Abstract
BACKGROUND: Although advancements in systemic therapy have improved the outlook for pancreatic adenocarcinoma, it is not known if patients get access to these therapies. We aimed to examine the patterns and factors associated with access to specialized cancer consultations and subsequent receipt of cancer-directed therapy for patients with non-curative pancreatic adenocarcinoma.
METHODS: We conducted a population-based analysis of noncurative pancreatic adenocarcinoma diagnosed over 2005–2016 in Ontario by linking administrative health care data sets. Our primary outcomes were specialized cancer consultation and receipt of cancer-directed therapy (chemotherapy or a combination of chemo- and radiation therapy [chemoradiation therapy]). We examined specialized cancer consultation with hepato-pancreatico-biliary surgery, medical and radiation oncology. We used multivariable logistic regression to identify factors associated with medical oncology consultation and cancer-directed therapy.
RESULTS: Of 10 881 patients, 64.9% had a consultation with specialists in medical oncology, 35.1% with hepatopancreatico-biliary surgery and 24.7% with radiation oncology. Sociodemographic characteristics were not associated with the likelihood of medical oncology consultation. Of these patients, 4144 received cancer-directed therapy, representing 38.1% of all patients and 58.6% of those who consulted with medical oncology. Of 6737 patients not receiving cancer-directed therapy, 2988 (44.4%) had a consultation with medical oncology. Older age and lowest income quintile were independently associated with lower likelihood of cancer-directed therapy. If the first specialized cancer consultation was with medical or radiation oncology, the likelihood of cancer-directed therapy was significantly higher compared with surgery.
INTERPRETATION: A considerable proportion of patients with noncurable pancreatic adenocarcinoma in Ontario did not have a specialized cancer consultation and most did not receive cancer-directed therapy. We identified disparities in specialized cancer consultation and receipt of systemic cancer-directed therapy that indicate potential gaps in assessment.
Despite substantial improvements in cancer care, mortality from pancreatic adenocarcinoma is largely unchanged.1,2 The only option for curative treatment is pancreatectomy.3 However, 80% of patients who present with advanced or metastatic disease are not candidates for resection and require noncurative management.4
Contemporary multiagent chemotherapy regimens can improve symptoms, extend median survival for up to 11 months and delay clinical deterioration in patients with advanced pancreatic adenocarcinoma.5–7 The American Society of Clinical Oncology guideline update in 2018 included chemotherapy for the noncurative management of pancreatic adenocarcinoma in most patients.6 In addition, radiation therapy, nerve blocks and other modalities can be employed to reduce symptom burden.8–10 However, these multimodality treatments require specialized management by a multidisciplinary team of cancer specialists.
Despite those established benefits, a large proportion of patients may not access these treatments.11–15 However, little information is available about the mechanisms underlying these disparities. To optimize delivery of care in patients with noncurative pancreatic adenocarcinoma, it is imperative to roadmap the patterns of health care delivery, and identify barriers and disparities in the delivery of cancer-directed therapy. Therefore, we conducted a population-based study to examine the patterns of and factors associated with access to specialized cancer care and subsequent receipt of cancer-directed therapy for patients with noncurative pancreatic adenocarcinoma. We hypothesized that a substantial proportion of patients with noncurative pancreatic adenocarcinoma do not get access to specialized cancer consultation and cancer-directed therapy.
Methods
Study design
Using data linked from prospectively maintained administrative databases stored at ICES in Toronto, we conducted a population-based cohort study. Under the Canada Health Act, the population of Ontario benefits from universally accessible and publicly funded health care though the Ontario Health Insurance Plan.
This study was reported following the Reporting of studies Conducted using Observational Routinely-collected health Data (RECORD) statement.16
Data sources
The Ontario Cancer Registry is a provincial database that includes all patients with a cancer diagnosis (excluding nonmelanoma skin cancer) in Ontario.17,18 We used the Registered Persons Database (a population-based registry that is maintained by the Ontario Ministry of Health and Long-Term Care) to obtain vital status and demographic data.19 We obtained information about health services from the Canadian Institute for Health Information Discharge Abstract Database, the National Ambulatory Care Reporting System and the Ontario Health Insurance Plan Claims Database (for billing by health care providers). We used the Cancer Activity Level Reporting database to determine what chemotherapeutics and other medications were administered to patients with cancer.
Study population and cohort
We identified patients with a new diagnosis of pancreatic adenocarcinoma over 2005–2016 in the Ontario Cancer Registry using the International Classification of Diseases for Oncology, 3rd Edition (ICD-O-3) codes. We included patients who did not undergo a pancreatectomy from 180 days before the date of diagnosis to the end of follow-up (Supplementary Table 1, Appendix 1, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.190211/-/DC1). We excluded patients if they died before or on the date of diagnosis, had another cancer diagnosis before or after the diagnosis of pancreatic adenocarcinoma, or were less than 18 or greater than 99 years of age at the time of diagnosis.
Demographic and socioeconomic characteristics of patients with noncurative pancreatic cancer, stratified by therapy received
Outcome measures
Our primary outcomes were receipt of specialized cancer consultation and cancer-directed therapy. Specialized cancer consultation refers to the first encounter (initial consultation as opposed to a follow-up visit) with an oncology specialist; the structure and content of the consultation was up to the physician and was not standardized. We defined specialized cancer consultation as 1 or more consultations with specialists in medical oncology, radiation oncology or hepato-pancreatico-biliary surgery from the date of diagnosis to the end of follow-up. We created the following categories: medical oncology, radiation oncology, hepato-pancreaticobiliary surgery and no consultation, whereby 1 patient might have received consultation with more than 1 specialist.
We defined medical and radiation oncologists as physicians who submitted billings using the administration codes for chemotherapy and for radiation therapy, respectively.20 We defined hepato-pancreatico-biliary surgeons as physicians who submitted billings for at least 2 pancreaticoduodenectomies per year over the study period. Hepato-pancreatico-biliary surgery services are provided exclusively in Ontario in 10 designated centres staffed with at least 2 surgeons trained with hepato-pancreaticobiliary fellowships, as per governmental policy.21
To gain insight into initial access to cancer care, we further defined the type of first consultation following diagnosis, either hepato-pancreatico-biliary surgery or oncology (medical or radiation oncology). Each patient was assigned to 1 group only; if assessments for both hepato-pancreatico-biliary surgery and oncology were received on the same day, we assigned the patient to the oncology group. We defined 3 treatment regimens: chemotherapy, a combination of chemo- and radiation therapy (chemoradiation therapy) and best supportive care (including palliative radiation therapy), using physician claims from the Ontario Health Insurance Plan Claims Database.22,23 Cancer-directed therapy included chemotherapy and chemoradiation therapy.
Patients were followed from the date of diagnosis until the date of death, the date of last contact or Mar. 31, 2017, for a minimum of 12 months follow-up for all patients.
Covariates
Variable sources and definitions are detailed in Supplementary Table 1 (Appendix 1). We determined rural living using the postal code of residence and defined it as a Rurality Index of Ontario score greater than or equal to 40.24 We capture income quintile as the median income of a patient’s postal code of residence using national census data.25,26 We measured comorbidity burden using the Johns Hopkins Adjusted Clinical Groups system score dichotomized with a cut-off of 10 for high comorbidity burden, which is consistent with previous reports.27,28 We measured straight-line distances (reported in km) from a patient’s place of residence to both the institution where chemotherapy was administered and to the nearest chemotherapy delivery facility, as a surrogate measure of access to medical oncology care, using latitude and longitude for those geographical points (based on Statistics Canada equations). Finally, we measured survival from the date of diagnosis to the date of death from any cause according to the Registered Persons Database. Details on staging, extent of disease, performance status classification and referring physicians were not available in the data sets.
Statistical analysis
We used descriptive analyses to define baseline characteristics and outcomes. Categorical variables are reported as absolute numbers (n) and proportions (%), and continuous variables as means with standard deviations (SDs) or medians with interquartile ranges (IQRs), as appropriate. We used χ2 tests to compare categorical variables; we used either the Student t test or Kruskal–Wallis test to compare continuous variables, as appropriate. We estimated overall survival using the Kaplan–Meier method, calculated with the life-table method and compared with the log-rank test.
We examined factors associated with specialized cancer consultation and cancer-directed therapy using multivariable regression models. Considering that a medical oncology assessment is the gateway to receiving cancer-directed therapy for noncurative pancreatic adenocarcinoma (chemotherapy), our first model examined medical oncology consultation in the entire cohort of patients. We then assessed factors associated with cancer-directed therapy. For this analysis, the cohort was restricted to patients who had a medical oncology consultation. Relevant demographic and clinical characteristics were identified a priori for inclusion in the models based on clinical relevance (markers of complexity of cancer care) and existing literature (known relation with treatment of pancreatic adenocarcinoma). We included the following covariates: age (categorical), sex, comorbidity burden, income quintile, rural living, time period of diagnosis (2005–2010 v. 2011–2016) and distance to the nearest chemotherapy centre (categorical). For the model examining cancer-directed therapy, we added the type of first specialized cancer consultation (oncology v. hepato-pancreatico-biliary surgery) to the covariates. We used a modified Poisson regression with robust variance and offset for length of follow-up for modelling. The results are reported as risk ratios (RRs) with 95% confidence intervals (CIs).
Considering the high fatality of pancreatic adenocarcinoma, patients may die before having the opportunity to experience the outcome of specialized cancer consultation or cancer-directed therapy. We conducted a sensitivity analysis that was restricted to patients surviving a minimum of 30 days after the date of diagnosis.
All analyses were 2-sided with statistical significance at p ≤ 0.05. We conducted the analyses using SAS Enterprise Guide 6.1 (SAS Institute).
Ethics approval
The study was approved by the Sunnybrook Health Sciences Centre Research Ethics Board.
Results
We included 10 881 patients with a new diagnosis of noncurative pancreatic adenocarcinoma between 2005 and 2016 (Supplementary Figure 1, Appendix 1). The median age at diagnosis was 72 (IQR 62–80) years, and 5450 of the patients (50.1%) were female. The median distance from a patient’s residence to the nearest chemotherapy centre was 6 (IQR 3–14) km. Median overall survival for the cohort was 3.3 (IQR 1.2–8.5) months. Overall, 26.6% of patients (n = 2889) received systemic chemotherapy, 11.5% (n = 1255) received chemoradiation and 61.9% (n = 6737) received best supportive care (Table 1).
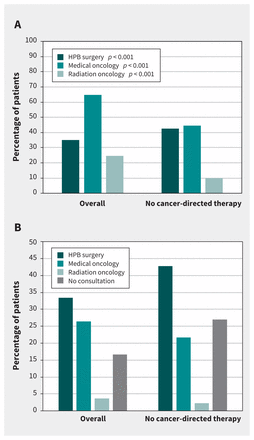
Specialized cancer consultation stratified by receipt of cancer-directed therapy (A) at any time after cancer diagnosis and (B) as the first consultation after diagnosis. Proportions may not add up to 100% because some patients may have seen more than 1 specialist at any time after diagnosis or on the same day as the first consultation. Note: HPB = hepato-pancreatico-biliary.
Patterns of specialized cancer consultation
Overall, 64.9% (n = 7062) of the patients had a consultation with a medical oncologist, 35.1% (n = 3819) with a hepatopancreatico-biliary surgeon and 24.7% (n = 2688) with a radiation oncologist at any time after diagnosis. Of those patients who did not receive cancer-directed therapy, 44.4% (n = 2988) had a consultation with medical oncology (Figure 1A). Median overall survival for patients who consulted with medical oncology was 5.1 (IQR 4.9–5.3) months.
We considered the first specialized cancer consultation as point of access to care, and only 26.4% of all patients consulted with medical oncology first (Figure 1B). Of those patients who did not receive cancer-directed therapy, 73.0% had a specialized cancer consultation; there was a higher proportion who initially consulted with hepato-pancreatico-biliary surgery (42.7%) compared with medical or radiation oncology. Most patients who first consulted with hepato-pancreatico-biliary surgery subsequently had a consultation with medical or radiation oncology (Supplementary Table 2, Appendix 1).
The factors associated with medical oncology consultation are presented in Table 2. A more recent diagnosis over 2011–2016 was associated with a higher likelihood of consultation with medical oncology.
Demographic and socioeconomic characteristics associated with medical oncology consultation
Receipt of cancer-directed therapy
Overall, 4144 patients received cancer-directed therapy, representing 38.1% of all patients and 58.6% of those who consulted with medical oncology. To analyze factors associated with receipt of cancer-directed therapy, we restricted the cohort to patients who consulted with medical oncology (Table 3). Patients over 81 years of age had a lower likelihood of receiving cancer-directed therapy compared with those less than 60 years of age (RR 0.48, 95% CI 0.40–0.57). Patients with a lower income in the 1st quintile (RR 0.80, 95% CI 0.69–0.93) were less likely to receive cancer-directed therapy. However, comorbidity burden, rural residence and distance to the nearest chemotherapy centre were not associated with cancer-directed therapy. Patients who consulted with a medical or radiation oncologist as a first point of cancer care were significantly more likely to receive cancer-directed therapy (RR 1.61, 95% CI 1.46–1.76).
Demographic and socioeconomic characteristics associated with receipt of cancer-directed therapy in patients with specialized cancer assessment
Restricting the analysis to patients who survived a minimum of 30 days (n = 8542) after diagnosis did not alter the results significantly. Of those patients, 75.6% (n = 6458) had a medical oncology consultation and 47.7% received cancer-directed therapy (n = 4075). Among those patients who did not receive cancer-directed therapy, 45.3% (n = 2022) did not see a medical oncologist. We found no difference in the factors associated with medical oncology consultation and receipt of cancer-directed therapy on multivariable analysis or on the size of the observed effect estimates.
Interpretation
In this large population-based study, we investigated access to specialized cancer care and patterns of therapy for noncurative pancreas adenocarcinoma. Two-thirds of patients had consultation with medical oncology, which understandably affected the therapy received. Indeed, of patients not receiving cancer-directed therapy, over half did not have a consultation with medical oncology. We also observed disparities in both specialized cancer consultation and cancer-directed therapy that provide insight into processes of care.
The World Health Organization (WHO) describes 3 dimensions for access to health care: physical accessibility, financial affordability and acceptability.29 Our analysis of patterns of access to specialized cancer care addresses the accessibility dimension of the WHO framework. Previous studies have most commonly addressed acceptability of therapy by focusing on patients receiving chemotherapy and describing the use of different regimens.14,30–32 Some small studies that examined referral patterns were limited by smaller sample sizes or also encompassed curative management, which confused the results.11,12 Rates of medical oncology consultations for noncurative pancreatic adenocarcinoma have been reported at 31% in Nova Scotia and 54% in Alberta, with older age and higher comorbidity burden associated with no consultation.11,33
Our analysis adds to knowledge in this area, as it involves over 10 000 patients, combines data on both access to care and receipt of therapy, and assesses the trajectory in accessing specialized cancer care. We found that Ontario had higher rates of medical oncology consultation compared with other provinces, which may be related to several factors. These factors include different strategies to capture noncurative pancreatic cancer or cancer care systems (including regionalization of pancreatic cancer surgery) that may also influence patterns of referrals for nonsurgical patients. Nevertheless, the observed rates of oncology consultations and cancer-directed therapy for noncurative pancreatic adenocarcinoma contrast with high rates of treatment for other metastatic cancers, despite the ability to prolong median survival to 11 months with cancer-directed therapy for pancreatic adenocarcinoma compared with 6 months without therapy.5,7,34
Although not providing cancer-directed therapy may be appropriate when chemotherapy is not feasible or not aligned with patient preferences, patients should be given equal chances potentially to receive cancer-directed therapy. Our results suggest a possible lack of informed discussion regarding cancer-directed therapy. Knowing that a patient’s initial negative perception of the risks and benefits of chemotherapy is not associated with receipt of chemotherapy, communication and shared decision-making with oncology providers is critical.36 We highlighted issues with under-assessment of patients with noncurative pancreatic adenocarcinoma and a need to change practice to increase the number of consultations with medical oncology. The ideal rate of patients receiving consultation is unknown and could be examined in future health-policy work.
Factors associated with having medical oncology consultation suggest patterns of referrals related to practices, perceptions and beliefs of the primary care providers rather than patient characteristics and physical accessibility of care. Substantial improvement in outcomes with modern chemotherapy was established in 2011, but this information may not have been disseminated widely enough.5,7 Although we observed encouraging higher odds of consultation with medical oncology in more recent years, further work is needed. Patient factors were more relevant with regard to receipt of cancer-directed therapy. It is possible that oncologists are reluctant to provide chemotherapy to older or more socially fragile patients, despite the evidence of benefit.37,38 The main factor associated with receiving cancer-directed therapy was having a first consultation with oncology, highlighting a need to facilitate access to medical oncology.
The reasons underlying the low rate of consultation with medical oncology are likely multifactorial, including provider, patient, and system level factors. From a provider perspective, perceptions of the need and benefits of oncology consultation and therapy by primary care providers can influence referrals. Patients’ perceptions of oncology treatments may also lead to declining referrals. Education for health care providers about the current gap in practice, as well as the rationale and benefit for consultation and treatment, should be established. Patient information is also key. Both should include debunking of the potential stigma of pancreatic cancer. Future work should engage patients to gain further insight into their value of benefits and drawbacks of therapy. The perceptions, attitudes, and heuristics of healthcare providers should also be examined.
Limitations
We conducted a retrospective cohort study using health care administrative data sets that were not collected specifically to address the research question. As such, we lacked some patient, provider and disease details to decipher the decision-making process or indications for consultation and cancer-directed therapy. Therefore, we cannot comment on the reasons why patients were not assessed or not treated, or where the referrals originated. Although surgical cancer care in Ontario is regionalized, this is not the case for medical cancer care, which can be delivered in any centre. There are emerging data showing superior outcomes with high-volume oncology care for cancer-directed therapy.35 However, a detailed analysis of the effect of institution volume on outcomes for medical cancer care was beyond the scope of this study. In particular, it is difficult to identify the institution where a consultation happened, such that the analysis would be limited to volume of cancer-directed therapy delivered by institution and inherently biased.
Conclusion
In our study, a worrisome proportion of patients with noncurative pancreatic adenocarcinoma did not have a specialized cancer consultation, and most did not receive cancer-directed therapy. Although some patients may not have been eligible for therapy, we identified disparities in the receipt of consultation with medical oncology and cancer-directed therapy. We have highlighted the potential under-assessment of patients with noncurative pancreatic adenocarcinoma that can serve as the rationale and foundation for future research, and the design of pathways and policies to optimize the delivery of equitable patient-centred care.
Footnotes
Visual abstract available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.190211/-/DC2
Competing interests: Julie Hallet has received speaking honoraria from Ipsen Biopharmaceuticals Canada and Novartis Oncology. Sten Myrehaug has received speaking honoraria from Ipsen Biopharmaceuticals Canada and Novartis Oncology, and travel support from Novartis Oncology. Natalie Coburn receives salary support from Cancer Care Ontario as the lead for patient-reported outcomes. No other competing interests were declared.
This article has been peer reviewed.
Contributors: Julie Hallet and Natalie Coburn conceived and designed the work. Julie Hallet, Natalie Coburn, Laura Davis, Alyson Mahar, Ying Liu and Kaitlyn Beyfuss acquired the data. Michail Mavros and Julie Hallet drafted the manuscript. All of the authors analyzed and interpretated the data, revised the manuscript critically for important intellectual content, gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Funding: This study was supported by an operating grant from the Canadian Institutes of Health Research (grant no. 154131). Natalie Coburn is the Sherif and Mary-Lou Hanna Chair in Surgical Oncology Research.
Data sharing: The data set from this study is held securely in coded form at ICES. Although data-sharing agreements prohibit ICES from making the data set publicly available, access may be granted to those who meet prespecified criteria for confidential access, available at www.ices.on.ca/DAS. The full data set creation plan and underlying analytic code are available from the authors upon request, understanding that the programs may rely upon coding templates or macros that are unique to ICES.
Disclaimer: This study was supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health and Long-Term Care (MOHLTC). The opinions, results and conclusions reported in this paper are those of the authors and are independent from the funding sources. No endorsement by ICES or the Ontario MOHLTC is intended or should be inferred.
Part of this work was presented as a poster presentation at the ASCO Gastrointestinal Cancers Symposium in San Francisco, California, Jan. 17–19, 2019, and was presented at the Annual Cancer Symposium of the Society of Surgical Oncology held in San Diego, California, Mar. 27–30, 2019.
- Accepted April 26, 2019.