Technology: Continuous venovenous hemodiafiltration (CVVHD)
Use: CVVHD is a form of continuous renal replacement therapy that is used for critically ill patients with multisystem organ failure in whom acute renal failure develops. This form of dialysis differs from intermittent hemodialysis in that it is a slower, continuous mode of dialysis that permits the clearance of blood solutes both by diffusion across a semipermeable membrane (dialysis) and by convection of solutes across a membrane as they are separated from whole blood in response to hydrostatic pressure.1 Hydrostatic pressure is generated by simultaneously circulating fluid in conjunction with blood through a dialysis circuit before it enters the dialysis hemofilter.
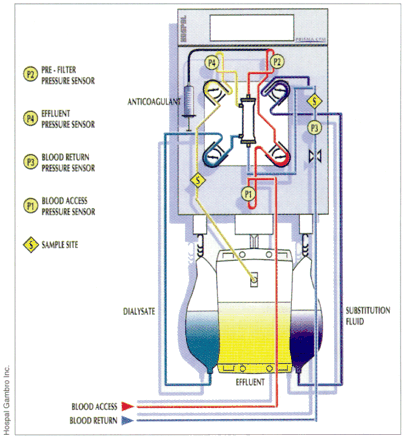
History: Continuous renal replacement therapy was first described by Scribner and associates2 in 1960. It was initially performed through an arteriovenous circuit that incorporated a dialysis hemofilter and relied on the patient's arterial blood pressure to circulate blood through the hemofilter. This technology was inspired by the limitation in the use of intermittent hemodialysis in hemodynamically unstable patients and has been demonstrated to improve hemodynamic and cerebrovascular stability compared with intermittent hemodialysis in patients with acute renal failure who also have liver failure or cerebral edema.[3, 4] Continuous renal replacement therapy has evolved rapidly. With CVVHD, sophisticated pump-driven devices with venovenous access are used to circulate blood through a dialysis hemofilter (Fig. 1), and thus the risks inherent with arterial access are avoided. CVVHD is used to treat acute renal failure complicated by refractory fluid overload or hemodynamic instability, and to treat life-threatening electrolyte and acid-base disorders.5 In Canada, its use has been limited to critically ill patients in tertiary care facilities with renal failure, fluid overload and major electrolyte abnormalities.
Fig. 1: Multi-mode continuous renal replacement machine.
Promise: Sepsis due to bacterial infection and systemic inflammatory response syndrome (SIRS) due to noninfectious causes such as trauma, burns and pancreatitis develop from the release of a multitude of biologically active inflammatory mediators. It has been hypothesized that the removal of these noxious molecules by hemofiltration may blunt the exaggerated inflammatory response and improve clinical outcome. Although the removal of several cytokines such as interleukin-1, interleukin-6, tumour necrosis factor-alpha and myocardial depressant substance has been demonstrated in animal and human studies, the lack of large randomized clinical trials in this area has made the role of continuous renal replacement therapy for sepsis and SIRS controversial.[6, 7] Specific limitations with the use of continuous renal replacement therapy for sepsis and SIRS include the middle-molecular weight of most cytokines (< 30 000 daltons), which require clearance through convection (not diffusion), and the need for more than 100 L of replacement fluid daily to achieve clinically meaningful clearances of some inflammatory mediators.
Problems: Anticoagulation remains a major problem with the use of continuous renal replacement therapy. Systemic heparin has been the predominant mode of anticoagulation; however, a high risk of bleeding has been encountered in subgroups of critically ill patients. Alternative modes such as intravenous prostacycline and trisodium citrate have been used successfully.8 Other drawbacks include the need for expensive dialysis machines and hemofiltration fluid for solute exchange.
Prospects: CVVHD has gained acceptance as an effective mode of renal replacement therapy in critically ill patients with renal failure, as evidenced by the publication of large series of patients given this treatment.9 However, with the emergence of this technology, physicians caring for critically ill patients will have to face the ethical decisions regarding the futility of sustaining certain patients on yet another form of life support.
[Visit www.crrt.com for current references and conference information relating to continuous renal replacement therapy.]
Competing interests: Dr. Kutsogiannis is currently conducting research comparing modes of anticoagulation in CVVHD that has received funding from several sources, including Hospal Gambro Inc., manufacturer of continuous renal replacement therapy equipment.
Acknowledgments
CMAJ publishes papers that advance the understanding of medicine and health care, stimulate debate, educate and entertain. Manuscripts are usually published within 16 to 18 weeks of acceptance. Please refer to "Uniform requirements for manuscripts submitted to biomedical journals" found on our Web site (www.cma.ca/publications/mwc/uniform.htm).
Research articles (2500 words, excluding the abstract, figures, tables and references) report original clinical findings of interest to a general medical audience. Systematic reviews and meta-analyses also fall under this category. Research letters may present interim findings or the results of a small study.
Although most Commentaries are invited, we welcome unsolicited submissions and suggestions for topics and contributors. Commentaries should be restricted to 1000 words and 10 references. They require a succinct and confident style, a clear point of view and a degree of balance.
The Review section includes narrative reviews, program descriptions, case reports and other papers of no more than 2000 words that contribute to the professional development of practising physicians.
Narrative reviews present a practical and highly readable overview of recent advances in basic science or clinical practice. Program descriptions present teaching programs, new clinical procedures, innovative approaches to the management of a disease, pilot projects, and so forth. With rare exceptions, Case reports must describe conditions or events that have not been previously reported, or present a useful teaching point. Signed consent must be obtained from all patients or their surrogates.
The Left Atrium gives readers room for reflection through book and film reviews, creative writing and features on the visual and performing arts. We welcome unsolicited poetry, fiction and creative non-fiction; the writing should be candid, but confidentiality must be respected in accounts of experiences with patients. If you would like to be added to our list of book reviewers or discuss ideas for contributions please contact Anne Marie Todkill, editor of The Left Atrium (todkia{at}cma.ca). FIGURE

Figure.
Public Health presents brief reports on current issues in public health of interest to clinicians. Prospective contributors should contact the editor-in-chief.
Letters to the Editor commenting on published articles or of general interest are welcome. Letters are edited for length (ideally 300 words) and style, and authors whose work is discussed are given an opportunity to respond.
For the unabridged version of Writing for CMAJ see the Jan. 11, 2000, issue (page 113) or visit our Web site (www.cma.ca/cmaj).
Editor-in-chief; John Hoey, MD; 800 663-7336 x2118;hoeyj{at}cma.ca
Associate Editors; Tom Elmslie, MD; Ken Flegel, MD; K.S. Joseph, MD; Anita Palepu, MD; telmslie{at}scohs.on.ca; kflegel{at}rvhmed.lan.mcgill.ca; ks.joseph{at}np.inkgrace.ns.ca; anita{at}hivnet.ubc.ca