Miettinen OS, Henschke CI, Pasmantier MW, Smith JP, Libby DM, Yankelevitz DF. Mammographic screening: no reliable supporting evidence? Lancet 2002;359:404-5.
Background: A Cochrane meta-analysis by Olsen and Gøtzsche,1 in which the authors conclude that “there is no reliable evidence that screening for breast cancer reduces mortality,” has fuelled the debate about whether mammography saves lives. This conclusion has confused clinicians and their patients, who have been told that screening is recommended for women over age 50 in Canada2 and over age 40 in the United States.3
The Cochrane analysis reviewed all clinical trials of breast cancer screening, but only 2 of them met the authors' standards for trial quality: a study conducted in Malmö, Sweden, that followed women for 11 years after study entry4 and a Canadian randomized clinical trial by Miller and colleagues that had a follow-up of 3 to 4 years.5,6 In the Malmö study, there were 63 deaths from breast cancer in the screened group and 66 in the control group, a clinically and statistically nonsignificant difference (relative risk 0.96, 95% confidence interval [CI] 0.68–1.35).
However, a reanalysis of the Malmö study data by Miettinen and colleagues7 has produced a different conclusion and has highlighted the importance of determining the appropriate measure of the usefulness of a screening regimen. Miettinen previously pointed out that screening for early disease detection must be founded on the assumption that starting treatment earlier in detected cases is more likely to cure the disease than starting treatment later in unscreened patients.8 Thus, if we are evaluating screening trials, we should expect to see a benefit, not immediately at the time of screening, but some time later when patients, after having received the treatment, would begin to show the benefit of earlier treatment. For an indolent disease such as breast cancer, this period could easily be delayed for several years. Miettinen and colleagues thus set out to determine if this delayed effect was true in the Malmö study. (They did not examine the Canadian studies because, they reasoned, the follow-up of only 3 to 4 years after screening was too short to show an effect on breast cancer deaths from the screening program.)
Question: Does the Malmö study of breast cancer screening show a benefit of screening and early treatment, after an appropriate amount of time has elapsed in the study period?
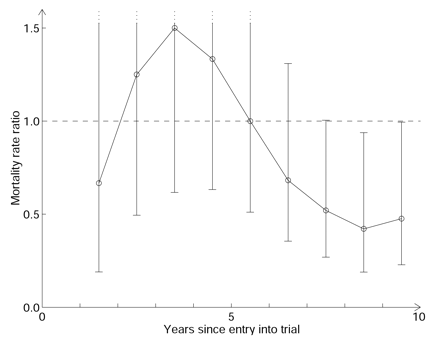
Methods: Miettinen and colleagues examined data available from the Malmö trial for women 55 years of age or older at study entry. They compared deaths from breast cancer in the screened and control groups by year since entry to the trial and determined 3-year moving averages for the 2 populations. They reported the mortality rate ratio and 95% CI specific to each of the successive years after entry into the trial.
Results: There was a statistically significant drop in breast cancer mortality rates that began 6 years after entry into the trial (Fig. 1). In the first 5 years after entry the rates were higher in the screened group than in the control group. After the sixth year, the rates were lower in the screened group than in the control group. On the basis of data for years 8 to 11 (the last year for which data were available), the rate ratio is 0.45 (95% CI 0.24–0.84).
Fig. 1: Breast cancer mortality ratio for women at least 55 years of age in the Malmö study. Shown are point estimates and 95% confidence intervals, based on the deaths in the year at issue together with those in the preceding and following years. Reprinted with permission from Elsevier Science ( Lancet 2002;359:404-5).
Commentary: This is an important study. It has a clinical resonance that makes sense. As Miettinen previously argued,9 it is imperative that trialists get the purpose or object of their study right. One might quibble with the choice of interval for the analysis. Is a 6-year delay too long, or too short? The authors, although they chose years 6 to 11, present the data for all years.
Implications for practice: Physicians should not be too hasty in rejecting mammography for women 55 years of age or older. In addition, they should be skeptical when evaluating reports of screening trials that measure outcomes from the time of entry into the trial. Doing so usually makes no clinical sense.
John Hoey CMAJ