Abstract
Background: High-sensitivity troponin assays are now available for clinical use. We investigated whether early measurement with such an assay is superior to a conventional assay in the evaluation of acute coronary syndromes.
Methods: Patients presenting to an emergency department with chest pain who did not have ST-segment elevation were prospectively recruited from November 2007 to December 2010. Patients underwent serial testing with a conventional cardiac troponin I assay. Samples were also obtained at presentation and two hours later for measurement of troponin T levels using a high-sensitivity assay. The primary outcome was diagnosis of myocardial infarction on admission; secondary outcomes were death, myocardial infarction and heart failure at one year.
Results: Of the 939 patients enrolled in the study, 205 (21.8%) had myocardial infarction. By two hours after presentation, the high-sensitivity troponin T assay at the cut-off point of the 99th percentile of the general population (14 ng/L) had a sensitivity of 92.2% (95% confidence interval [CI] 88.1%–95.0%) and a specificity of 79.7% (95% CI 78.6%–80.5%) for the diagnosis of non–ST-segment myocardial infarction. The sensitivity of the assay at presentation was 100% among patients who presented four to six hours after symptom onset. By one year, the high-sensitivity troponin T assay was found to be superior than the conventional assay in predicting death (hazard ratio [HR] 5.4, 95% CI 2.7–10.7) and heart failure (HR 27.8, 95% CI 6.6–116.4), whereas the conventional assay was superior in predicting nonfatal myocardial infarction (HR 4.0, 95% CI 2.4–6.7).
Interpretation: The high-sensitivity troponin T assay at the cut-off point of the 99th percentile was highly sensitive for the diagnosis of myocardial infarction by two hours after presentation and had prognostic utility beyond that of the conventional assay. To rule out myocardial infarction, the optimal time to test a second sample using the high-sensitivity troponin T level may be four to six hours after symptom onset, but this finding needs verification in future studies before it can become routine practice.
For novel cardiac markers to be clinically useful in diagnosing acute coronary syndromes, they need to show their incremental utility beyond that of existing markers, with therapeutic implications designed to improve patient care. Recent improvement in the performance of troponin assays to comply with current guidelines for the diagnosis of acute myocardial infarction1 has resulted in a new generation of assays with enhanced clinical sensitivity that are now available for use in clinical care. Assays with high sensitivity have been shown to detect myocardial injury earlier2–8 and identify more patients at risk of future adverse outcomes8–10 than conventional assays.
We conducted a study to assess whether early measurement (at presentation and two hours later) with a high-sensitivity troponin T assay could (a) effectively rule out myocardial infarction without the need for later measurement of troponin levels and (b) identify more patients at risk of adverse cardiac events within one year follow-up compared with a conventional troponin assay.
Methods
Study design
Our study is a secondary analysis from a multinational Asia–Pacific collaboration to study the utility of early measurement (at presentation and two hours later) of multiple biomarkers and electrocardiograms in patients with chest pain assessed as low risk, as a tool to rule out adverse cardiac events such as myocardial infarction.11 In our study, we evaluated the performance of early measurement with a high-sensitivity troponin T assay in all patients recruited irrespective of their pretest risk.
Patients presenting to the emergency department between 0530 and 2000 between November 2007 and December 2010 were recruited by research nursing staff. Those with symptoms suggestive of cardiac ischemia (acute chest, epigastric, neck, jaw or arm pain or discomfort or pressure without an apparent noncardiac source12) were included. Patients were excluded if they were less than 18 years old, had ST-segment elevation on an electrocardiogram, were unable to provide informed consent, were unwilling to participate or would not be available for follow-up.
Cardiac troponin T levels were measured with a high-sensitivity assay at presentation and two hours later. Troponin I levels were measured with a conventional assay at presentation and at least six hours later as per hospital protocol, and at two hours after presentation as per the study protocol. Electrocardiograms were recorded at presentation and at least six hours later as well as during episodes of symptoms and if requested by the medical staff. The decision to perform stress testing, coronary angiography and any other management plan was at the discretion of the attending clinician without knowledge of the high-sensitivity troponin T assay results.
Patients were followed for one year by telephone contact, review of patient notes and a search of the National Health Index database (identifies national hospital attendances using an alphanumeric identifier unique to each patient).
The research protocol was approved by the Upper South A Regional Ethics Committee of the New Zealand Ministry of Health. All participants gave informed consent.
Troponin assays
The reference test was the conventional troponin I assay (Architect Troponin I, Abbott Diagnostics, Chicago, Illinois). Blood samples for clinical decision-making were obtained at presentation and at 2 hours and 6–12 hours after presentation; they were sent in tubes coated with lithium heparin for measurement of troponin I. The assay’s limit of detection is 0.01 μg/L, the 99th percentile in a general population is 0.028 μg/L, and the coefficient of variation of less than 10% is 0.032 μg/L. The decision cut-off point, as recommended by the manufacturer, is 0.03 μg/L.
For the high-sensitivity assay (Elecsys troponin T assay, Roche Diagnostics, Indianapolis, Indiana), blood samples were collected at presentation and two hours later into tubes containing ethylenediaminetetraacetic acid; they were centrifuged, and the plasma was stored frozen at −80°C for later analysis in a blinded fashion in batches. For the high-sensitivity assay, the limit of blank (the highest apparent analyte concentration expected to be found in a sample containing no analyte) is 3 ng/L, the limit of detection is 5 ng/L, the 99th percentile in the general population is 14 ng/L, and the coefficient of variation of less than 10% is 13 ng/L. The decision cut-off point, as recommended by the manufacturer and the universal definition of myocardial infarction,1 is the 99th percentile.
Adjudication
Patient data were recorded according to the American College of Cardiology’s key data elements and definitions for measuring the clinical management and outcomes of patients with acute coronary syndromes,13 and to standardized guidelines for reporting data for patients with acute coronary syndromes.14 Diagnoses on admission and at follow-up were independently adjudicated by one of us (S.J.A.), who was unaware of the results of the high-sensitivity troponin T assay.
Outcome measures
The primary outcome measure was a diagnosis of non–ST-segment elevation myocardial infarction on admission. The diagnostic criteria were a troponin I level of 0.03 μg/L or higher plus (a) a rise or fall in the troponin I level of at least 20% from baseline to peak;15 (b) objective evidence of myocardial ischemia (new ST-segment deviation of at least 0.5 mm or T-wave inversion of at least 1 mm in two or more contiguous leads on resting electrocardiogram); or (c) significant coronary artery disease on coronary angiography (stenosis of 70% or greater in at least one coronary artery or revascularization).
The secondary outcome measures were all-cause mortality, nonfatal myocardial infarction (defined as above but including both ST- and non–ST-segment elevation myocardial infarctions) and admission because of heart failure (pulmonary edema on chest radiograph or symptoms of heart failure with an elevated brain natriuretic peptide level) between discharge and one year.
Statistical analysis
Continuous variables are presented as medians and interquartile ranges, and categorical variables as numbers and percentages.
We calculated the sensitivity, specificity, positive predictive value, negative predictive value and accuracy of the high-sensitivity troponin T assay at presentation and two hours after presentation for diagnosing non–ST-segment elevation myocardial infarction using the cut-off points of the 99th percentile, the limit of detection and the limit of blank. We also calculated the sensitivity of the assay at presentation according to the time from onset of symptoms.
We constructed receiver operating characteristic curves for the high-sensitivity troponin T assay for the diagnosis of non–ST-segment elevation myocardial infarction, and for the high-sensitivity troponin T and conventional troponin I assays for adverse events by one year.
Differences in event-free survival were assessed by means of Kaplan–Meier survival curves. Using the log-rank test, we compared these differences between three groups of patients: those who had a positive troponin I result (irrespective of their high-sensitivity troponin T level); those who had a negative troponin I result and a positive high-sensitivity troponin T result; and those who had a negative result for both assays.
We used Cox proportional hazards models to generate hazard ratios (HRs). In one model, we adjusted by age and sex for adverse events in the three groups described above, with the reference group being patients who had a negative result for both assays. In another model, we adjusted by type of assay using the forward conditional method to identify the incremental benefit or the superiority of one assay over the other. The net gain or loss in false-negative and false-positive results for adverse events was calculated.
Results
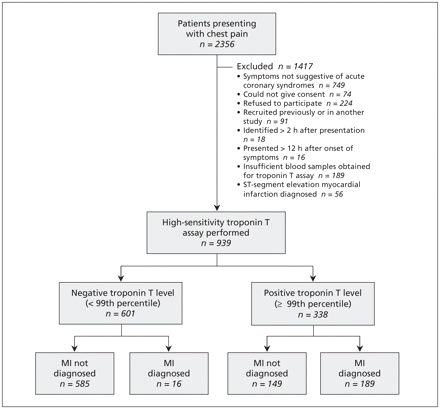
We recruited 939 patients (Figure 1). The baseline characteristics of the patients are shown in Table 1. A total of 205 (21.8%) of the patients received a diagnosis of non–ST-segment elevation myocardial infarction.
Recruitment of patients presenting with chest pain, and results of early measurement of high-sensitivity troponin T levels according to recommended cut-off point of 99th percentile of the general population (14 ng/L). MI = non–ST-segment elevation myocardial infarction.
Characteristics of 939 patients presenting with chest pain and no ST-segment elevation who underwent early measurement of cardiac troponin levels with a conventional troponin I assay and a high-sensitivity troponin T assay
Diagnostic utility
At presentation, the high-sensitivity troponin T level was positive (≥ 99th percentile) in 181 (88.3%) of the 205 patients with non–ST-segment elevation myocardial infarction; it was at or above the limit of detection (≥ 5 ng/L) in 192 (93.7%) and above the limit of blank (≥ 3 ng/L) in 196 (95.6%). At two hours after presentation, the high-sensitivity troponin T level was positive in 189 (92.2%), detected in 196 (95.6%) and above the limit of blank in 202 (98.5%) of the 205 patients. Among the 16 patients who had a diagnosis of non–ST-segment elevation myocardial infarction but whose high-sensitivity troponin T level was below the 99th percentile, the median peak troponin I level was 0.10 (interquartile range [IQR] 0.06–0.18) μg/L.
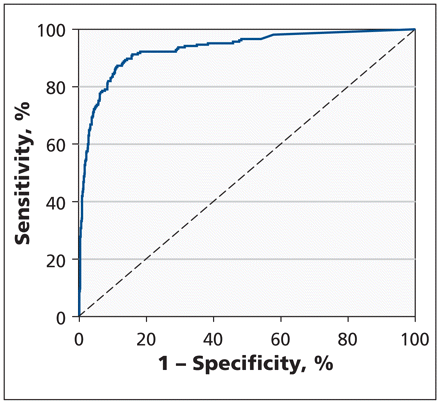
The sensitivity, specificity, positive predictive value, negative predictive value and diagnostic accuracy of the high-sensitivity troponin T assay at different cut-off values at presentation and two hours later are shown in Table 2. The optimum cut-off value for the assay for non–ST-segment elevation myocardial infarction by receiver operating characteristics curve (Figure 2) was 17 ng/L, which corresponded to a sensitivity of 90.7% and a specificity of 84.5%. The level at which there was 98% sensitivity was 3 ng/L.
Performance of high-sensitivity troponin T assay in diagnosing myocardial infarction at presentation and two hours later
Receiver operating characteristics curve for the high-sensitivity troponin T assay at two hours after presentation for the diagnosis of non–ST-segment elevation myocardial infarction. Area under the curve = 0.93 (95% confidence interval 0.91–0.95).
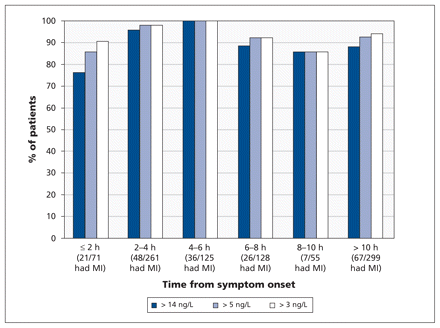
The median time from symptom onset to presentation was 6.3 hours (IQR 3.3–13.3). Of the 16 patients with a false-negative result based on the 99th percentile cut-off point, 6 presented early (≤ 4 hours) and 7 presented late (≥ 10 hours). Figure 3 shows the sensitivity of the high-sensitivity troponin T assay at presentation at the different cut-off points according to the time from symptom onset. The assay had 100% sensitivity at all cut-off values among patients who presented four to six hours after symptom onset.
Sensitivity of the high-sensitivity troponin T assay at presentation at different cut-off points for the diagnosis of non–ST-segment elevation myocardial infarction (MI), by time from symptom onset. Cutoff points: 14 ng/L = 99th percentile of the general population; 5 ng/L = limit of detection (the lowest quantity or concentration of an analyte that can be reliably detected with a given analytical method); 3 ng/L = limit of blank (the highest apparent concentration of an analyte expected to be found in a sample containing no analyte).
Prognostic utility
Overall, 246 patients had a positive troponin I result (irrespective of their high-sensitivity troponin T level), 582 had a negative result for both assays, and 111 had a negative troponin I result and a positive high-sensitivity troponin T result. In the last group, the median level of high-sensitivity troponin T was 21 (IQR 17–30) ng/L.
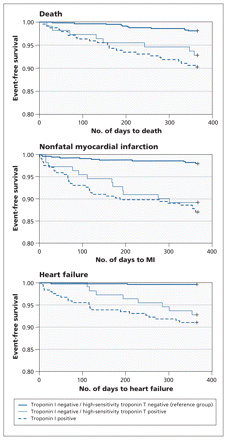
Figure 4 shows the Kaplan–Meier event-free survival curves by one year. The rates of adverse events (death from any cause, nonfatal myocardial infarction and heart failure) in the first year after discharge were significantly lower among patients who had a negative result for both assays (reference group) than among patients who had a positive result for either troponin (p < 0.001). The rates among patients who had a positive troponin I result did not differ significantly from the rates among patients who had a negative troponin I result and a positive high-sensitivity troponin T result (p > 0.1 for each comparison). In the Cox model, the HRs adjusted by age and sex showed graded risk stratification. Compared with patients who had a negative result for both assays (reference group), patients whose troponin I level was negative but whose high-sensitivity troponin T level was positive were more likely to have a nonfatal myocardial infarction (HR 4.5, 95% confidence interval [CI] 2.0–10.3) and heart failure (HR 11.9, 95% CI 2.5–56.9) within the first year after discharge; they were also more likely to die within the first year (HR 1.8, 95% CI 0.7–4.4), although the likelihood was not statistically significant. Patients whose troponin I result was positive, regardless of their high-sensitivity troponin T result, were significantly more likely than those in the reference group to die (HR 2.9, 95% CI 1.4–6.0), to have a nonfatal myocardial infarction (HR 5.7, 95% CI 2.9–11.3) and to have heart failure (17.2, 95% CI 4.0–74.8) within the first year.
Kaplan–Meier survival curves for adverse outcomes (death, nonfatal myocardial infarction and heart failure) by one year after presentation according to results of conventional troponin I and high-sensitivity troponin T assays.
Compared with the troponin I assay, the high-sensitivity troponin T assay identified more patients at risk of adverse outcomes by one year: 32 versus 24 of the 43 patients who died, 37 versus 32 of the 56 patients who had a nonfatal myocardial infarction and 30 versus 22 of the 32 patients who had heart failure by one year. Among the 43 patients who died, there were 11 false-negative results with the high-sensitivity troponin T assay versus 19 with the troponin I assay (net gain 18.6% of those who died) and 306 versus 222 false-positive results (net loss 9.4% of those who did not die). Among the 56 patients who had a nonfatal myocardial infarction, there were 19 false-negative results with the high-sensitivity assay versus 24 with the troponin I assay (net gain 8.9% of those who had nonfatal myocardial infarction) and 301 versus 214 false-positive results (net loss 9.9% of those who did not have the outcome). Among the 32 patients admitted to hospital with heart failure, there were 2 false-negative results with the high-sensitivity assay versus 10 with the conventional assay (net gain 25.0% of those admitted with heart failure) and 308 versus 224 false-positive results (net loss 9.3% of those who were not admitted with heart failure).
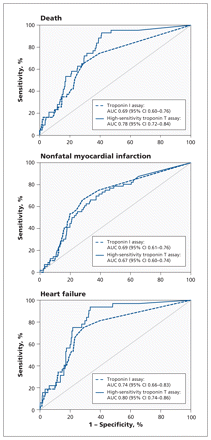
Figure 5 shows the receiver operating characteristics curves for the two assays for the prediction of adverse events by one year. The high-sensitivity troponin T assay performed superiorly to the troponin I assay in predicting death (p = 0.003) and heart failure (p = 0.04) by one year, but not nonfatal myocardial infarction (p = 0.5). In the Cox model, we found no net gain by using both assays for the prediction of any outcome. The high-sensitivity troponin T assay had an incremental utility beyond that of the troponin I assay for predicting death (HR 5.4, 95% CI 2.7–10.7) and heart failure (HR 27.8, 95% CI 6.6–116.4), whereas the troponin I assay was superior to the high-sensitivity assay for predicting the outcome of nonfatal myocardial infarction (HR 4.0, 95% CI 2.4–6.7).
Receiver operating characteristics curves for the high-sensitivity troponin T assay and the conventional troponin I assay for predicting adverse events (death, nonfatal myocardial infarction or heart failure) by one year after presentation. AUC = area under the curve, CI = confidence interval.
Interpretation
The high-sensitivity troponin T assay at the cutoff point of the 99th percentile was highly sensitive for the diagnosis of non–ST-segment elevation myocardial infarction by two hours after presentation. However, the false-negative rate was substantial at 7.8%, which suggests that later measurement with the assay would still be required. This false-negative rate appeared to be due mainly to poor performance of the assay among patients presenting early (≤ 4 hours after symptom onset). Instead of performing a second test with the high-sensitivity troponin T assay at two hours after presentation, it may be more reliable to perform the second test four to six hours after symptom onset. Use of a lower cut-off point, such as the limit of blank, to rule out myocardial infarction (but not rule it in, which by definition requires an elevation in the troponin T level at or above the 99th percentile) improved the sensitivity of the assay, with a false-negative rate of 1.9%. More patients at risk of adverse events (death and heart failure in particular) were identified with the high-sensitivity troponin T assay than with the conventional troponin I assay.
Compared with previous assays, sensitive troponin assays allow earlier detection of myocardial infarction,3–7 even in patients presenting early after symptom onset.3,5,7,14 The result may be fewer hospital admissions, with the potential for savings in health care costs and reduced overcrowding in emergency departments and hospitals.14 In previous studies, the sensitivity of the high-sensitivity troponin T assay at presentation was reported to be 61.5% (84.6% at three hours) among patients with confirmed non–ST-segment elevation acute coronary syndromes (incidence of non–ST-segment myocardial infarction 44.8%);7 88% among patients with low- to intermediate-risk chest pain (incidence of non–ST-segment elevation myocardial infarction 3.2%);15 and 95% among patients similar to those in our study (incidence of non–ST-segment elevation myocardial infarction 17%).5 This compares with the baseline sensitivity of 88.3% in our study.
Current international guidelines recommend serial troponin measurement at presentation and again at 8–12 hours after symptom onset16 or 6–9 hours after presentation.1 Only recently has there been national recommendations in Australia and New Zealand to change the timing of the second sample to three hours after presentation for patients presenting more than six hours after symptom onset when high-sensitivity assays are used, although evidence for this proposal was level C.17
Our study showed that the high-sensitivity troponin T assay at the 99th percentile cut-off was not sensitive enough to detect all patients with non–ST-segment elevation myocardial infarction by two hours after presentation. This early measurement may have failed to identify patients with “minor” infarctions who had only low-level increases in troponin I. Also, the sensitivity of the high-sensitivity troponin T assay was less reliable among patients who presented earlier than four hours after symptom onset, achieving 100% sensitivity among those presenting four to six hours after symptom onset. Such findings support the aforementioned Australasian guidelines17 and could result in a change in international protocols, with a second troponin test performed four to six hours earlier than currently recommended.16 This strategy requires further validation. The high-sensitivity troponin T assay’s sensitivity appears to fall again among patients presenting later than six hours after symptom onset. These decreases may have been because the numbers of patients in many of these periods were small and the 95% CIs large.
A recent cohort study suggested that the high-sensitivity troponin T assay be used at a very low cut-off point (3 ng/L = limit of blank) to rule out myocardial infarction on presentation.18 The sensitivity for myocardial infarction in that study was 100% even among patients presenting early and 99.8% in the subsequent prospective audit. The authors suggested that up to 17.5% of patients would not require further troponin testing. In our study, which had a similar incidence of myocardial infarction, the sensitivity of the high-sensitivity troponin T assay at a cut-off point of 3 ng/L was only 95.6% at presentation, rising to 98.1% two hours later. The use of this cut-off point requires testing in other cohorts.
Troponin is well known for its prognostic utility.2,3,14 In our study, 100 patients (10.6%) experienced adverse events during the one-year follow-up period. There were significantly more events among those with an elevated troponin I level and those with a positive high-sensitivity troponin T assay result even when the troponin I result was negative, than among patients in whom neither marker was elevated. Hazard ratio analysis adjusted by age and sex showed that patients with a negative troponin I result and a positive high-sensitivity troponin T result were less likely to experience adverse events than patients with a positive troponin I result. This was possibly because risk was graded by the magnitude of the troponin elevation. The high-sensitivity troponin T levels were lower in patients with a negative troponin I result than in those with an elevated troponin I level.
Further analysis of the incremental utility of the high-sensitivity troponin T assay over the conventional troponin I assay, or vice versa, suggested that only one troponin assay has utility for risk stratification. The high-sensitivity troponin T assay was superior in stratifying patients at risk of death and heart failure, but the conventional troponin I assay was superior at stratifying patients at risk of nonfatal myocardial infarction. This may have been because more patients had a positive high-sensitivity troponin T result because of noncoronary causes. It also may have been because the troponin I assay was used to adjudicate the diagnosis of myocardial infarction, which may have overestimated the performance of the troponin I assay.
Past studies that used first-generation troponin assays showed that outcomes were improved when treatment decisions such as revascularization were guided by troponin results. In our study, more patients had a positive result with the high-sensitivity troponin T assay than with the conventional assay and were shown to be at risk of future adverse events. This prognostic utility has the potential to translate into improved outcomes, but only prospective randomized trials directly comparing high-sensitivity and conventional troponin assays for clinical decision-making will answer this question.
Limitations
The incidence of non–ST-segment elevation myocardial infarction during our study period was consistent with that seen at this institution and in New Zealand in general. However, it may be higher than that seen elsewhere and should be considered if applying these findings to a different population. Blood samples for the high-sensitivity troponin T assay were taken only at presentation and two hours later because this was a substudy analysis; samples obtained after two hours would have been useful. The troponin I assay is an imperfect “gold standard” for assessment of another troponin assay. Although this represents a major limitation, it is the comparative method used in similar studies.
Conclusion
The high-sensitivity troponin T assay at the cutoff point of the 99th percentile was highly sensitive for the diagnosis of myocardial infarction by two hours after presentation. Current international protocols for the diagnosis of acute myocardial infarction could possibly be accelerated by up to four to six hours. Randomized controlled trials to prospectively validate the timing of serial samples are required. The use of a lower cut-off point for the high-sensitivity troponin T assay may identify a subgroup of patients who do not require further troponin testing, but again this needs validation in a randomized controlled trial. Compared with the conventional troponin I assay, the high-sensitivity troponin T assay identified more patients at risk of future adverse events, and its use may therefore influence outcomes.
Acknowledgements
Sally Aldous was supported by a National Heart Foundation of New Zealand Research Fellowship. Mark Richards holds the National Heart Foundation of New Zealand Chair of Cardiovascular Studies and is Director of the Cardiovascular Research Institute, National University Health System, Singapore.
Footnotes
Competing interests: Louise Cullen has received consultancy fees from Abbott Diagnostics, research grants from Abbott Diagnostics and Alere, speaker fees from Alere, Pfizer and Radiometer Pacific, and travel assistance from Boehringer Ingelheim. Richard Troughton has received speaker fees from St. Jude Medical Inc. No competing interests declared by Sally Aldous, Mark Richards and Martin Than.
This article has been peer reviewed.
Contributors: All of the authors were involved in the study concept and design. Sally Aldous was responsible for the analysis and interpretation of the data and the drafting of the manuscript. All of the authors revised the manuscript critically for important intellectual content and approved the final version submitted for publication.
Funding: This study was supported by the National Heart Foundation of New Zealand and the Health Research Council of New Zealand. The high-sensitivity troponin T assays used in this study were provided free of charge by the manufacturer.