Abstract
BACKGROUND: Patients who continue to smoke after acute coronary syndrome are at increased risk of reinfarction and death. We previously found use of varenicline to increase abstinence 24 weeks after acute coronary syndrome; here we report results through 52 weeks.
METHODS: The EVITA trial was a multicentre, double-blind, randomized, placebo-controlled trial of varenicline for smoking cessation in patients admitted to hospital with acute coronary syndrome. Participants were randomly assigned (1:1) to receive varenicline or placebo for 12 weeks, in conjunction with low-intensity counselling. Smoking abstinence was assessed via 7-day recall, with biochemical validation using exhaled carbon monoxide. Participants lost to follow-up or withdrawn were assumed to have returned to smoking.
RESULTS: Among the 302 participants, abstinence declined over the course of the trial, with 34.4% abstinent 52 weeks after acute coronary syndrome. Compared with placebo, point estimates suggest use of varenicline increased point-prevalence abstinence (39.9% v. 29.1%, difference 10.7%, 95% confidence interval [CI] 0.01% to 21.44%; number needed to treat 10), continuous abstinence (31.1% v. 21.2%, difference 9.9%, 95% CI −0.01% to 19.8%) and reduction in daily cigarette smoking by 50% or greater (57.8% v. 49.7%, difference 8.1%, 95% CI −3.1% to 19.4%). Varenicline and placebo groups had similar occurrence of serious adverse events (24.5% v. 21.9%, risk difference 2.7%, 95% CI −7.3% to 12.6%) and major adverse cardiovascular events (8.6% v. 9.3%, risk difference −0.7%, 95% CI −7.8% to 6.5%).
INTERPRETATION: Varenicline was efficacious for smoking cessation in this high-risk patient population. However, 60% of patients who received treatment with varenicline still returned to smoking. Trial registration: ClinicalTrials.gov, no. NCT00794573
Smoking cessation is of critical importance for patients immediately following an acute coronary syndrome, substantially reducing negative outcomes such as reinfarction and death among successful quitters.1–4 The acute hospital admission is a “teachable moment” in which smokers may be more receptive to information about quitting and more motivated to make a quit attempt.5 It is also a valuable opportunity to provide smokers, particularly those who may not have been in recent contact with a health care provider, with assistance in quitting and to organize longitudinal support for a quit attempt, as patients return for regular follow-up after acute coronary syndrome.
However, there is limited evidence available concerning the use of smoking cessation therapies in patients with acute cardiovascular conditions. Although the use of nicotine replacement therapies (e.g., nicotine patch and nicotine gum) is common, there is a lack of clinical trial data supporting the efficacy and safety of these therapies in patients with cardiovascular disease.6,7 Additionally, data from a number of trials suggest that bupropion is not efficacious in this population.8–10 Varenicline, a partial agonist of α4β2 nicotinic acetylcholine receptors, has been widely studied in the general population, with efficacy appearing to meet or exceed that of nicotine replacement therapies and bupropion.11–13 Along with bupropion, varenicline has been shown to increase abstinence in patients with stable cardiovascular disease; however, its efficacy in patients with acute coronary syndrome was previously unknown.6 We have recently shown that use of varenicline increases smoking abstinence 24 weeks after acute coronary syndrome.14 Here we report evidence concerning the efficacy and safety of varenicline for smoking cessation 52 weeks after acute coronary syndrome.
Methods
Study design
The Evaluation of Varenicline in Smoking Cessation for Patients Post–Acute Coronary Syndrome (EVITA) trial is a randomized, placebo-controlled trial that enrolled 302 participants across Canada and the United States. The methods of this trial have been previously described in detail.15 Enrolment took place during hospital admission for acute coronary syndrome, including myocardial infarction and unstable angina with clinically significant coronary artery disease. To be eligible, patients had to be motivated to quit and have smoked 10 or more cigarettes per day for the past year. Patients with a history of mental illness were excluded, as was standard practice for clinical trials at the time of study design, owing to concerns about serious neuropsychiatric events in individuals with pre-existing psychiatric disease taking varenicline. Those who consented to participate and met the inclusion and exclusion criteria were randomly assigned 1:1 to varenicline tartrate (0.5 mg daily for 3 days, then 0.5 mg twice daily for 4 days, followed by 1.0 mg for 11 weeks) or matching placebo for 12 weeks, combined with low-intensity counselling for smoking cessation and relapse prevention. Treatment was initiated in-hospital.
Follow-up and end points
Participants were followed for 52 weeks after acute coronary syndrome, with telephone calls at weeks 1, 2 and 8, and clinic visits at weeks 4, 12, 24 and 52. Self-reported smoking abstinence was biochemically validated at clinic visits using exhaled carbon monoxide (Micro 3/4 Smokerlyzer, Bedfont Scientific Ltd.). Measurement of exhaled carbon monoxide is a practical and reliable method of assessing smoking status, which correlates with the frequency and quantity of cigarettes smoked.16 Although its ability to detect nonrecent smoking (> 8 h) is limited,16 participant perception of greater efficacy may increase the validity of self-report data.17
The prespecified primary end point was smoking abstinence at week 24 (previously reported).14 Point-prevalence abstinence was defined as self-reported abstinence in the past week (no smoking, not even a puff), with exhaled carbon monoxide levels of 10 ppm or less. Continuous abstinence was defined as self-reported abstinence in the past week at all follow-ups since baseline, with exhaled carbon monoxide levels of 10 ppm or less at all clinic visits. Reduction in daily cigarette consumption by 50% or greater was also assessed.
Statistical analysis
We used the intention-to-treat principle for all analyses. As in other trials of smoking cessation, our analyses assumed that participants who were lost to follow-up or withdrew returned to smoking at their baseline rate. Sensitivity analyses were conducted to examine the effect of this assumption. Participants who died were censored from analyses after death. The number needed to treat (NNT) represents the number of patients with acute coronary syndrome who needed to receive treatment with varenicline for 1 patient to be abstinent. Statistical analyses were performed using SAS software (version 9.3).
Ethics approval
This study complied with the Declaration of Helsinki; locally appointed ethics committees approved the research protocol, and informed consent was obtained from participants before enrolment.
Results
Participant characteristics
Participants enrolled in the trial (n = 302) (Figure 1) were primarily male (75.2%) with a mean age of 55.0 (standard deviation [SD] 9.3) years, and had been smoking for the past 3–4 decades (35.9 [SD 11.6] yr). At the time of their acute coronary syndrome (56.0% ST-segment elevation myocardial infarction, 37.8% non–ST segment elevation myocardial infarction and 6.3% unstable angina), participants were smoking a mean of 21.4 (SD 10.6) cigarettes per day. Most (80.4%) had a score of 4 or greater on the Fagerström Test for Nicotine Dependence, indicating moderate or severe dependence on nicotine. Most had made at least 1 previous attempt to quit smoking (81.8%) and many had another smoker living at home (41.7%). Participant characteristics (Table 1) were well-balanced between the varenicline and placebo groups,14 with the exception of the proportion of participants with another smoker at home, which was higher in the varenicline group (48.3% v. 35.1%, difference 13.2%).
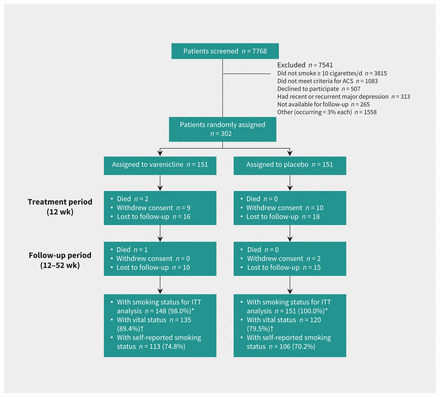
Randomization and follow-up of study patients. *Includes all patients except those who died. For the intention-to-treat (ITT) analysis, patients who were lost to follow-up or withdrew were assumed to have returned to smoking at their baseline rate. †In the event of loss to follow-up, vital status was obtained from chart review if possible. ACS = acute coronary syndrome.
Characteristics of smokers with acute coronary syndrome, by treatment group
Drug course
The median length of hospital stay was 3 (interquartile range [IQR] 2–4) days, and the median time from admission to the first dose of study medication was 2 (IQR 1–3) days. At the conclusion of the treatment period (12 wk), most participants reported taking 2 pills per day (per the protocol): 70.5% of participants in the varenicline group compared with 82.1% of participants in the placebo group (p = 0.07; adherence data available for 218 participants). An additional 7.1% in the varenicline group and 2.8% in the placebo group reported taking 1 pill per day (as may be recommended to reduce adverse effects). Study personnel and participants were unaware of treatment allocation until the conclusion of the trial, with participant guesses of treatment assignment no better than chance (49.1% in the varenicline group and 48.8% in the placebo group correctly guessed their treatment assignment at week 12).
Smoking cessation
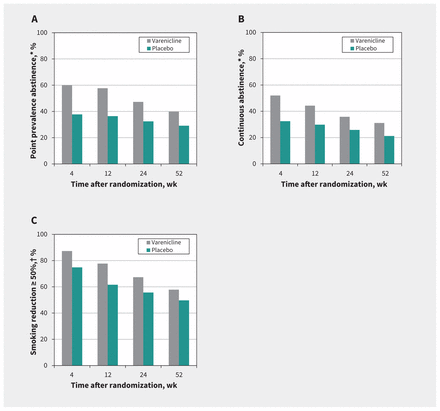
Point-prevalence smoking abstinence declined over the course of the trial (Figure 2), from a high of 58.6% of all participants at week 1 to a low of 34.4% of all participants at week 52. There was an immediate difference in abstinence between the varenicline and placebo groups beginning at the first follow-up telephone call at week 1: 66.2% versus 51.0%, respectively. The difference in point-prevalence abstinence remained significant at all follow-ups through the end of the 12-week treatment period and 52-week follow-up (differences and 95% confidence intervals [CIs] presented in Appendix 1, supplementary Table 1, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.170377/-/DC1). A similar trend was observed for continuous abstinence, with significant differences observed between groups throughout the 12-week treatment period, although CIs included the null at week-24 and -52 follow-up (Appendix 1, supplementary Table 1).
Trends in point-prevalence smoking abstinence from baseline to week 52. All analyses were intention-to-treat. Patients who withdrew consent or were lost to follow-up were assumed to have returned to smoking at their baseline rate. Participants who died were censored after the time of death. Participants were considered abstinent if they abstained from smoking in the 7 days before the visit through a self-report of 0 cigarettes smoked per day, confirmed by exhaled carbon monoxide levels of 10 ppm or less at clinic visits (available for 94.7%, 87.3%, 79.7% and 81.7% of self-reported abstinent participants at weeks 4, 12, 24 and 52, respectively).
Smoking abstinence and reduction data from clinic visits at weeks 4, 12, 24 and 52 are shown in Figure 3. Abstinence data at the primary end point of week 24 have been previously reported.14 At week 24, use of varenicline was found to increase point-prevalence smoking abstinence (47.3% v. 32.5%) and reduction in daily cigarette consumption by 50% or greater (67.4% v. 55.6%) compared with placebo. Continuous abstinence rates were 35.8% in the varenicline group versus 25.8% in the placebo group at week 24. At week 52, point-prevalence smoking abstinence was significantly higher in the varenicline group (39.9.%) compared with the placebo group (29.1%) (difference 10.7, 95% CI 0.01% to 21.44%; NNT 10). Rates of continuous abstinence at week 52 were 31.1% in the varenicline group versus 21.2% in the placebo group (difference 9.9%, 95% CI −0.01% to 19.8%). Reduction in daily cigarette consumption by 50% or greater was 57.8% in the varenicline group compared with 49.7% in the placebo group (difference 8.1%, 95% CI −3.1% to 19.4%). Among only those who continued to smoke, reduction by 50% or greater was 29.5% in the varenicline group and 29.0% in the placebo group (difference 0.5%, 95% CI −12.3% to 13.4%).
Smoking cessation and reduction by treatment group. All analyses were intention-to-treat. Participants who withdrew consent or were lost to follow-up were assumed to have returned to smoking at their baseline rate. Participants who died were censored after the time of death. A) Smoking cessation: point-prevalence abstinence. Participants were considered abstinent if they abstained from smoking in the 7 days before the visit through a self-report of 0 cigarettes smoked per day, confirmed by exhaled carbon monoxide levels of 10 ppm or less (available for 94.7%, 87.3%, 79.7% and 81.7% of self-reported abstinent participants at weeks 4, 12, 24 and 52, respectively). Risk differences: 4 weeks, 22.3 (95% confidence interval [CI] 11.2 to 33.3); 12 weeks, 21.3 (95% CI 10.3 to 32.3); 24 weeks, 14.9 (95% CI 3.9 to 25.8); 52 weeks, 10.7 (95% CI 0.01 to 21.4). B) Smoking cessation: continuous abstinence. Participants were considered abstinent if they abstained from smoking through a self-report of 0 cigarettes smoked per day, confirmed by exhaled carbon monoxide levels of 10 ppm or less (available for 96.9%, 91.0%, 82.6% and 88.5% of self-reported continuously abstinent participants at weeks 4, 12, 24 and 52, respectively) at all follow-up visits. Risk differences: 4 weeks, 19.6 (95% CI 8.6 to 30.5); 12 weeks, 14.5 (95% CI 3.7 to 25.3); 24 weeks, 10.0 (95% CI −0.4 to 20.4); 52 weeks, 9.9 (95% CI −0.1 to 19.8). C) Reduction in daily cigarette consumption of 50% or more from baseline; self-report only. Risk differences: 4 weeks, 12.4 (95% CI 3.7 to 21.2); 12 weeks, 16.1 (95% CI 5.9 to 26.4); 24 weeks, 11.7 (95% CI 0.8 to 22.7); 52 weeks, 8.2 (95% CI −3.1 to 19.4). *Adjusted for loss to follow-up. †In self-reported daily cigarette consumption from baseline.
Abstinence was biochemically validated using exhaled carbon monoxide at clinic visits. A total of 81.7% of participants self-reporting abstinence at week 52 provided biochemical validation (owing to completion of some clinic visits by telephone, as necessary to obtain participant follow-up). Of these, only 1 participant had a carbon monoxide reading greater than 10 ppm and was classified as having returned to smoking. Sensitivity analyses were conducted for smoking abstinence and reduction at weeks 4, 12, 24 and 52, which included only participants who returned for follow-up (in order to assess the effect of the assumption that participants who withdrew or were lost to follow-up returned to smoking at their baseline rate). These unadjusted analyses found similar differences in smoking abstinence and reduction end points (Appendix 1, supplementary Tables 1 and 2).
Low-intensity counselling and nonstudy cessation therapies
As part of the trial, participants received low-intensity counseling, with a mean of 51.4 (SD 33.5) minutes for all participants from baseline to week 52. Participants were also permitted to seek counselling outside of the study; however, only 2.7% of participants did so at any point (equal in each treatment arm). Following the 12-week treatment period, participants who had relapsed were also permitted to use nonstudy pharmacotherapy treatments for smoking cessation. Use of a nonstudy treatment at any point in the trial was 18.3% overall (14.9% in the varenicline group v. 21.8% in the placebo group). Participants reported using (non–mutually exclusive categories): 6.6% e-cigarette, 6.0% nicotine patch, 4.3% short-acting nicotine replacement therapy (e.g., gum, inhaler or lozenge), 2.6% varenicline and 2.0% other.
Safety
The occurrence of serious adverse events was similar between trial arms (Table 2). Overall, there were 93 serious adverse events between baseline and week 52, occurring in 70 patients (37 in the treatment arm [24.5%] and 33 in the placebo arm [21.9%]). A total of 30 major adverse cardiovascular events were reported in 27 patients (13 in the treatment arm [8.6%] and 14 in the placebo arm [9.3%]), including 17 myocardial infarctions, 11 hospital admissions for unstable angina and 2 cardiovascular deaths (2 patients experienced both myocardial infarction and unstable angina, and 1 patient had 2 separate instances of unstable angina). Both deaths occurred within 30 days of treatment discontinuation in patients in the varenicline arm (1 because of congestive heart failure 40 days after randomization and 18 days after discontinuing study drug; the other due to sudden death 25 days after randomization in a patient presumed to have taken the study drug until the time of death). A single noncardiovascular death occurred in a patient in the varenicline arm due to a perforated ulcer 63 days after study drug discontinuation.
Serious adverse events by treatment group
Interpretation
Use of varenicline significantly increased abstinence compared with placebo (39.9% v. 29.1%) 52 weeks after hospital admission for acute coronary syndrome. This finding is important given the substantial reduction in cardiovascular risk conferred by smoking cessation in this high-risk population.1,2,18,19 In addition, rates of serious adverse events (24.5% v. 21.9%) and major adverse cardiovascular events (8.6% v. 9.3%) were similar between varenicline and placebo arms. This suggests that varenicline is safe for use in these patients. However, new strategies for smoking cessation are still needed, given that 60% of smokers who received treatment with varenicline returned to smoking by 1 year after their acute coronary syndrome.
The use of varenicline in the EVITA trial differed in one significant way from the usual method of administration recommended. Typically, patients are instructed to begin taking the drug 8–14 days before a quit date to allow time for titration and bioaccumulation.20 In the EVITA trial, whereas the dose was titrated as recommended over 7 days, participants were already not smoking at the time of randomization (due to hospital admission) and were instructed not to resume smoking on discharge. This approach appears to have been efficacious, given that the differences between groups were apparent from week 1, with only half of participants in the placebo arm abstinent (51.0%) compared with two-thirds of participants in the varenicline arm (66.2%). However, this approach could have resulted in increased “slips” (i.e., temporary lapses to smoking) early in the trial, limiting our ability to detect significant differences between groups in regard to continuous smoking abstinence.
Safety concerns related to neuropsychiatric events and cardiovascular events associated with the use of varenicline21–25 have largely been resolved,26–30 with high-quality evidence suggesting that varenicline is safe for use in both populations with prior psychiatric illness and history of cardiovascular disease. In particular, compelling evidence has been generated by the EAGLES trial (n = 8144 participants),31 which found no difference in the incidence of neuropsychiatric events between individuals (with or without psychiatric disease) receiving varenicline, bupropion, nicotine patch or placebo. There were very few serious cardiovascular adverse events reported, with no apparent differences between groups. Likewise, the CATS trial (n = 4595 participants) found no difference in the incidence of major adverse cardiovascular events between the same groups.32 In addition, Sterling and colleagues conducted a meta-analysis of 38 randomized controlled trials (n = 12 706 participants), which found no difference in serious cardiovascular adverse events when comparing varenicline with placebo in populations either with or without cardiovascular disease.33 Overall, evidence accumulated from these trials and meta-analyses provides strong support that use of varenicline is safe for smoking cessation.
Limitations
Our trial had several potential limitations. First, enrolment was restricted to patients with acute cardiovascular disease who were motivated to quit smoking. Therefore, our findings may not be generalizable to patients with stable cardiovascular disease or to the general population of hospital-admitted smokers after acute coronary syndrome. Second, counselling provided to both groups was of low intensity, which may have reduced quit rates overall. However, our findings may represent a real-world scenario in which patients receive (or seek) little psychosocial intervention. Participants were encouraged to use additional counselling resources outside of the study, and only 2.7% did so at any point. Lastly, our findings may underestimate the efficacy of varenicline over time for several reasons. The first is that participants who had not successfully quit were permitted to use other smoking cessation therapies during trial follow-up. Given that 21.8% of participants in the placebo arm compared with 14.9% in the varenicline arm used other cessation therapies, this may have resulted in an underestimation of the efficacy of varenicline. Likewise, an imbalance in proportion of participants with another smoker living at home (48.3% in the varenicline arm v. 35.1% in the placebo arm) may have resulted in increased relapse in the varenicline group.
Conclusion
We examined the efficacy of varenicline versus placebo, initiated in-hospital and in conjunction with low-intensity counselling, for smoking cessation 52 weeks after acute coronary syndrome. We found that varenicline is efficacious for smoking cessation; however, 60% of patients who received treatment with varenicline still returned to smoking by 1 year. If varenicline were used routinely after acute coronary syndrome, for every 10 smokers who received treatment there would be 1 less smoker a year later.
Acknowledgements
The authors’ thanks go to the patients across North America who participated in the EVITA trial, as well as the research teams who conducted enrolment and follow-up. The authors additionally thank Joseph Mancini, Natalie Zacchia, and Bettina Habib for their assistance with trial coordination and data management, as well as Patrick Bélisle for his help with data analysis.
Footnotes
↵* A complete list of EVITA Investigators is provided in Appendix 1, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.170377/-/DC1.
Competing interests: Shamir Mehta reports funding from AstraZeneca, Boston Scientific, Bayer and Abbott. Beth Abramson has received grants or research support from AstraZeneca and Sanofi; honoraria from Amgen, AstraZeneca, Boehringer Ingelheim, Bristol–Myers Squibb, Novartis, Fournier, Merck, Pfizer, Servier and Sanofi; and consulting fees from Amgen, Bayer, Boehringer Ingelheim, Sanofi and Servier. She authored Heart Health for Canadians. Mark Eisenberg, Payam Dehghani, François Grondin and Mina Madan received honoraria from Pfizer for providing continuing medical education on smoking cessation. No other competing interests were declared
This article has been peer reviewed.
Contributors: The steering committee, comprising Mark Eisenberg (Chair), Beth Abramson, Iqbal Bata and Mina Madan, made substantial contributions to the conception or design of the work. All of the authors made substantial contributions to the acquisition, analysis or interpretation of data. Sarah Windle drafted the article, and all remaining authors revised it critically for important intellectual content. All of the authors gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Funding: EVITA was an investigator-initiated trial that received funding and the study drug and placebo from Pfizer Inc. Pfizer had no role in the design, conduct, analysis, interpretation of data, or reporting of the EVITA trial.
- Accepted November 28, 2017.