Abstract
Background: The Public Health Agency of Canada reviewed sexual transmission of HIV between serodiscordant partners to support examination of the criminal justice system response to HIV nondisclosure by the Department of Justice of Canada. We sought to determine HIV transmission risk when an HIV-positive partner takes antiretroviral therapy, has a suppressed viral load or uses condoms.
Methods: We conducted an overview and systematic review update by searching MEDLINE and other databases (Jan. 1, 2007, to Mar. 13, 2017; and Nov. 1, 2012, to Apr. 27, 2017, respectively). We considered reviews and studies about absolute risk of sexual transmission of HIV between serodiscordant partners to be eligible for inclusion. We used A Measurement Tool to Assess Systematic Reviews (AMSTAR) for review quality, Quality in Prognosis Studies (QUIPS) instrument for study risk of bias and then the Grading of Recommendations, Assessment, Development and Evaluation (GRADE) approach to assess the quality of evidence across studies. We calculated HIV incidence per 100 person-years with 95% confidence intervals (CIs). We assigned risk categories according to potential for and evidence of HIV transmission.
Results: We identified 12 reviews. We selected 1 review to estimate risk of HIV transmission for condom use without antiretroviral therapy (1.14 transmissions/100 person-years, 95% CI 0.56–2.04; low risk). We identified 11 studies with 23 transmissions over 10 511 person-years with antiretroviral therapy (0.22 transmissions/ 100 person-years, 95% CI 0.14–0.33; low risk). We found no transmissions with antiretroviral therapy and a viral load of less than 200 copies/mL across consecutive measurements 4 to 6 months apart (0.00 transmissions/100 person-years, 95% CI 0.00–0.28; negligible risk regardless of condom use).
Interpretation: Based on high-quality evidence, there is a negligible risk of sexual transmission of HIV when an HIV-positive sex partner adheres to antiretroviral therapy and maintains a suppressed viral load of less than 200 copies/mL measured every 4 to 6 months. Sexual transmissions of HIV have occurred when viral load was more than 200 copies/mL with antiretroviral therapy or condoms alone were used, although the risk remains low. These findings will help to support patient and clinician decision-making, affect public health case management and contact tracing, and inform justice system responses to HIV nondisclosure.
In Canada, an estimated 82% of people living with HIV acquired their infection through sexual transmission.1 Under current Canadian criminal law, people can be prosecuted for not disclosing their HIV-positive status when they engage in sexual activity that poses a “realistic possibility” of HIV transmission.2–5
Previous reviews have shown that both antiretroviral therapy and condoms reduce the possibility of HIV transmission.6–8 Antiretroviral therapy inhibits viral replication with the goal of suppressing viral load to an undetectable level, defined as a plasma HIV-1 RNA level below 20–50 copies/mL.9,10 In 2016, an estimated 86% of the 63 110 people living with HIV in Canada were aware of their infection, 81% of those diagnosed were taking antiretroviral therapy and 91% of those being treated had a suppressed viral load of less than 200 copies/mL.11 Although previous reviews have shown the preventive benefit of antiretroviral therapy, use of condoms or both, they have not included the most recent studies that have changed clinical guidance12–14 and public messaging,15 and could affect Canadian criminal law.
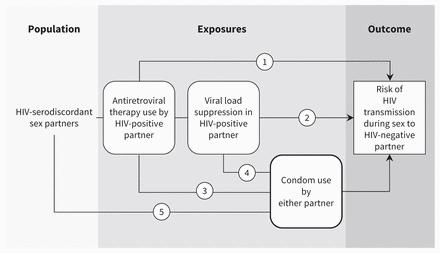
To support the examination of the criminal justice system’s response to HIV nondisclosure by the Department of Justice Canada,16 we synthesized evidence on the absolute risk of HIV transmission during sex between serodiscordant partners. Our objectives were to determine risk when a sex partner who is HIV-positive (Q1) is taking antiretroviral therapy (with varying levels of viral load); (Q2) is taking antiretroviral therapy and has a suppressed viral load; (Q3) is taking antiretroviral therapy and either partner uses condoms (or other barrier methods); (Q4) is taking antiretroviral therapy and has a suppressed viral load, and either partner uses condoms (or other barrier methods); and when (Q5) either partner uses condoms (or barrier methods) alone.
The Public Health Agency of Canada, in consultation with the Department of Justice Canada, chose these questions and the outcome of interest (absolute risk of HIV transmission) as most relevant to informing public health and legal responses.
Methods
We conducted a systematic review of systematic reviews (overview) and a systematic review of more recent studies that were not available for consideration in previously published reviews (update). Figure 1 presents the analytic framework for the overview and update. We prepared unregistered protocols a priori (Appendices 1 and 2, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.180311/-/DC1; summarized in the following section) by following the criteria from the Preferred Reporting Items for Systematic review and Meta-Analysis Protocols (PRISMA-P).17 We followed PRISMA and Cochrane guidance (Supplementary Table S1, Appendix 3, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.180311/-/DC1).18,19
Analytic framework illustrating exposure to preventive measures that reduce the risk of transmission between HIV-serodiscordant sex partners. The numbers in the corresponding pathways illustrate the risk of HIV transmission from the HIV-positive partner to the HIV-negative partner during sex for Q1 to Q5. Note: Q1 = to determine risk when a sex partner who is HIV-positive is taking antiretroviral therapy (with varying levels of viral load); Q2 = to determine risk when a sex partner who is HIV-positive is taking antiretroviral therapy and has a suppressed viral load; Q3 = to determine risk when a sex partner who is HIV-positive is taking antiretroviral therapy and either partner uses condoms (or other barrier methods); Q4 = to determine risk when a sex partner who is HIV-positive is taking antiretroviral therapy and has a suppressed viral load, and either partner uses condoms (or other barrier methods); Q5 = to determine risk when either partner uses condoms (or barrier methods) alone.
Overview review
We developed a comprehensive search strategy (Appendix 1) with assistance from research librarians at Health Canada. We searched MEDLINE, Embase, Global Health and the Cochrane Library from Jan. 1, 2007, to Mar. 13, 2017, in addition to review registries (PROSPERO and Cochrane Library) and reference lists.
We included reviews that provided quantitative estimates of absolute risk of sexual transmission of HIV between serodiscordant sex partners (number of HIV transmissions in a given number of sex acts or person-years). Appendix 1 gives the detailed eligibility criteria.
We performed title and abstract screening, full-text screening, data extraction and quality assessments using the A Measurement Tool to Assess Systematic Reviews (AMSTAR)20 independently and in duplicate, with disagreements resolved by a third reviewer. Our AMSTAR ratings informed the selection of reviews for the update.
Update review
We identified 2 high-quality reviews that addressed Q1 to Q4 to update6,21 (see Appendix 2, section 3.3). Research librarians at Health Canada helped to develop the search strategy for the update that encompassed any sexual orientation or type of partnership. The strategy was externally peer reviewed using Peer Review of Electronic Search Strategies (PRESS).22 We searched MEDLINE, Embase, the Cochrane Register of Controlled Trials (CENTRAL) and Web of Science from Nov. 1, 2012, to Apr. 27, 2017, in addition to trial registries and reference lists. We contacted experts to identify unpublished studies. We did not conduct an update for Q5 because a Cochrane review addressing condom use alone7 was declared in 2012 to be stable and conclusive evidence not requiring further updates.7,23,24
We included studies that reported or provided data to calculate an absolute risk of sexual transmission of HIV between serodiscordant sex partners. These studies included people living with HIV who were taking antiretroviral therapy with monitoring of their viral load and reported on condom use. We combined the primary studies from the update search with those from the reviews.6,21 Appendix 2 gives the detailed eligibility criteria.
We performed title and abstract screening, full-text screening, data extraction and study-level risk of bias assessment using the Quality in Prognosis Studies (QUIPS) tool25 independently and in duplicate, with disagreements resolved by a third reviewer. Our QUIPS ratings informed the risk of bias domain of our overall quality assessments.
We used the Grading of Recommendations, Assessment, Development and Evaluation (GRADE) approach for prognostic studies to assess certainty (i.e., confidence or quality) in the evidence as high, moderate, low or very low.26–28 For questions with no direct evidence, we used indirect evidence from other questions, populations and sex acts to estimate transmission risk and rated down for indirectness.
Statistical analysis
We present descriptive statistics at the study level including aggregate person-years and transmission events. We also describe individual transmission events from all studies in the update. We confirmed that the studies contained independent data. We converted per person-year risk of HIV transmission to incidence per 100 person-years and per-act risk of HIV transmission to incidence per 1000 acts. We calculated incidence of HIV transmission per 100 person-years and exact 95% confidence intervals (CIs) assuming a Poisson distribution.29 We used SAS for all analyses (SAS Enterprise Guide version 5.1, SAS Institute Inc.). When more than 1 cohort was available, we used the sum of transmission events divided by the sum of person-years to calculate a pooled absolute transmission risk (i.e., incidence) and 95% CIs. We assessed statistical heterogeneity visually with forest plots (because we did not examine comparisons between groups)30 to inform the “inconsistency” criterion of our GRADE assessment.
Categories of transmission risk
To facilitate knowledge translation, we assigned risk categories (no risk, negligible risk, low risk and high risk) to our findings using the criteria outlined in the guideline from the Canadian AIDS Society (Supplementary Table S3, Appendix 3).31 We considered 2 criteria when assessing the level of transmission risk associated with an activity: whether there was potential for HIV transmission, and whether there was documented evidence of transmission.31
Ethics approval
Because we systematically reviewed published studies, no ethics approval was required.
Results
In the overview, of 1414 citations screened, we found 12 systematic reviews6–8,21,32–39 that met the eligibility criteria (Supplementary Figure S1, Appendix 3). Table 1 and Supplementary Tables S4–S6 (Appendix 3) summarize characteristics of the included reviews and their key findings (Appendix 4, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.180311/-/DC1 presents full narrative summaries). Our AMSTAR scores ranged from 2 to 9 out of 11 (Supplementary Table S7, Appendix 3).
Characteristics of included reviews
We updated 2 reviews on use of antiretroviral therapy6,21 that included the same 6 studies;40,44–48 2 of those studies had more recent results identified through our update search.49,50 Of 7266 citations screened in our update, 7 met our eligibility criteria.49–55 Therefore, 11 studies (4 from the original reviews44–47 and 7 from the update search49–55) met the eligibility criteria (Supplementary Figure S2, Appendix 3). We identified 13 companion articles (Supplementary Table S20, Appendix 3).40,48,56–66
Table 2 (abridged; complete table is available as Supplementary Table S8 in Appendix 3) and Supplementary Tables S9–S12 (Appendix 3) summarize study characteristics and key findings (Appendix 5, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.180311/-/DC1, presents full narrative summaries). Most of the studies enrolled participants who were 18 years of age or older and had a low or moderate risk of bias (Supplementary Table S13, Appendix 3).
Characteristics of included studies
We use the terms “male/man,” “female/woman” and “heterosexual” according to the language used by the authors as sex, gender and sexual orientation were not otherwise described. We refer to male serodiscordant couples as “gay, bisexual and other men who have sex with men (MSM)” and to female serodiscordant partners as “lesbian, bisexual and other women who have sex with women (WSW).” No reviews or studies reported on barrier methods other than male condoms, specific sex acts other than penile–vaginal or penile–anal, or HIV transmission risk for WSW. We applied indirect evidence from heterosexual sex partners to WSW for Q1 and Q2, which did not involve male condom use.
Table 3 lists the risk of HIV transmission for Q1 to Q5. Supplementary Figures S3–S5 (Appendix 3) provide forest plots, and Supplementary Tables S14–S19 (Appendix 3) show the GRADE assessments.
Risk of HIV transmission among HIV-serodiscordant sex partners when antiretroviral therapy (with or without viral load suppression), condoms or both were used
Q1: The HIV-positive sex partner is taking antiretroviral therapy (with varying levels of viral load)
The included studies reported 23 phylogenetically linked HIV transmissions across 10 511 person-years of follow-up on antiretroviral therapy (pooled incidence 0.22 transmissions/100 person-years, 95% CI 0.14–0.33).44–47,49–55 Among heterosexual sex partners, there were 23 linked transmissions over 9922 person-years (pooled incidence 0.23 transmissions/100 person-years, 95% CI 0.15–0.35).44–47,49,50,52–55 We rated the quality of the evidence as high for heterosexual sex partners and as moderate for WSW because of the application of indirect evidence from heterosexual sex partners. Two studies involving MSM found 0 transmissions over 588.96 person-years (pooled incidence 0.00 transmissions/100 person-years, 95% CI 0.00–0.63).51,55 We rated the quality of evidence for MSM as moderate. Based on the criteria from the Canadian AIDS Society, we assigned a low risk of sexual HIV transmission for Q1, because of the potential for transmission during the exchange of bodily fluids, and the small number of reported transmissions in the included studies.
Most of the person-years came from cohorts with high levels of adherence to antiretroviral therapy or viral load suppression (Table 2). Overall condom use varied across studies (Table 2) and was not usually characterized for individual participants. Appendix 6 (available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.180311/-/DC1) presents a narrative description of the 23 linked transmission events. Across all studies (10 511 person-years) where the HIV-positive partner was taking antiretroviral therapy, no linked transmissions were characterized by documented consecutive (> 1) viral load measurements of less than 200 copies/mL immediately before or around the estimated time of transmission (Supplementary Table S9, Appendix 3; Appendix 6). Only 2 studies quantified person-years of follow-up with a viral load of less than 200 copies/mL.
Q2: The HIV-positive sex partner is taking antiretroviral therapy and has a suppressed viral load
Two studies reported transmission risk when an HIV-positive partner was taking antiretroviral therapy with a suppressed viral load of less than 200 copies/mL with measurements taken every 4 to 6 months66 or at least twice per year.51 During follow-up, studies of sex acts without the use of condoms reported 0 transmissions over 1327 person-years (pooled incidence 0.00 transmissions/100 person-years, 95% CI 0.00–0.28).51,55 Based on high-quality evidence from a single study that included heterosexual sex partners, 0 transmissions occurred over 799 person-years (0.00 transmissions/100 person-years, 95% CI 0.00–0.46).55 We used this estimate as indirect evidence for WSW and rated it as moderate quality. Based on high-quality evidence from both studies that included MSM, we found 0 transmissions over 527.59 person-years (pooled incidence 0.00 transmissions/100 person-years, 95% CI 0.00–0.70).51,55 We assigned a negligible risk of sexual HIV transmission for Q2, when an HIV- positive partner is taking antiretroviral therapy and has a suppressed viral load of less than 200 copies/mL on consecutive measurements 4 to 6 months apart, because, despite the potential for transmission during the exchange of bodily fluids, there were no reported transmissions in the included studies under these circumstances. We selected a testing interval of 4 to 6 months given that greater than 90% of the person-years occurred under these conditions.
The same 2 studies51,55 reported HIV transmissions per person-years of specific sex acts without condoms, although the follow-up periods for these acts may not have been mutually exclusive (Supplementary Tables S10, S11 and S14–S19, Appendix 3). In the absence of direct evidence for oral sex, we applied estimates for unspecified heterosexual sex.
Q3: The HIV-positive sex partner is taking antiretroviral therapy (with varying levels of viral load) and either partner uses condoms
No studies reported empirical estimates for Q3; however, a systematic review published in 20148 modelled the combined effect of antiretroviral therapy and condoms to derive per-act risks, ranging from 0.003 transmissions per 1000 acts (95% CI 0.00–0.03) for insertive vaginal sex to 0.11 transmissions per 1000 acts (95% CI 0.02–0.73) for receptive anal sex. Furthermore, 4 transmissions were found in 1 study when the HIV-positive partner was taking antiretroviral therapy and reported consistent condom use,52 although there was inadequate information to calculate an estimate of transmission risk. We assigned a low risk of sexual HIV transmission for penile–vaginal sex and penile–anal sex for Q3 because of the potential for transmission during the exchange of bodily fluids that may occur during a slip, break or other incorrect use of the condom when viral load is not suppressed and the few reports of transmission under these circumstances. Given the paucity of evidence, we did not estimate transmission risk for oral sex when antiretroviral therapy (with variable viral load) and condoms were used together.
Q4: The HIV-positive sex partner is taking antiretroviral therapy and has a suppressed viral load, and either partner uses condoms
No reviews or studies provided direct evidence for Q4. We used indirect evidence from studies that reported on antiretroviral therapy, viral load suppression and sex without the use of condoms to provide a risk estimate (pooled incidence 0.00 transmissions/100 person-years, 95% CI 0.00–0.28).51,55 We rated the quality of the indirect evidence as moderate for heterosexual sex partners and MSM. We assigned a negligible risk of sexual HIV transmission for Q4, when an HIV-positive partner is taking antiretroviral therapy, has a suppressed viral load of less than 200 copies/mL on consecutive measurements 4 to 6 months apart and uses condoms, per the rationale for Q2.
Q5: Either partner uses condoms alone
Five reviews provided pooled estimates of the risk of HIV transmission7,8,32,33,35 when heterosexual sex partners used condoms (Supplementary Tables S4–S6, Appendix 3). Because the findings of a Cochrane systematic review7 were less likely to have been confounded by antiretroviral therapy use and were declared as stable conclusive evidence in 2012 by The Cochrane Collaboration,23,24 we did not update this review. The authors of the review7 found that among serodiscordant couples who reported “always” using condoms, there were 1.14 HIV transmissions per 100 person-years (95% CI 0.56–2.04). We assigned a low risk of sexual HIV transmission for Q5 because there is potential for transmission during the exchange of bodily fluids that may occur during a slip, break or other incorrect condom use, and transmissions have occurred among partners reporting consistent condom use.
Interpretation
We found high-quality evidence showing a negligible risk of sexual HIV transmission when an HIV-positive sex partner had a suppressed viral load of less than 200 copies/mL that was maintained through adherence to antiretroviral therapy and confirmed on consecutive measurements every 4 to 6 months. In the GRADE approach, high-quality evidence means we are very confident that the true transmission risk is not higher than the upper limit of the reported confidence interval. We found that the risk of sexual HIV transmission is low when antiretroviral therapy (with varying levels of viral load), condoms or both were used. Based on our findings, relevant case law and other factors, the Department of Justice Canada concluded that the criminal law should not apply to people living with HIV who maintain a suppressed viral load of less than 200 copies/mL.5 The department also concluded that the criminal law should generally not apply to “persons living with HIV who: are on treatment; are not on treatment but use condoms; or, engage only in oral sex (unless other risk factors are present and the person living with HIV is aware of those risks).”5
The risk of HIV transmission with varying levels of viral load (Q1) may have been underestimated because most of our analyses were based on cohorts with high levels of adherence to antiretroviral therapy or viral load suppression. There may also have been confounding caused by condom use. For Q2 and Q4, we could not statistically rule out a risk of approximately 27 transmissions in 1000 serodiscordant couples who were followed for 10 years (based on the upper limit of the confidence interval for the estimate);68 however, a risk this high is unlikely given there were no confirmed transmissions while the viral load was less than 200 copies/mL on consecutive measurements in any study. Where there were 0 transmission events, the upper limits of the 95% CIs were solely a function of the studies’ person-years of follow-up.46,47,49,51,55 For Q3, the transmission risk a priori should be lower than with either antiretroviral therapy or condom use alone, although it does not meet the criteria from the Canadian AIDS Society for negligible risk. Some practices within a particular Canadian AIDS Society risk category may pose a lower risk than others (e.g., Q3 v. Q5).69 For Q5, risk of HIV transmission with condom use may have been overestimated (i.e., condom effectiveness is underestimated) because consistent use may have been overreported owing to recall or social desirability bias, confounding by indication (e.g., individuals engaged in higher risk activities more likely to use condoms) and the possible inclusion of nonphylogenetically linked cases in estimates.7
Our review had a number of strengths including using an equity-based approach in considering all genders, sexual orientations and relationship types in our searches, and developing conclusions for seldom-studied groups including WSW. Our observed transmission rate for Q1 was consistent with that found in a 2013 systematic review6 (0.14 transmissions/100 person-years, 95% CI 0.04–0.31; Supplementary Table S4, Appendix 3). Unlike previous reviews6,21 that relied on modelling and assumptions about eligible person-years of follow-up to develop estimates for Q2, our review relied on empirical data. We also included a rating of certainty in our risk estimates,28 and assigned levels of risk using a priori objective criteria;31 both of which may aid the public, as well as the medical and legal communities, in understanding the risks of transmission.
Limitations
Our study protocols (Appendices 1 and 2), although developed a priori and peer reviewed, were not registered or published. Our ability to conduct certain analyses (e.g., per-act risk estimates, risk with concomitant sexually transmitted infections or for specific sex acts) was limited by both the level of detail and inconsistent measures across the included studies. Had the data allowed calculation of per-act estimates, however, our conclusions would not have changed. Updates of 2 studies that were included in our review were recently released.51,55,70,71 If all of the additional follow-up from these studies were eligible for incorporation into our meta-analysis, the upper confidence interval of our estimate for Q2 and Q4 would be lowered (from 0.28 to 0.13), but our point estimate and conclusions would not change. A recent expert consensus has, in any case, cautioned against overreliance on theoretical risks in this context.72 Finally, the risk of HIV transmission in study populations may not reflect the risks and circumstances in individual relationships.
Conclusion
Our findings show that there is a negligible risk of sexually transmitting HIV when an HIV-positive sex partner adheres to antiretroviral therapy and maintains a suppressed viral load of less than 200 copies/mL on consecutive measurements every 4 to 6 months. The risk of sexual HIV transmission is low when an HIV-positive sex partner is taking antiretroviral therapy without a suppressed viral load of less than 200 copies/mL, condoms are used or both. These findings will support individual patient and clinician decision-making, and will have implications for public health case management and contact tracing. The Department of Justice Canada used these findings to inform their 2017 report on the justice system’s response to HIV nondisclosure,5 and they may inform the responses of other justice systems.
Acknowledgements
The authors thank the peer reviewers nominated by the Communicable and Infectious Disease Steering Committee (CIDSC) of the Pan-Canadian Public Health Network whose thoughtful comments and feedback on earlier versions of this evidence synthesis strengthened their work: Ian Davis, Riyas Fadel, Évelyne Fleury, Todd Hatchette, Laurie Ireland, Claude Laberge, Cynthia Lyles, David McKeown, Darrell Tan and Denise Werker. The authors also thank members of the CIDSC and the Council of Chief Medical Officers of Health of the Pan-Canadian Public Health Network for their reviews and comments. The authors acknowledge the advice provided by Dr. James Hanley with respect to our statistical analysis and interpretation. The authors also thank Jane Falconer for her external peer review of the search strategy for the update. We also acknowledge the unique contributions of our colleagues at the Public Health Agency of Canada, including Lisa Glandon, Lynda Gamble and Robyn Hocking at the Federal Science Library for preparing and conducting our electronic database searches; Fowsia Abdulkadir for assisting with reference screening; and Kanchana Amaratunga, Saskia Arnold, Marion Doull, Marsha Hay-Snyder, Howard Njoo, Toju Ogunremi, Dana Paquette, Shamila Shanmugasegaram, Winnie Siu and Karen Timmerman for their thoughtful input and insight.
Footnotes
See related article at www.cmaj.ca/lookup/doi/10.1503/cmaj.181405
Competing interests: None declared.
This article has been peer reviewed.
Contributors: Rachel Rodin conceptualized and oversaw this project in response to the Department of Justice Canada’s request to the Public Health Agency of Canada for an assessment of the most recent medical science on sexual HIV transmission. Rachel Rodin and Jennifer LeMessurier designed the project with input from Gregory Traversy, Olivia Varsaneux, Makenzie Weekes, Marc Avey and Oscar Niragira. Jennifer LeMessurier, Gregory Traversy, Olivia Varsaneux, Makenzie Weekes and Oscar Niragira screened references and extracted data. Gregory Traversy, Olivia Varsaneux, Makenzie Weekes and Oscar Niragira conducted quality (AMSTAR) and risk-of-bias (QUIPS) assessments. Jennifer LeMessurier, Gregory Traversy, Olivia Varsaneux and Makenzie Weekes assessed the quality of evidence across studies (GRADE) with methodological assistance from Gordon Guyatt. Jennifer LeMessurier, Gregory Traversy, Olivia Varsaneux, Makenzie Weekes, Marc Avey, Robert Gervais and Rachel Rodin analyzed and interpreted data. Jennifer LeMessurier, Gregory Traversy, Olivia Varsaneux, Makenzie Weekes, Marc Avey and Rachel Rodin drafted the manuscript. All of the authors critically revised the manuscript for important intellectual content, gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Funding: This research was funded, conducted and approved by the Public Health Agency of Canada.
Data sharing: Data extracted from the included studies are presented largely in the Results and Appendices; however, full extraction data tables are available upon reasonable request from the corresponding author.
- Accepted September 20, 2018.