Article Figures & Tables
Figures
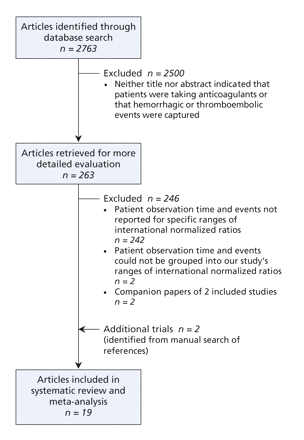
Figure 1: Retrieval and selection of studies involving patients taking oral anticoagulants that captured hemorrhagic or thromboembolic events and person-years of observation.
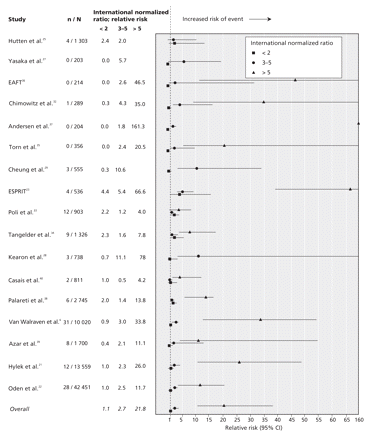
Figure 2: Anticoagulation intensity and risk of hemorrhagic events. An international normalized ratio of 2–3 was the reference range. Studies that did not report events at a ratio greater than 5 reported a maximum range of ratio greater than 3. We grouped these data into the range of 3–5. CI = confidence interval, EAFT = European Atrial Fibrillation Trial Study Group, ESPRIT = European and Australian Stroke Prevention in Reversible Ischaemia Trial, RR = relative risk. Confidence intervals are in Appendix 5, available online at www.cmaj.ca/cgi/content/full/179/3/235/DC2.
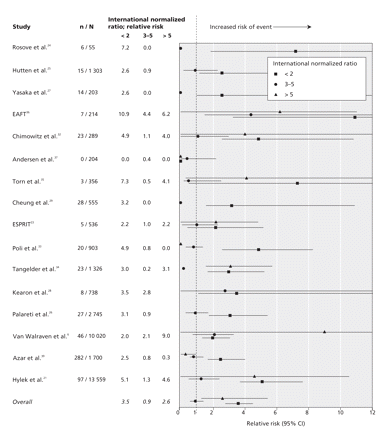
Figure 3: Anticoagulation intensity and risk of thromboembolic events. An international normalized ratio of 2–3 was the reference range. Studies that did not report events at a ratio greater than 5 reported a maximum range of ratio greater than 3. We grouped these data into the range of 3–5. CI = confidence interval, EAFT = European Atrial Fibrillation Trial Study Group, ESPRIT = European and Australian Stroke Prevention in Reversible Ischaemia Trial, RR = relative risk. Confidence intervals are in Appendix 5, available online at www.cmaj.ca/cgi/content/full/179/3/235/DC2.
Figure 4: Anticoagulation intensity and risk of hemorrhagic or thromboembolic events combined. An international normalized ratio of 2–3 was the reference range. Studies that did not report events at a ratio greater than 5 reported a maximum range of ratio greater than 3. We grouped these data into the range of 3–5. CI = confidence interval, EAFT = European Atrial Fibrillation Trial Study Group, ESPRIT = European and Australian Stroke Prevention in Reversible Ischaemia Trial, RR = relative risk. Confidence intervals are in Appendix 5, available online at www.cmaj.ca/cgi/content/full/179/3/235/DC2.