Abstract
Objective: To systematically review the benefits and risks associated with the use of benzodiazepines to treat insomnia in adults.
Data sources: MEDLINE and the Cochrane Controlled Trials Registry were searched for English-language articles published from 1966 to December 1998 that described randomized controlled trials of benzodiazepines for the treatment of insomnia. Key words included "benzodiazepines" (exploded), "randomized controlled trial" and "insomnia." Bibliographies of relevant articles were reviewed for additional studies and manufacturers of benzodiazepines were asked to submit additional randomized controlled trial reports not in the literature.
Study selection: Articles were considered for the meta-analysis if they were randomized controlled trials involving patients with insomnia and compared a benzodiazepine with placebo or another active agent. Of the 89 trials originally identified, 45 met our criteria, representing a total of 2672 patients.
Data extraction: Data were extracted regarding the participants, the setting, details of the intervention, the outcomes (including adverse effects) and the methodologic quality of the studies.
Data synthesis: The meta-analyses of sleep records indicated that, when compared with placebo, benzodiazepines decreased sleep latency by 4.2 minutes (non-significant; 95% confidence interval [CI] −0.7 to 9.2) and significantly increased total sleep duration by 61.8 minutes (95% CI 37.4 to 86.2). Patient-reported outcomes were more optimistic for sleep latency; those randomized to benzodiazepine treatment estimated a sleep latency decrease of 14.3 minutes (95% CI 10.6 to 18.0). Although more patients receiving benzodiazepine treatment reported adverse effects, especially daytime drowsiness and dizziness or light-headedness (common odds ratio 1.8, 95% CI 1.4 to 2.4), dropout rates for the benzodiazepine and placebo groups were similar. Cognitive function decline including memory impairment was reported in several of the studies. Zopiclone was not found to be superior to benzodiazepines on any of the outcome measures examined.
Interpretation: The use of benzodiazepines in the treatment of insomnia is associated with an increase in sleep duration, but this is countered by a number of adverse effects. Additional studies evaluating the efficacy of nonpharmacological interventions would be valuable.
Insomnia is a common reason for visiting a primary care physician.1-3 Although treatment of the underlying causes of insomnia and nonpharmacological therapies are recommended, benzodiazepines remain a treatment of choice.3-9 The 5 benzodiazepines promoted as hypnotics in Canada accounted for approximately $40 million in medication expenditures in 1993 (Dorothy Rhodes, IMS Canada, Mississauga, Ont.: personal communication, 1996). Various studies have raised concerns about prolonged use, as well as higher rates of use among women and older people and in certain regions of the country.1,4,6,9 The association of benzodiazepine use with confusion, falls and motor vehicle accidents and the uncertainty regarding their benefit beyond that of placebo make benzodiazepines a logical topic for review.8,10-15
Unlike many other target endpoints, sleep can be measured objectively. Polysomnography (analysis of sleep) involves the documentation of sleep onset, sleep duration and the number of awakenings during the night with EEG recordings.16 However, these objective measures may not capture subjective experience regarding sleep quality, and there is currently no universally accepted measure of sleep quality (e.g., a validated sleep-specific quality-of-life questionnaire).
This systematic overview of studies on benzodiazepine use in the treatment of insomnia was prepared to provide a background paper for the practice guidelines initiative sponsored by the Canadian Medical Association and the Canadian Pharmaceutical Association. It is the second in a 3-part series of meta-analyses of benzodiazepine use; benzodiazepine use in the treatment of alcohol withdrawal was addressed in a previous issue of CMAJ,17,18 and an evaluation of its use in the treatment of anxiety will appear in an upcoming issue. Our objective here was to obtain precise summary estimates of the efficacy and common adverse effects of benzodiazepines compared with those of placebo and other treatments.
Methods
A comprehensive search of MEDLINE was conducted for articles of randomized controlled trials published from 1966 to December 1998 on the use of benzodiazepines in the treatment of insomnia. The MeSH search terms used were "benzodiazepine" (exploded) or "benzodiazepine tranquillizers" (exploded) or "clonazepam"; "drug therapy"; "randomized controlled trial" or "random allocation" or "all random"; "human" and "English language." A similar search was carried out in the Cochrane Controlled Trials Registry. Relevant articles were then retrieved and appraised for original data comparing therapies for insomnia. Bibliographies of retrieved articles were scanned for additional articles, and each manufacturer of a brand-name benzodiazepine was asked to contribute reports of early trials not published in the literature. Reports of randomized controlled trials of benzodiazepine therapy for primary insomnia were considered for the meta-analysis if they compared a benzodiazepine with a placebo or an alternative active drug.
Individual reports were rated for quality with the use of a scale from 0 to 5; for therapeutic efficacy this meant taking into account the quality of randomization, blinding and follow-up, and for harmful effects it meant examining randomization, blinding and control for baseline differences between groups.19
Descriptive data were recorded on the study design, conditions treated, patient characteristics, setting and duration of the trial and outcomes measured. Interrater reliability was checked through duplicate, independent abstraction of the first 21 articles. Overall agreement on classification and descriptive data extracted from the studies was 98% (κ value 0.95). Agreement that all validity criteria were met for a study of therapy was 95% (κ value 0.90) and for a study of harmful effects was 76% (κ value 0.51). Disagreement was resolved by consensus, and subsequent abstraction was carried out by one reviewer.
The meta-analysis of the endpoints from the selected studies was necessarily limited to those presented in a comparable way. Fixed-effects methods were used, and heterogeneous results were checked with a random-effects model.20 Mantel-Haenszel common odds ratios, along with 95% confidence intervals (CIs, calculated by the method of Cornfield) were obtained for discrete data (e.g., number of patients with an outcome).21 The Breslow-Day test for homogeneity was applied, and if study results were heterogeneous, the studies were subdivided into predefined groups and the common odds ratios were recalculated. The subdivisions examined included the type of benzodiazepine, dosage level (e.g., high versus low), setting (e.g., primary care versus tertiary care) and quality of the methodology. For continuous variables (e.g., minutes of sleep), effect sizes were calculated for each study as the difference between the outcome means of the groups divided by the pooled standard deviations. An overall weighted effect size was obtained and converted into natural units for the overall difference (with the 95% CI) in outcome between the benzodiazepine groups and the control groups.21 Results were tested for homogeneity using the Breslow-Day test. If a measure of variability was not reported for study results, standard deviations were calculated by means of substitution in the formula for the coefficient of variation using the study results most similar in outcome means and sample size to the study with missing data.22 In studies with a crossover design, the number of patients was counted once for each arm in which they were included.
Results
Of the 89 randomized controlled trials we identified, 44 were excluded from the meta-analysis: in 24 a benzodiazepine was compared only to another benzodiazepine,23-46 in 1 report original data was not included,47 in 8 closer review revealed the study was not a true randomized controlled trial,48-55 in 7 insomnia was related to another disorder56-62 and in 4 the alternative therapy was not available in Canada.63-66 The remaining 45 randomized controlled trials67-111 represented a total of 2672 patients, 47% of whom were women. Twenty-five studies were based in the community and 9 involved inpatients. Twenty-seven studies compared a benzodiazepine with a placebo, 13 compared a benzodiazepine with an alternate active treatment, and 5 studies involved a combination of the above.
The mean age of patients (reported in 33 of 45 studies) ranged from 29 to 82 years; 15 studies included patients over 65 years of age. The duration of the studies ranged from 1 day to 6 weeks, with a mean of 12.2 days and median of 7.5 days.
Sixteen studies involved triazolam, 14 used flurazepam, 13 involved temazepam, 5 used midazolam, 4 reported on nitrazepam and 2 involved estazolam; lorazepam, diazepam, brotizolam, quazepam, loprazolam and flunitrazepam were each evaluated in 1 study. Alternative drug therapies included zopiclone in 13 studies and diphenhydramine, glutethimide and promethazine in 1 study each. Only 1 article reported on a nonpharmacological treatment (behavioural therapy).100
Exclusion criteria for patients in individual studies were diverse. Patients were excluded if they were undergoing or had recently undergone treatment for insomnia (in 24 studies), if they had ever been diagnosed with a psychiatric disorder (in 25 studies) or had a "serious medical problem," usually underfined (in 24 studies), or a history of drug or alcohol abuse (in 21); 16 studies excluded women who were pregnant or lactating.
The methodologic quality of the studies was not uniform. For the 41 studies reporting on benefit, only 26 (63.4%) met the rudimentary criteria of good follow-up and double blinding. Similarly, for the 45 studies reporting on harm, 25 (55.6%) met all of our criteria (random allocation of patients, double blind and baseline differences controlled). The diversity in outcomes used and the methods of summarizing (e.g., difference in time to sleep versus proportion of patients falling asleep in less than 30 minutes) prevented the pooling of many trials. Data on sleep outcomes documented by sleep records (objective) were kept separate from patient-reported outcomes (subjective).
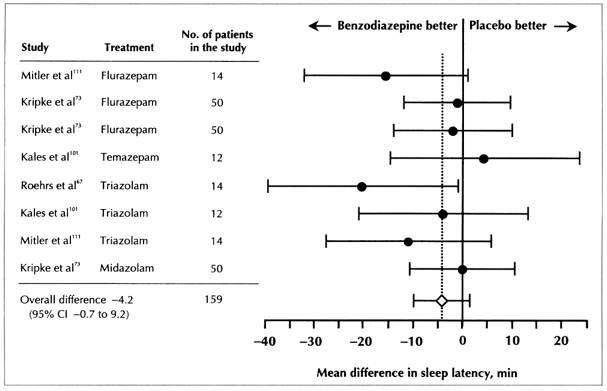
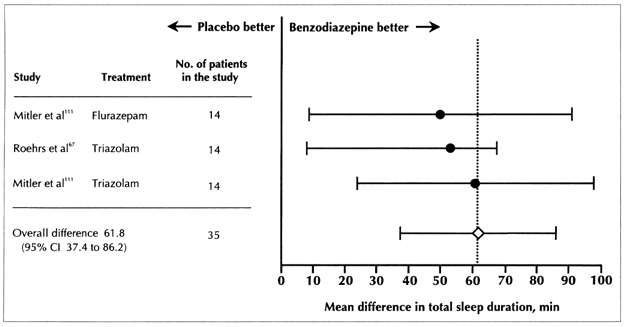
Eight comparisons between a benzodiazepine and placebo in 4 studies67,73,101,111 involving 159 subjects were made on sleep-record latency (time to fall asleep) data (Fig. 1). The pooled difference indicated that the latency to sleep for patients receiving a benzodiazepine was 4.2 minutes (95% CI −0.7 to 9.2) shorter than for those receiving placebo. Results of the meta-analysis of 2 studies in which total sleep duration (using sleep records) was compared (n = 35 patients)67,111 indicated that patients in the benzodiazepine groups slept for an average of 61.8 minutes (95% CI 37.4 to 86.2) longer than those in the placebo groups (Fig. 2).
Fig. 1: Mean difference in sleep latency from sleep records of trials analysing the effect of benzodiazepine versus placebo for the treatment of insomnia; 1-7 day treatment (test for homogeneity, p > 0.05).
Fig. 2: Mean difference in total sleep duration from sleep records of trials analysing the effect of benzodiazepine versus placebo for the treatment of insomnia; 1-7 day treatment (test for homogeneity, p > 0.05).
Patients' estimates of sleep latency were examined in 8 studies (n = 539),73,75,80,90,92,103,107,111 and the summary estimate of the superiority of benzodiazepines over placebo was 14.3 minutes (95% CI 10.6 to 18.0). A priori hypotheses regarding differences between studies were tested because a statistical test for heterogeneity reached significance. When the randomized controlled trials that received a high-quality rating were separated from those with lower ratings, the heterogeneity disappeared, and the estimate of benefit was smaller but still statistically significantly. The patient-estimated sleep latency from pooled data from the higher quality studies was 11.7 minutes (95% CI 7.6 to 15.8) compared with 23.7 minutes (95% CI 15.8 to 31.5) from pooled data of the lower quality studies. Patients' estimates of sleep duration from 8 studies (n = 566)73,75,80,90,93,103,107,111 were pooled, and total sleep duration was calculated to be 48.4 minutes (95% CI 39.6 to 57.1) longer for patients taking benzodiazepines than for those on placebo.
Unfortunately, we could not answer one of our key research questions - whether tolerance to any sleep-promoting effect of benzodiazepines occurs - because all of the trials eligible for the meta-analyses were of short duration (i.e., 14 days or less).
Seven studies (n = 821)25,81,87,103,107,109,110 provided data on the proportion of subjects reporting adverse effects. As shown in Fig. 3, patients randomized to a benzodiazepine group were more likely to report adverse effects (odds ratio [OR] 1.8, 95% CI 1.4 to 2.4) over the 3-7 days of therapy. Data were then pooled on 2 specific adverse outcomes - daytime drowsiness and dizziness or lightheadedness. In a meta-analysis of 8 studies involving 889 patients, benzodiazepines were more likely than placebo to be associated with complaints of daytime drowsiness (OR 2.4, 95% CI 1.8 to 3.4).71,73,79,81,87,94,103,109 Likewise, based on results of 4 studies involving 326 patients,79,81,103,110 a benzodiazepine was more likely to be associated with dizziness or lightheadedness (OR 2.6, 95% CI 0.7 to 10.3), although this effect did not reach statistical significance. Although more adverse effects were experienced by patients taking a benzodiazepine for the treatment of insomnia, dropout rates in the benzodiazepine and placebo groups were similar.79,84,93
Fig. 3: Odd ratio of trials analysing effect of benzodiazepine versus placebo in terms of total number of adverse events; 3-7 day treatment (test for homogeneity, p > 0.05).
The relationship between benzodiazepine dose and outcome could only be evaluated for patient-reported outcomes; there was no strong correlation for sleep latency data (r = 0.4, 95% CI −0.3 to 0.9) or for sleep duration (r = 0.2, 95% CI −0.8 to 0.4).
There were 6 studies that rated high on our quality rating25,70,76,79,83,109 that could not be combined in the meta-analyses; these were examined separately (see Table 1 at www.cma.ca/cmaj/vol-162/issue2/225tab1.htm). The sleep outcome results from these studies mirrored the pooled results obtained in the meta-analyses, with decreased sleep latency and increased sleep duration for the benzodiazepine groups; 3 of the 4 studies examining sleep quality reported a significant improvement in sleep quality with benzodiazepine therapy, but no assessment of the clinical importance of these results was provided.
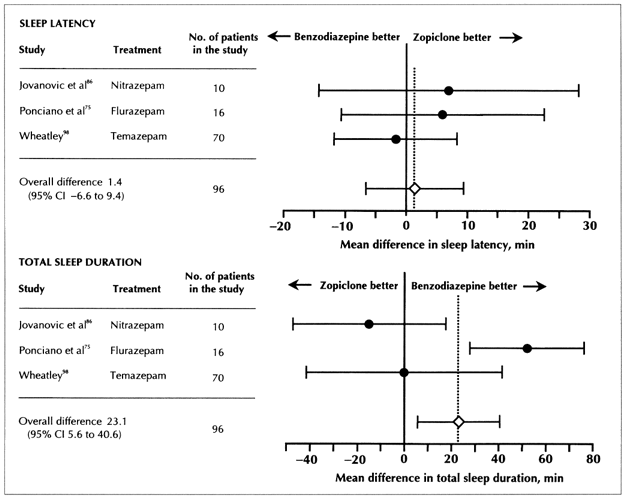
Since 1990 zopiclone (Imovane) has been marketed in Canada as a short-acting hypnotic, advertised as safer than benzodiazepines and less disruptive of sleep architecture.112 However, proof of its clinical superiority, especially its long-term safety, is lacking. Few trials were amenable to a meta-analysis for comparison purposes. The pooling of the results of 3 trials (n = 96) indicated there was no significant difference between benzodiazepine and zopiclone treatment in terms of effects on sleep latency, but benzodiazepine therapy might lead to a longer sleep (23.1 min, 95% CI 5.6 to 40.6)75,86,98 (Fig. 4). Six other studies70,76-79,96 that could not be included in the meta-analysis reported no significant differences for any sleep parameter (Table 2 available at www.cma.ca/cmaj/vol-162/issue-2/225tab2.htm).
Fig. 4: Mean difference in sleep latency (above) and total sleep duration (below) of trials analysing the effect of benzodiazepine versus zoplicone for the treatment of insomnia; 3-7 day treatment (sleep latency studies test for homogeneity, p > 0.05; sleep duration studies test for homogeneity, p < 0.01).
The data from 4 trials (n = 252)77,78,91,95 comparing benzodiazepines and zopiclone were combined to calculate a summary odds ratio for adverse effects.79,91,96,98 There was a nonsignificant trend toward more side effects with the use of benzodiazepines (OR 1.5, 95% CI 0.8 to 2.9) but also a trend toward a lower dropout rate for those in the benzodiazepine groups.
There were only a few studies comparing the efficacy of benzodiazepines in the treatment of insomnia with other alternatives. Comparisons with antihistamines, including diphenhydramine83 and promethazine,85 did not detect any significant differences on sleep outcomes. Results of the single small trial that compared triazolam with behavioural therapy100 supported a priori hypotheses; although triazolam was more effective than behavioural therapy early in treatment to decrease sleep latency, its efficacy declined by the second week of treatment. Behavioural therapy, however, remained effective throughout the 9-week follow-up.
The global term "cognitive impairment" is often used to encompass negative effects on memory, reaction time and thought processing speed - all concepts that are thought to be causally related to drowsiness, confusion and accidents. Most of the relevant trials evaluating cognitive impairment and the use of benzodiazepines could not be combined, but each was reviewed separately. Each of the studies evaluating memory impairment involved a small sample and was of short duration69,75,80 2 of the 3 trials we assessed reported significant memory impairment associated with benzodiazepine use (see Table 3 at www.cma.ca/cmaj/vol-162/issue-2/225tab3.htm).
Studies examining other cognitive or psychomotor adverse effects,70-72,75,80,97,106,111 primarily in middle-aged adults (see Table 4 at www.cma.ca/cmaj/vol-162/issue-2/225tab4.htm), noted significant impairment particularly with flurazepam. The studies on temazepam, triazolam and zopiclone are conflicting and plagued by small samples.
Four small trials evaluating temazepam, flurazepam, triazolam and nitrazepam involved elderly patients exclusively83,93,96,104 (see Table 5 at www.cma.ca/cmaj/vol-162/issue-2/225tab5.htm). Only 1 trial followed subjects beyond 2 weeks, and again, results were mixed regarding benefit on sleep outcomes; adverse cognitive effects were poorly reported.
Interpretation
Uncertainty regarding the risk:benefit ratio for the use of benzodiazepines in the treatment of insomnia has led to controversy over their appropriate level of use. A previously published meta-analysis113 suggests that these medications are beneficial for those with insomnia, but their analysis combined benzodiazepines and alternative agents in the same group, limited the sample to younger patients, did not examine risk and did not present results in clinically interpretable units. Although our meta-analyses does address these deficiencies regarding benefit and risk, it but does not entirely resolve the debate.
As our analysis of sleep records has shown, benzodiazepines are associated with an insignificant decrease in sleep latency compared with placebo. Their effect on overall sleep duration is more marked (approximately 1 hour) and perhaps clinically meaningful. In agreement with findings of a recent survey,5 patients taking benzodiazepines tended to overestimate sleep outcome measures and the efficacy of their medication. The association of higher quality trials with more conservative results is an important finding but not a new one. An analysis of the Oxford Perinatal Trials Database also showed that more rigorous methods of randomization and blinding were associated with differences that were not as impressive.114,115 Current evidence does not appear to support a dose-response gradient for benefit with benzodiazepines; the data are scant, however. Restricting the analyses to benzodiazepines available in Canada did not affect the results of the meta-analyses.
Benzodiazepines were associated with more reports of adverse effects including drowsiness, dizziness or lightheadedness and cognitive impairment, but this did not translate into higher discontinuation rates. None of these adverse effects were unexpected, given the pharmacology of benzodiazepines. The maintenance of subjects within a trial despite adverse effects could be associated with a compliance effect, and it would be unlikely that it would generalize to real practice, or it could reflect a real preference of subjects to remain on the drug despite adverse effects. This preference may relate to benefits perceived but not measured or be a consequence of dependence. Rebound insomnia associated with the abrupt withdrawal of benzodiazepine treatment is another factor likely to promote continuance of the drug.116,117 These adverse effects are potential surrogates for the serious morbidity associated with benzodiazepine use that has been detected in nonrandomized trials.15
The apparent conflicting information on cognitive impairment and the lack of an accepted validated scale for measuring global cognitive impairment suggest a need for further research in this area. Because elderly volunteers show greater cognitive impairment and sedative effects after benzodiazepine administration,118 elderly patients with comorbid conditions should be a research priority. Further research on the treatment of patients with insomnia should address:
• the impact of insomnia on patients' everyday lives
• factors that contribute to a patient's decision to seek medical attention for their insomnia
• factors that determine dissatisfaction with sleep hygiene education alone
• the degree to which sociologic factors such as loneliness, work dissatisfaction and family stress contribute to a request for benzodiazepine medication
• the criteria and circumstances that influence a physician's decision to prescribe a benzodiazepine for sleep
• patients' and physicians' perceptions regarding the overall risk:benefit ratio of benzodiazepines for the treatment of insomnia
Zopiclone was the only alternative pharmacological therapy that could be studied with any precision. To date, there is not enough data available to conclude that one should be chosen over the other for the treatment of insomnia. However, because zopiclone is less well known and more expensive than most benzodiazepines, it cannot be recommended as a substitute for benzodiazepines. A recent study noted that both users of benzodiazepines and zopiclone were at increased risk of experiencing a road traffic accident.119
The lack of data comparing benzodiazepine treatment to nonpharmacological alternatives is disappointing. Ample evidence exists to suggest that psychological interventions including stimulus control, sleep restriction and relaxation techniques are efficacious over a number of months.120,121 A small randomized controlled trial published after our overview was complete compared behavioural and pharmacological (temazepam) therapies for late-life insomnia. Cognitive-behaviour therapy was rated by patients and clinicians as more effective, and the beneficial effects were sustained for longer.122
A potential limitation to our methodology was the restriction of our search to studies published in English; we may have missed some relevant studies. Although the pooling of available data was limited by the wide variety of outcome measures reported, the variables that we pooled were highly relevant to our analyses. Also, we selected only randomized controlled trials, over 90% of which were also double-blinded, but the adequacy of blinding in placebo-controlled insomnia trials has been questioned.123 Our results may appear to be at variance with perceptions of benefit in usual clinical practice; this variance may be due to a large placebo effect in insomnia therapies.117 Finally, although the long-term use of benzodiazepines is relatively common among elderly patients,12 we cannot comment on the benefit and safety of long-term use because the trials analysed were of short duration.
Conclusions
It is unfortunate that 2 major clinical questions remain unanswered. First, how do benzodiazepines impact on the quality of patients' sleep, overall quality of life and functional status compared with placebo or health-promoting interventions such as exercise? Secondly, how do the benefits of benzodiazepines for the treatment of insomnia counterbalance with the associated risks? In other words, can we adequately weigh any extra minutes of sleep gained against the possibility of cognitive impairment or dependence? It is clear that benzodiazepines do not provide a major advantage over placebo and that they are are not free of adverse effects. In light of the strength of the placebo effect in patients with insomnia, physicians and patients who believe that benzodiazepines are highly effective may wish to reconsider their treatment choice. None of the data extracted in this review support long-term use (i.e., longer than 2 weeks).
Regrettably, there does not appear to be a clearly efficacious and safe pharmaceutical alternative to benzodiazepines.
Nonpharmacological alternatives currently hold the most promise for the treatment of insomnia, but the techniques are not well understood by generalist clinicians and their efficacy has not been adequately studied. Additional reports comparing the efficacy of educational, health promotional or psychological interventions with benzodiazepines would be valuable.
This study was funded in part by the Canadian Pharmaceutical Association (CPhA) and the Canadian Medical Association (CMA). The overview was prepared for a CPhA-CMA clinical practice guideline initiative on benzodiazepines. Dr. Holbrook is a recipient of an Ontario Ministry of Health Research Personnel Award (no. 04698).
Competing interests: None declared.
Footnotes
-
This article has been peer reviewed.
An overview of the diagnosis and management of insomnia in clinical practice appears on page 216.
Correspondence to: Dr. Anne M. Holbrook, Centre for Evaluation of Medicines; St. Joseph's Hospital and McMaster University, 50 Charlton Ave. E, Hamilton ON L8N 4A6; fax 905 521-6136; holbrook{at}mcmaster.ca Reprints will not be available.
References
- 1.↵
- 2.
- 3.↵
- 4.
- 5.↵
- 6.
- 7.
- 8.↵
- 9.
- 10.
- 11.
- 12.↵
- 13.
- 14.
- 15.↵
- 16.↵
- 17.↵
- 18.
- 19.↵
- 20.↵
- 21.↵
- 22.↵
- 23.↵
- 24.
- 25.↵
- 26.
- 27.
- 28.
- 29.
- 30.
- 31.
- 32.
- 33.
- 34.
- 35.
- 36.
- 37.
- 38.
- 39.
- 40.
- 41.
- 42.
- 43.
- 44.
- 45.
- 46.
- 47.↵
- 48.↵
- 49.
- 50.
- 51.
- 52.
- 53.
- 54.
- 55.
- 56.↵
- 57.
- 58.
- 59.
- 60.
- 61.
- 62.
- 63.↵
- 64.
- 65.
- 66.
- 67.↵
- 68.
- 69.↵
- 70.↵
- 71.↵
- 72.
- 73.↵
- 74.
- 75.↵
- 76.
- 77.↵
- 78.
- 79.↵
- 80.
- 81.
- 82.
- 83.↵
- 84.
- 85.↵
- 86.
- 87.
- 88.
- 89.
- 90.
- 91.
- 92.
- 93.
- 94.
- 95.
- 96.
- 97.
- 98.
- 99.
- 100.↵
- 101.
- 102.
- 103.
- 104.
- 105.
- 106.
- 107.
- 108.
- 109.
- 110.
- 111.
- 112.↵
- 113.↵
- 114.↵
- 115.
- 116.↵
- 117.↵
- 118.↵
- 119.↵
- 120.↵
- 121.
- 122.↵
- 123.↵