The case
Mr. K, a 57-year-old man, presents to the emergency department with sudden onset of chest pain radiating to the left arm of 30 minutes' duration. Ten days earlier he experienced a similar episode of pain that lasted 10 minutes as well as several intermittent episodes of pain over the subsequent week that lasted about 5 to 10 minutes each and were not related to physical activity. His medical history includes peptic ulcer and arterial hypertension. He had an acute ischemic stroke 1 year ago, at which time a stenosis of 80% in the left internal carotid artery was detected and treated by carotid endarterectomy. His risk factors for ischemic heart disease are hypertension, dyslipidemia and cigarette smoking. At presentation, his blood pressure is 190/90 mm Hg and he is given ASA, atorvastatin, enalapril, amlodipine and omeprazole. An electrocardiogram (ECG) shows ST-segment depression in the inferior leads, and the cardiac troponin I level is normal. Intravenous therapy with heparin and nitrates is begun, with a good initial response. However, Mr. K experiences chest pain the day after hospital admission. Holter monitoring shows persistent ST-segment depression in the inferior leads. On day 3 coronary angiography shows critical stenosis of the proximal right coronary artery. Percutaneous transluminal coronary angioplasty with stenting is performed. Mr. K is subsequently discharged pain free with a normal ECG and is prescribed ASA and clopidogrel.
Questions surrounding this case: Should low-molecular-weight (LMW) heparin be used instead of unfractionated heparin? If so, which LMW heparin? What is the optimal antiplatelet regimen? When should clopidogrel be started? How long should it be continued?
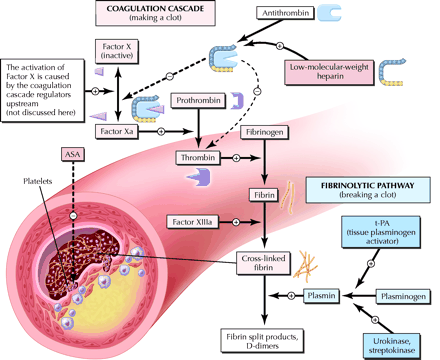
Fissuring of atherosclerotic plaque triggers thrombus formation and is the main pathogenetic mechanism underlying acute coronary syndromes such as unstable angina, non-Q-wave myocardial infarction (MI) and Q-wave MI.1 Plaque rupture exposes flowing blood to subendothelial tissues and to potent stimuli that result in platelet aggregation followed by the generation of thrombin, which converts fibrinogen to fibrin (Fig. 1).2 Antiplatelet and anticoagulant drugs, therefore, represent the cornerstone of therapy for acute coronary syndromes and have resulted in a substantial reduction in morbidity and mortality. ASA remains the standard antiplatelet agent and reduces the risk of death or MI by at least 50% in patients with non-ST-segment elevation acute coronary syndromes. ASA has also been shown to reduce the risk of death or reinfarction in the acute phase of transmural MI and in the longer term in post-MI patients.3 Glycoprotein IIb/IIIa inhibitors, a new class of antiplatelet agents, also improve the outcomes of patients with unstable angina and are gaining acceptance as important players in the treatment of acute coronary syndromes, particularly in patients who undergo coronary intervention.4
Fig 1: Low-molecular-weight (LMW) heparin in the treatment armamentarium for acute coronary syndromes. Whether or not a coronary artery is blocked depends in part on a dynamic balance between clot formation (the coagulation cascade) and clot dissolution (the fibrinolytic pathway). LMW heparin interacts with antithrombin causing a physical transformation in the protein that catalyzes its inhibition of the activation of Factor X. Antithrombin also inhibits thrombin directly, which eventually leads to reduced formation of cross-linked fibrin and a shift in favour of clot dissolution. Other therapies used for acute coronary syndromes (ASA, t-PA, urokinase and streptokinase), and their relative points of action, are also depicted. + = promotion of activity, – = inhibition of activity. Photo by: Christine Kenney
Because of the key role of thrombin in the pathogenesis of thrombosis, there is a strong rationale for anticoagulant therapy in addition to antiplatelet therapy for acute coronary syndromes. Unfractionated heparin has been the antithrombotic drug of choice in patients presenting with unstable angina. Several randomized trials conducted in the 1980s and early 1990s suggested its effectiveness in reducing the risk of death and MI following an episode of unstable angina.5 Thus, for the medical management of patients with unstable coronary syndromes, it was recommended that ASA be given in addition to about 3 to 5 days of intravenous therapy with unfractionated heparin to obtain therapeutic partial thromboplastin times between 1.5 and 2.5 times slower than control.6
However, unfractionated heparin has a number of limitations related to its pharmacokinetic and pharmacodynamic properties that make its anticoagulant effect unpredictable and inconsistent in different patients.7 New antithrombotic compounds have thus been developed as possible alternatives to unfractionated heparin and have been tested in the clinical setting of acute coronary syndromes. Among these are low-molecular-weight (LMW) heparins.
Low-molecular-weight heparins
LMW heparins are on average one-third the molecular size of standard heparin. They have been widely tested in rigorous clinical trials in a broad spectrum of thromboembolic disorders and are currently recommended as an alternative to unfractionated heparin in acute coronary syndromes.8 LMW heparins offer a number of potential advantages over heparin (Table 1). The antithrombotic response is much more predictable and, as a consequence, laboratory monitoring of LMW heparin therapy is not required. In addition, LMW heparins have good bioavailability following subcutaneous injections and have a longer half-life than unfractionated heparin.9 Thus, an adequate and persistent anticoagulant effect can be achieved with 1 or 2 daily subcutaneous injections at fixed, weight-adjusted doses. It has been suggested that the rate of anti-factor Xa (anti-Xa) to anti-factor IIa (anti-IIa) activity may be important in differentiating the efficacy and safety of the various LMW heparins, but there are no data to support this hypothesis.8
Table 1.
A number of LMW heparins are available in Canada (Table 2). Of these, dalteparin, enoxaparin and nadroparin are approved for the treatment of unstable angina and are given subcutaneously twice daily in fixed, weight-adjusted doses.
Table 2.
Large-scale clinical trials have been conducted to evaluate LMW heparins in the treatment of unstable angina and non-Q-wave MI. Results from the first randomized clinical trial, a single-blind study that compared nadroparin and heparin, were published in 1995.10 In this trial, 211 patients with unstable angina were randomly assigned to 1 of 3 treatment groups: ASA plus unfractionated heparin, ASA plus nadroparin, or ASA alone. Patients with non-Q-wave MI were excluded. After 5–7 days, when the antithrombotic treatment was stopped, there was a decrease of more than 50% in the rate of recurrent angina and a significant decrease in the rates of silent ischemia and the need for revascularization in the nadroparin group compared with the other 2 groups. Rates of major bleeding were comparable between the LMW and unfractionated heparin groups, and minor bleeding during the acute phase occurred significantly less often in the LMW heparin group. The promising results of this study along with positive data in the management of venous thromboembolism stimulated interest in LMW heparins, and large-scale studies were designed to confirm their role in acute coronary syndromes.
Dalteparin
The first large-scale trial to be published was the Fragmin during Instability in Coronary Artery Disease (FRISC) study,11 which compared the LMW heparin dalteparin with placebo in 1506 patients; all patients received ASA as well. Dalteparin was given in a dose of 120 IU/kg twice daily for 6 days and then as a fixed dose of 7500 IU once daily for 35–45 days to test whether extended use might be beneficial.12,13 During the first 6 days of treatment the rate of death or new MI was lower in the dalteparin group than in the placebo group (13 [1.8%] v. 36 [4.8%]; risk ratio 0.37 [95% confidence interval 0.20–0.68]). The difference persisted at 40 days, but the effect was confined to nonsmokers. The rate did not differ statistically between the 2 groups after 150 days.
In a second trial of dalteparin14 1482 patients with unstable coronary artery disease were randomly assigned to receive ASA plus either dalteparin or unfractionated heparin during the acute phase. The 2 treatments were found to be equivalent in efficacy and safety. As in the previous trial, patients were randomly assigned to continue antithrombotic treatment, with a fixed, low dose of dalteparin once daily, or to take placebo for an additional 39 days. Again, the prolonged treatment did not confer any additional benefit over ASA alone.
The third trial of dalteparin in unstable coronary artery disease was the FRISC II study.15 In addition to comparing an invasive and a noninvasive strategy, the investigators specifically evaluated the benefits of long-term treatment with a significantly higher dose of dalteparin than had been used in the previous trials. All patients received at least 5 days' treatment with dalteparin at therapeutic doses (120 IU/kg twice daily) and were then randomly assigned to receive placebo or to continue taking dalteparin for 3 months at a fixed dose of 7500 IU twice daily (or 5000 IU twice daily in men weighing less than 70 kg and women weighing less than 80 kg). There was a significant decrease in the composite endpoint of death or MI at 30 days in the dalteparin group and a nonsignificant decrease at 3 months. There was no difference between the 2 groups at 6 months. However, during the extended treatment period, rates of major and minor bleeding complications were increased in the dalteparin group. The FRISC II study demonstrated a clear benefit of early intervention with revascularization in moderate- and high-risk patients with unstable coronary artery disease.
Enoxaparin
The third LMW heparin to be tested was enoxaparin. In the ESSENCE trial16 3171 patients with unstable angina or non-Q-wave MI were randomly assigned to receive either enoxaparin (1 mg/kg subcutaneously twice daily) or unfractionated heparin intravenously for at least 48 hours and at most 8 days. At 14 days, the risk of the composite endpoint of death, MI or recurrent angina was significantly lower in the enoxaparin group than in the heparin group (16.6% v. 19.8%, p = 0.019). This benefit was maintained at 30 days and at 1 year.17
In the second study of enoxaparin (the TIMI 11B trial18), patients were randomly assigned to receive either unfractionated heparin for at least 72 hours or therapeutic doses of enoxaparin for up to 8 days, followed by a fixed dose of enoxaparin or placebo once daily for an additional 35 days. At 14 days, the proportion of patients with the primary endpoint of death, MI or need for revascularization was significantly lower in the enoxaparin group than in the heparin group (14.2% v. 16.7%, relative risk reduction 14.9%, p = 0.029). The early benefit was maintained at 43 days, although no additional benefit was observed. The rate of major bleeding events did not differ significantly between the unfractionated and LMW heparin groups, but there was a significant increase in the rate when enoxaparin was compared with placebo in the long-term phase of the study.
A meta-analysis of the 2 studies21 showed that the risk of death, MI or need for urgent revascularization was 20% lower in the enoxaparin group than in the standard heparin group on days 8 and 43 and that there was also a reduction in the risk of death or MI of 23% and 18% at 8 and 43 days respectively (p = 0.02). The incidence of major bleeding events during acute treatment did not differ between the enoxaparin group and the standard heparin group (1.3% and 1.1% respectively, p = 0.35).
Cost analyses of the results of the ESSENCE study in the United States19 and Canada20 have shown an economic benefit for the use of enoxaparin in the management of acute unstable angina or non-Q-wave MI over standard treatment with heparin. Despite the incremental cost of administering the LMW heparin, the improved clinical effectiveness and additional cost savings at hospital discharge resulted in a cost advantage for enoxaparin.
Nadroparin
A large-scale study with nadroparin was recently completed (the FRAXIS study).22 In this study, involving 3468 patients, standard therapeutic doses of unfractionated heparin administered for 6 days were compared to either 6 or 14 days of nadroparin. The composite endpoint of death, MI or refractory angina did not differ among the 3 groups after 6 and 14 days, but was significantly worse at 3 months in the group treated with nadroparin for 14 days. Major bleeding rates were also increased in this group at 14 days and at 3 months.
Advantages of low-molecular-weight heparins over unfractionated heparin
A recently published meta-analysis23 concluded that there was no benefit of LMW heparins over unfractionated heparin in the management of unstable angina and non-ST-segment elevation MI and that there was no benefit to extending the treatment period. However, it has been pointed out that there are a number of differences in the design of the trials that may affect the interpretation of the analysis.24 The LMW heparins used in the studies have different chemical structures, different antithrombotic activities and different pharmacokinetic properties, all of which could result in different clinical outcomes. In addition, the prespecified endpoints of the trials differed from those used in the meta-analysis. Thus, the conclusions drawn from the meta-analysis should not negate the results of the individual trials, which were adequately powered to answer specific clinical questions. A summary of the results of the LMW heparin trials in unstable angina appears in Table 3.
Table 3.
Conclusion
In the past decade, LMW heparins have been extensively evaluated in many clinical settings. For the treatment of venous thromboembolism, they have been proven to be at least as effective as unfractionated heparin in reducing the rate of recurrent thromboembolic events. In acute coronary syndromes LMW heparins have been proven effective and safe in reducing ischemic events, including death, MI and the need for urgent revascularization. For venous thromboembolism, LMW heparins have offered new management perspectives such as outpatient treatment. However, clinical trials that addressed the question of long-term treatment in acute coronary syndromes have failed to show any clear advantage of LMW heparins over standard antiplatelet treatment, despite the evidence of persistent thrombin generation and resultant risk of clinical recurrence with standard antiplatelet treatment. The reason for the lack of a significant clinical benefit with LMW heparin in this setting is unclear. Therefore, further clinical trials of long-term treatment with these agents are needed.
Future studies will examine the effect of combining an LMW heparin and a glycoprotein IIb/IIIa inhibitor, which have shown encouraging results in the setting of acute coronary syndromes, particularly during coronary intervention.
Finally, the issue of which LMW heparin is best has yet to be resolved. So far, only enoxaparin has been shown to have a clear advantage over unfractionated heparin. Whether this result is due by chance to the specific characteristics of the various LMW heparins or by the different designs of the trials is still a matter of debate.
A practical approach to antithrombotic therapy for non-ST-segment elevation acute coronary syndromes has been developed by Fitchett and colleagues.25
Articles to date in this series
-
Armstrong PW. New advances in the management of acute coronary syndromes [editorial]. CMAJ 2001; 164 (9): 1303-4.
-
Fitchett D, Goodman S, Langer A. New advances in the management of acute coronary syndromes: 1. Matching treatment to risk. CMAJ 2001;164(9):1309-16.
-
Armstrong PW. New advances in the management of acute coronary syndromes: 2. Fibrinolytic therapy for acute ST-segment elevation myocardial infarction. CMAJ 2001;165(6):791-7.
-
Buller CE, Carere RG. New advances in the management of acute coronary syndromes: 3. The role of catheter-based procedures. CMAJ 2002;166(1):51-61.
Footnotes
-
An information sheet for patients appears as an appendix 1 at the end of the article.
A patient information sheet appears on the next page.
This article has been peer reviewed.
Contributors: Drs. Ageno and Turpie contributed equally to the preparation of the manuscript.
Competing interests: None declared for Dr. Ageno. Dr. Turpie was an investigator in the TIMI IIB trial (enoxaparin), the ESSENCE trial (enoxaparin) and the FRIC trial (dalteparin) and was a coauthor of the articles reporting their results.
Series editor: Dr. Paul W. Armstrong