Article Figures & Tables
Figures
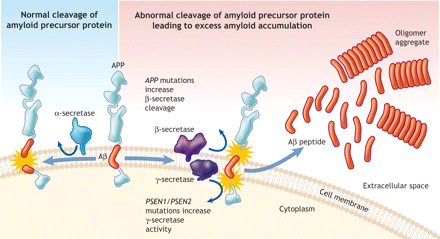
Figure 1: The amyloid precursor protein (APP) is a transmembrane protein that can undergo a series of proteolytic cleavage by secretase enzymes. When it is cleaved by α-secretase in the middle of the β-amyloid domain (Aβ), it is not amyloidogenic. However, when APP is cleaved by β- and γ-secretase enzymes, neurotoxic Aβ peptides are released, which can accumulate into oligomer aggregate. Mutations in the APP gene tend to inhibit cleavage by α-secretase and consequently enable preferential cleavage by β-secretase. Mutations in the presenilin-1 and presenilin-2 genes (PSEN1 and PSEN2), which are components of the γ-secretase complex, increase cleavage by γ-secretase at this site. In both situations, the result is excess Aβ peptide production. The current Aβ hypothesis suggests that the soluble oligomers can impair synaptic function between neurons. Simultaneously, the oligomers may aggregate into insoluble β-sheet amyloid fibrils, which can trigger a local inflammatory response.22 Over time, the subsequent oxidative stress and biochemical changes ultimately lead to neuronal death and the development of neuritic plaques typical of Alzheimer disease. Image by: Lianne Friesen and Nicholas Woolridge
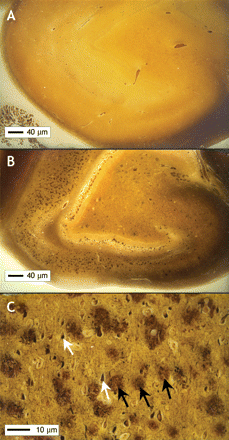
Figure 2: Images of normal hippocampus (A) and hippocampus of a patient with Alzheimer disease (B) [Bielschowsky stain]. The numerous dark brown spots seen in the abnormal hippocampus are the neuritic plaques typical of Alzheimer disease. At higher magnification (C), these plaques (black arrows) and tangles (white arrows) seen in Alzheimer disease are clearly visible. Image by: Images courtesy of Dr. Ian R.A. MacKenzie, Department of Pathology, University of British Columbia