration may be due to drug deposition within dermal cells in sun-exposed skin.4
There appears to be a dose threshold, and the effect is reversible if the total daily dose is reduced to 200 mg per day or less, with adequate sun protection.5 All patients taking amiodarone should be counselled on sun avoidance and the importance of sunscreen application and protective clothing. In addition, using the lowest possible dose of amiodarone will minimize the risk of adverse effects. Although other adverse effects of amiodarone require monitoring, they have no known association to this bluish-grey skin discolouration.
Footnotes
Competing interests: None declared.
This article has been peer reviewed.
The authors have obtained patient consent.