Osteoarthritis in primary care
Osteoarthritis is a common chronic condition that affects many older Canadians and is a considerable cause of disability. Researchers looked at the epidemiology of osteoarthritis in primary care in Canada, using the Canadian Primary Care Sentinel Surveillance Network database, which includes electronic medical record (EMR) data from 340 primary care practices across the country. They analyzed the records of 207 610 patients who were 30 years of age or older and had at least 1 clinic visit in 2011–2012. The estimated prevalence of diagnosed osteoarthritis was 14.2% and was more common in women (15.6% in women v. 12.4% in men). As expected, the prevalence of this condition increased with age. The diagnosis of osteoarthritis was associated with both high and low body mass index. Those with osteoarthritis had an increased risk of comorbidities, including hypertension, depression and chronic obstructive pulmonary disease (Table 1). Over half the patients with osteoarthritis (56.6%) had received a prescription for nonsteroidal anti-inflammatory drugs, 45% of which were topical. Only about one-quarter had acetaminophen in their medication lists, but use of this over-the-counter medication may have been underrecorded in the EMR. About one-third received prescriptions for opioid medications for pain management. CMAJ Open 2015;3:E270–5.
Age- and sex-adjusted prevalence ratios for comorbidity among patients with osteoarthritis
Postmarket safety of drugs approved on basis of clinical and surrogate outcomes
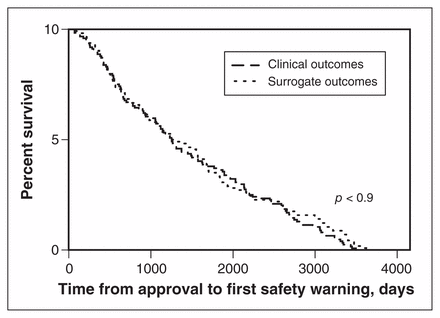
Health Canada approves drugs on the basis of clinical trials that use clinical outcomes, surrogate outcomes or a combination of both. The use of surrogate outcomes allows trials to be done less expensively with fewer patients in a shorter period, with the result that new drugs can reach patients faster. However, some drugs approved on the basis of surrogate outcomes had serious safety problems and were withdrawn from the market or had their indications substantially restricted. This cohort study looked at drugs approved by Health Canada from 2005 to 2014; 124 were approved using clinical outcomes and 114 using surrogate outcomes. The likelihood of drugs acquiring a serious safety warning (as posted on the MedEffect Canada website) after they were marketed was the same in both groups (Figure 1). The median time from market authorization to the first serious safety warning was also similar: 722 days in the clinical outcome group and 818 days in the surrogate outcome group (difference 96 days, 95% confidence interval −295 to 425). Although there was no statistically significant difference in postmarket safety between these two groups of drugs, the authors stress that drugs receiving approval using surrogate outcomes should be used with caution until their clinical benefits are better understood. CMAJ Open 2015;3: E286–91.
Kaplan–Meier curves showing time from approval to first serious safety warning or removal from market for drugs approved by Health Canada on the basis of clinical and surrogate outcomes.