Abstract
Background: Human parathyroid hormone (hPTH)(1–34) was approved in 2004 for the treatment of severe osteoporosis. Members of the Osteoporosis Canada clinical guidelines committee conducted a systematic review of randomized controlled trials (RCTs) to assess the efficacy and safety of hPTH for fracture prevention in postmenopausal women and men with osteoporosis.
Methods: We searched MEDLINE, EMBASE, HTA, Current Contents and the Cochrane Controlled Trials Registry for published data from 1966 to February 2005. A systematic literature search for RCTs was conducted using the Cochrane Collaborative approach. We identified 12 trials that randomly assigned patients either to hPTH or placebo or to hPTH or an active comparator and were at least 1 year in duration. Outcomes included change in bone mineral density (BMD), fractures, back pain and adverse events. Two independent reviewers abstracted data on study characteristics and outcomes.
Results: hPTH(1–34) significantly increases lumbar spine BMD, with smaller increases at the femoral neck and total hip. hPTH(1–84) significantly increases lumbar spine BMD. The data show a significant reduction in both vertebral and nonvertebral fractures with hPTH(1–34) in postmenopausal women with previous vertebral fractures. There were no data on fractures comparing the approved dose of hPTH(1–34) with active comparators.
Interpretation: There is Level I evidence that hPTH(1–34) significantly increases BMD at all skeletal sites except the radius and significantly reduces the risk of new vertebral and nonvertebral fractures in postmenopausal women with prior fractures.
The 2002 Osteoporosis Canada clinical guidelines committee reviewed the evidence for several classes of drugs that reduce bone resorption and inhibit bone formation.1 Anabolic therapies are capable of inducing new bone formation through an increase in bone modelling and remodelling.2,3 Human parathyroid hormone (hPTH) is an 84-amino acid peptide hormone that plays a key role in the maintenance of calcium homeostasis. hPTH binds to a target cell surface G-protein-coupled hPTH/hPTHrP receptor, which results in activation of adenylate cyclase and phospholipases and increased intracellular levels of cyclic AMP and calcium.4 Intermittent hPTH given by subcutaneous injection has been shown to exert potent anabolic effects on the skeleton.5 hPTH increases the rate of bone remodelling and results in a positive remodelling balance, leading to thicker osteons (structural units of remodelled bone).6,7 New bone formation occurs on quiescent surfaces and, as a result, trabecular architecture comes to more closely resemble normal bone.8,9 hPTH(1–34) induces new periosteal bone apposition, which results in the enlargement of the outer circumference of tubular bones such as the radius.10 This bone apposition results from decreased osteoblast apoptosis and enhanced differentiation of osteoblasts from preosteoblasts. Bisphosphonates preserve existing skeletal microarchitecture but do not restore it toward a more normal structure.11,12 Increases in bone mass with bisphosphonates are most likely due to enhanced secondary mineralization of preformed osteons.13
Two forms of recombinant hPTH have been evaluated in clinical trials, hPTH(1–34) and the intact 84-amino acid form, hPTH(1–84). hPTH(1–34) is approved for the treatment of severe osteoporosis.14 hPTH(1–84), although not currently approved, has been evaluated in clinical trials.15 hPTH(1–84) includes a C terminus, which may have discrete biologic properties and may therefore have different biologic actions from hPTH(1–34).3,4
In this article we present the results of our systematic review of the literature on the efficacy and safety of hPTH therapy.
Methods
The hPTH working group included members of the Osteoporosis Canada Clinical Guidelines Committee and scientific consultants with Osteoporosis Canada who have expertise in hPTH or in clinical epidemiology or both. This review was approved by all members of the Clinical Guidelines Committee.
We included RCTs of at least 1-year duration that compared hPTH with a placebo or with an active comparator and involved 1 of 3 distinct populations: postmenopausal women with osteoporosis, postmenopausal women with corticosteroid-induced osteoporosis or men with osteoporosis. The primary outcome was the effect of hPTH(1–34) or hPTH(1–84) on either bone mineral density (BMD) or fractures. Secondary outcomes were back pain and quality of life.
The Cochrane Collaborative approach for identifying RCTs guided our search strategy.16 We searched MEDLINE, EMBASE, HTA, Current Contents and the Cochrane Controlled Trials Registry from 1966 to September 2005 (see online Appendix 1, available at www.cmaj.ca/cgi/content/full/175/1/52/DC1). Citations of relevant articles and reviews were examined to identify other potential trials. Two reviewers (A.C. and N.Z.) evaluated all titles and abstracts for eligibility. Full articles of relevant trials were obtained to make final judgments about inclusion.
The 2 reviewers independently abstracted data on study characteristics, results and methodologic quality. Data examined included study design, population, dose, treatment duration, percent change in BMD and the number of subjects with fractures. Volumetric BMD data and biochemical markers were not included as outcomes in this review, since they are not widely accepted as surrogate outcomes. Missing data were requested from study investigators or sponsors. We decided a priori not to pool the BMD and fracture data from the various trials since different hPTH preparations (i.e., hPTH[1–34] v. hPTH[1–84]) may have different modes of action, and doses may not be comparable. For example, 20 μg of hPTH(1–34) may be roughly equivalent to 50 μg of hPTH(1–84) on a molar basis, but differences in peptide length may cause differences in tissue absorption or receptor activation.4
RCTS were classified as level 1 (an RCT of adequate sample size, with blinding of subjects and assessors) or as level 2 (RCT that does not meet level 1 criteria, owing to a small sample or methodologic limitations).
Results and interpretation
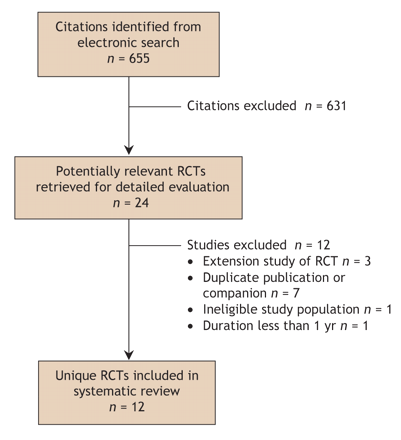
Fig. 1: Flow of articles through the systematic review. RCT = randomized controlled trial.
A total of 655 potentially relevant citations were identified and screened (Fig. 1). Of these, 24 were potentially eligible. Twelve trials were subsequently excluded: one included premenopausal women,17 10 were a duplicate report, companion or follow-up of a primary RCT,10,18–26 and in one BMD outcomes were for less than a year.27 Twelve published RCTs were included in this review.28–39 Nine trials involved postmenopausal women (Table 1),28–34,38,39 of which one involved women with corticosteroid-induced osteoporosis (Table 2).34 Three trials involved men with osteoporosis (Table 3).35–37 Ten trials evaluated hPTH(1–34)29–31,33–39 and 2 trials hPTH(1–84).28,32
Table 1.
Table 2.
Table 3.
Two trials had losses to follow-up of over 20%,29,33 8 trials had losses between 5% and 20%,28,30,31,34,35,37–39 and 2 had less than 5%.32,36 Allocation concealment was adequately reported in 2 trials30,39 and unclear in the remaining trials. Seven trials were double-blind.28,31–33,36–38
hPTH in postmenopausal women
In a large trial comparing hPTH(1–34) with calcium and vitamin D among women with severe osteoporosis and prior fractures, Neer and associates found a significant reduction in new vertebral and nonvertebral fractures associated with hPTH (Table 1 and Table 4).31 With 20 μg and 40 μg of hPTH(1–34) the relative risk (RR) of new vertebral fractures was 0.35 (95% confidence interval [CI] 0.22–0.55) and 0.31 (95% CI 0.19–0.50) respectively (absolute risk reduction [ARR] 9%–10%). Treatment with 20 μg hPTH(1–34) resulted in a RR of 0.47 (95% CI 0.25–0.88) for nonvertebral fractures and an ARR of 3%. In other hPTH(1–34) trials, the absolute number of fractures was too small to achieve significance28,38,39 (Table 4). Body and associates found a reduction in nonvertebral fractures with 40 μg/d hPTH(1–34) compared with 10 mg/d alendronate (4.1% v. 13.7%, p = 0.042).33
Table 4.
Six trials compared the effect of hPTH(1–34) with placebo or an active comparator on BMD (Table 4). Doses of hPTH(1–34) ranged from 20 μg/d to 50 μg/d and treatment durations from 1 to 3 years. There was a consistent increase in lumbar spine and femoral neck BMD but little effect on BMD at the distal radius. Increases in lumbar spine BMD with hPTH(1–34) were significant and ranged from 9.7% to 10.3% with 20 μg/d and from 13.7% to 14.3% with 40 μg/d (Table 4). Changes in femoral neck BMD were significant but smaller, ranging from 2.8% to 3.9% in the 20 μg/d group and 4.5%–5.1% in the 40 μg group.
Two trials compared the effect of hPTH(1–84) with that of placebo or alendronate. Increases in lumbar spine BMD ranged from 6.3% to 7.8% after one year with 50–100 μg of hPTH(1–84), which were larger but not significantly different than increases seen with alendronate.28,32 Increases in femoral neck BMD with hPTH(1–84) were small compared with placebo and less than that seen with alendronate (Table 4).
hPTH compared with bisphosphonates
Two trials compared hPTH(1–34) 20 μg or 40 μg with alendronate 10 mg/d in postmenopausal women.33,38 Body and associates compared 40 μg of hPTH(1–34) with alendronate 10 mg daily. Increases of lumbar spine and femoral neck BMD were greater with hPTH than with alendronate (p < 0.001). At the ultradistal radius the increase with alendronate was 1.4% compared with 0.23% with 40 μg hPTH(1–34) (p < 0.001).33
In a study by McClung and associates, hPTH(1–34) 20 μg/d resulted in significantly greater increases (10.3%) in lumbar spine BMD than with alendronate 10 mg/d.38
Combination therapy
Three trials evaluated hPTH therapy combined with either a bisphosphonate or calcitonin.29,30,32 In an RCT involving 238 postmenopausal women with osteoporosis, alendronate combined with hPTH(1–84) did not result in larger increases in lumbar spine BMD than with hPTH alone.32 Cosman and associates compared the effect of hPTH(1–34) 25 μg and hormone replacement therapy (HRT) with that of HRT alone in 52 postmenopausal women who had been taking HRT for at least a year.30 Three years of combination therapy resulted in increases in lumbar spine and total hip BMD of 13.4% and 4.4% respectively compared with a nonsignificant increase with HRT alone.30 In a small 2-year RCT, Hodsman and associates compared cyclical hPTH(1–34) 50 μg followed by placebo calcitonin with hPTH followed by sequential salmon calcitonin (Table 1). Combined sequential therapy with calcitonin did not offer any benefits for BMD over cyclical hPTH alone.29
hPTH therapy after prior bisphosphonate therapy
Although the anabolic effects of hPTH may be accentuated if bone resorption is suppressed, there is concern that the decreased remodelling rates induced by bisphosphonates might impair the ability of hPTH to stimulate new bone formation.40 In contrast, Cosman and associates randomly assigned women who had previously taken alendronate to either cyclic or daily hPTH(1–34) in combination with alendronate or to alendronate alone. Significant increases in lumbar spine BMD of 6.1% and 5.4% were noted in the daily and cyclic treatment arms respectively.39
Sequential use of bisphosphonate therapy after cessation of hPTH
After hPTH(1–34) is discontinued, BMD declines; this loss can be prevented with sequential use of a bisphosphonate.18,23 In an RCT of hPTH(1–84) and alendronate, Black and associates randomly assigned women to receive a second year of either alendronate or placebo and found that the alendronate group had significant increases in lumbar spine BMD (4.9%) compared with controls (–1.7%) (p < 0.01).41
Summary
There is level 1 evidence that hPTH(1–34) 20 μg/d and 40 μg/d increase BMD at the lumbar spine and proximal femur and decrease the risk of both vertebral and nonvertebral fractures in postmenopausal women with prior vertebral fractures. The effect of hPTH(1–34) on hip fractures has not been assessed. hPTH(1–34) results in larger increases in lumbar spine BMD than alendronate. There are no head-to-head fracture trials that compare the efficacy of 20 μg of hPTH(1–34) to bisphosphonates in decreasing the risk of fracture.
There is level 1 evidence that hPTH(1–84) increases lumbar spine BMD in postmenopausal women with osteoporosis.
The small increase in femoral neck BMD and decline in total body BMD may reflect a transient imbalance between cortical remodelling and bone formation. The decline in BMD at the distal radius seen in hPTH trials is not felt to be indicative of decreased bone strength and may occur as a result of the increased width of the radius.3 The absence of head-to-head fracture data suggests that hPTH(1–34) be limited to patients at high risk of fractures by virtue of very low BMD and pre-existing fractures.
There is level 1 evidence that combination therapy with alendronate and hPTH(1–84) may blunt the anabolic effect of hPTH on BMD. There are no fracture data comparing the effect of the combination of hPTH and alendronate with that of hPTH alone.
hPTH(1–34) in postmenopausal women with corticosteroid-induced osteoporosis
Lane and associates evaluated the effect of hPTH(1–34) in 51 postmenopausal women taking corticosteroids and HRT. Women were given hPTH(1–34) for one year and then followed for an additional year. hPTH(1–34) 25 μg in combination with HRT increased BMD by 12.6% at the lumbar spine and by 5.2% at the femoral neck after 2 years.34
Summary
There is level 2 evidence that hPTH(1–34) increases lumbar spine BMD in postmenopausal women with corticosteroid-induced osteoporosis. Fracture data are lacking in this population.
hPTH(1–34) in men with osteoporosis
Kurland and associates compared 25 μg hPTH(1–34) with placebo in a small trial involving men with idiopathic osteoporosis. After 18 months, BMD had increased significantly by 13.5% and 2.9% at the lumbar spine and femoral neck respectively. Total hip BMD did not change significantly, and there was a significant decrease at the 1/3 distal radius (–1.2%).36 Orwoll and associates, in an RCT involving 437 men with low BMD, compared 20 μg or 40 μg of hPTH(1–34) with calcium and vitamin D. Of these men, 49% had hypogonadism. After one year, lumbar spine BMD increased by 5.4% with 20 μg and by 8.5% with the 40 μg dose compared with no change with placebo. There was a nonsignificant decrease in nonvertebral fractures with hPTH(1–34) compared with placebo.37 Finkelstein and associates compared hPTH(1–34) 40 μg alone, hPTH(1–34) with alendronate 10 mg/d, and alendronate alone. Increases in lumbar spine and femoral neck BMD were 18.1% and 9.7% respectively in group receiving hPTH(1–34) alone compared with those taking combination hPTH(1–34) and alendronate or alendronate alone (p < 0.001) (Table 4).35
Summary
There is level 1 evidence that hPTH(1–34) increases BMD at the lumbar spine and femoral neck in men with osteoporosis, but there is no data on fractures in this population.
Health-related quality of life and back pain
Neer and associates assessed health-related quality of life with both generic (Nottingham Health Profile) and disease-specific measures (Osteoporosis Assessment Questionnaire) (OPAQ).31 Using the OPAQ, fractures in a subgroup of 365 women were associated with a decline in health-related quality of life. However, there were no significant differences between patients taking hPTH(1–34) and those taking placebo.42 Back pain was reported in 3 hPTH(1–34) trials,31,33,38 and one trial had baseline and follow-up spine radiography.31 A significant reduction in back pain was seen among postmenopausal women in the fracture trial of hPTH(1–34).31 In 2 trials comparing hPTH(1–34) and alendronate 10 mg, hPTH(1–34) was associated with a significant decrease in moderate to severe back pain.33,38 A meta-analysis of individual patient data from 5 trials comparing hPTH(1–34) with comparators found that patients taking hPTH(1–34) had a reduced risk of back pain (pooled RR 0.60, 95% CI 0.55–0.80).43
Summary
There is no evidence of improved health-related quality of life associated with hPTH(1–34) or calcium with vitamin D in postmenopausal women with osteoporosis. There is level 2 evidence that back pain may be reduced in patients given hPTH(1–34).
Adverse events
A potential safety issue with hPTH(1–34) is increased risk of osteosarcoma, which was reported in a life-long carcinogenicity study involving Fischer rats given high-dose hPTH(1–34) from infancy through senescence (8 weeks of age through 2 years).44 Because of this unexpected finding, trials of hPTH(1–34) for postmenopausal women with severe osteoporosis and men with osteoporosis31,33,37 were terminated prematurely by the sponsor. Osteosarcoma was found with all doses, and, in the lower-dose ranges, was first detected after about 20 months of therapy. There have been no reports of osteosarcoma in clinical trial subjects, and although there are isolated case reports of osteosarcoma in patients with long-standing hyperparathyroidism, there is no evidence to suggest that osteosarcoma is of increased frequency in hyperparathyroidism.45,46
Adverse events were included in all trials, but reporting was variable. Nine hPTH(1–34) trials29,31,33–39 reported post-dose hypercalcemia (serum calcium level > 2.6 mmol/L) that ranged from 3% to 11% among patients taking 20 μg of hPTH(1–34) and from 16.8% to 38% among patients taking 40 μg compared with 0%–3% among those taking the comparator.31,33 The overall pooled number of events of hypercalcemia was 282 in 1594 subjects from 7 trials. These episodes were mild: serum calcium levels usually returned to normal within 24 hours, and no clinical sequelae were reported in subjects with transient hypercalcemia. Hypercalcemia rates in the hPTH(1–84) trials ranged from 12% to 15.6%; there were 46 cases among patients taking hPTH out of a total of 340, compared with no cases among those taking the comparator.28,32
Transient hypercalciuria (24-hour urine collection showing calcium level of ≥ 8 mmol/d or urine calcium/creatinine ratio > 1.0) was reported in 6 hPTH(1–34) trials and ranged from 0% to 10% difference between hPTH and comparator.29,31,33,35,37,39 There were no reported increases in renal stones.
A 10% increase in serum creatinine levels was reported in one trial but did not exceed the normal range.29 In another trial, increases in creatinine clearance were reported with hPTH that were nonsignificant compared with increase seen with placebo.28
The frequency of headaches among treatment subjects in hPTH(1–34) trials ranged from 8.1% to 10.8% and was not significantly greater than among control subjects.31,37
hPTH(1–34) 20 μg significantly increased the proportion of patients experiencing dizziness (3%)31 and leg cramps (range 2%–8%),31,33 with a higher proportion in the 40 μg treatment arm. Hyperuricemia was reported in 2 hPTH(1–34) trials31,37 and ranged from 0% to 3% of subjects. Hyperuricemia was associated with gout in 3 subjects taking hPTH(1–84).32
The proportion of subjects with serious adverse events was not significantly different between treatment arms.
PTH treatment causes transient increases in serum calcium, but these episodes usually resolve. If they persist, they can be managed by decreasing calcium and vitamin D supplements.
@ See related article page 48
Footnotes
-
This article has been peer reviewed.
Contributors: Ann Cranney was involved in all phases of the review, including its design, literature search, and data extraction and interpretation, and was responsible for drafting the paper and incorporating revisions. Nicole Zytaruk was involved in reviewing the results of the literature search and in data extraction. Alexandra Papaioannou, David Hanley, Jonathan Adachi, Timothy Murray, David Goltzman and Anthony Hodsman were involved in the conception of the project and the interpretation of the data and results, and provided critical input on various drafts of the paper. All of the authors approved the final version of the manuscript and accompanying recommendations.
Acknowledgements: This work was funded by an unrestricted educational grant from Osteoporosis Canada. Ann Cranney is the recipient of a salary award from the Canadian Institutes of Health Research.
Competing interests: None declared for Nicole Zytaruk and David Goltzman.
Ann Cranney has been a member of the medical advisory boards for Merck Frosst Canada and Zelos Therapeutics and has received speaker fees from Aventis, Procter & Gamble Pharmaceuticals, Eli Lilly and Merck Frosst Canada. Alexandra Papaioannou has received speaker fees from Eli Lilly Canada, Novartis, Merck Frosst Canada, Amgen, Sanofi-Aventis and Procter & Gamble Pharmaceuticals; she has received unrestricted educational grants from Eli Lilly Canada, Merck Frosst Canada, Sanofi-Aventis and Procter & Gamble Pharmaceuticals. David Hanley has received an honorarium from Eli Lilly for being available for consultation during the past 2 years. He has participated in the major clinical trials of the 2 parathyroid hormone preparations that are either approved or under regulatory review for the treatment of osteoporosis; these trials were sponsored by the manufacturers, Eli Lilly and NPS Pharmaceuticals. David Hanley did not receive personal remuneration for his work as an investigator in these trials. He has been on the advisory board for the clinical trials conducted by NPS Pharmaceuticals and was a coauthor of the phase II and III clinical trial manuscripts for hPTH(1-84) but did not receive personal remuneration for his authorship of these articles. He has received speaker fees from Eli Lilly and NPS Pharmaceuticals and honoraria for attendance at their advisory board meetings, some of which were held in association with major scientific conferences. Jonathan Adachi has been a consultant and speaker and has received honoraria and travel assistance from Astra Zeneca, Eli Lilly, GlaxoSmithKline, Merck Frosst, Novartis, NPS Allelix, Pfizer, Procter & Gamble, Roche, Sanofi-Aventis, Servier and Wyeth. Timothy Murray has been a member of the advisory boards for NPS Pharmaceuticals, Eli Lilly Canada, Procter & Gamble and Aventis and has received speaker fees from Eli Lilly Canada and Novartis. He has participated in clinical trials funded by Eli Lilly and Novartis but did not receive personal remuneration in connection with these grants and received an unrestricted educational grant from Merck Frosst. Anthony Hodsman is a member of the medical advisory boards of Eli Lilly Canada, Sanofi Aventis, Zelos Therapeutics, NPS Allelix, Novartis Canada, Servier Canada and Merck Frosst Canada. He has participated in clinical trials funded by Eli Lilly, Novartis, NPS Allelix, Sanofi Aventis and Zelos Therapeutics.