Acute ischemic stroke may be the first clinical manifestation of occult cancer.
A comprehensive diagnostic approach to the causes of ischemic stroke may help identify uncommon etiologies, including occult cancer.
Ensure age- and sex-appropriate cancer screening tests are up to date in survivors of acute ischemic stroke.
A 77-year-old right-handed functionally independent woman presented to the emergency department about 12 hours after the acute onset of speech difficulties. The patient was a nonsmoker and was receiving treatment for hyper-cholesterolemia (low-density lipoprotein cholesterol level at presentation 2.48 mmol/L [target < 2.0 mmol/L or > 50% reduction on treatment) with a moderate dose of atorvastatin, 20 mg/d. She had no other modifiable cerebrovascular risk factors and no personal history of cancer or thromboembolic events. Although a sibling had been diagnosed with colorectal cancer at age 65 years, the patient had never undergone any screening for colorectal cancer. On examination, she had mild aphasia and right pronator drift. A noncontrast head computed tomography (CT) scan showed an ischemic nonlacunar infarct of the superior and middle left temporal gyri. We admitted her to our stroke unit for management and etiologic investigation.
The patient was afebrile, and there was no evidence of infection on chest radiography or urinalysis. Initial blood investigation showed mild normocytic anemia (hemoglobin concentration 116 g/L [normal 120–160 g/L], mean corpuscular volume 90.2 fL [normal 82.0–101.0 fL], normal serum ferritin level and a mildly elevated serum C-reactive protein level (18.6 mg/L [normal < 10.0 mg/L]). Her international normalized ratio and partial thromboplastin time were both within normal limits.
Twenty-four-hour ambulatory electrocardiography did not reveal atrial fibrillation, and transthoracic echocardiography showed mild left atrial enlargement, mild left ventricular diastolic dysfunction and nonsevere (≤ 5 mm) atheroma of the transverse aorta. Further investigations included brain magnetic resonance imaging, which corroborated the CT findings without additional ischemic lesions, and head and neck CT angiography, which did not show significant intracranial or extracranial stenosis.
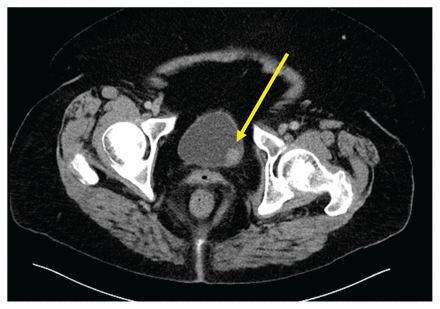
Given the patient’s mild anemia and elevated C-reactive protein level, we suspected occult cancer. We ordered chest–abdomen–pelvis positron emission tomography/CT, which showed a large hypermetabolic mass involving the transverse colon (Figure 1). Subsequent contrast-enhanced abdomen–pelvis CT failed to identify the lesion but, instead, revealed an additional, bulging mass involving the inner bladder wall (Figure 2). Colonoscopy confirmed the presence of an ulcerated infiltrative circumferential lesion located in the right portion of the transverse colon. The pathologic findings were consistent with low-grade colorectal adenocarcinoma (stage III, T3N1M0). Cystoscopy confirmed the presence of a bladder lesion, which was found to be stage I, low-grade urothelial bladder carcinoma.
Axial positron emission tomography–computed tomography scan of the abdomen of a 77-year-old woman with acute ischemic stroke, showing a large hypermetabolic mass involving the transverse colon (arrow).
Contrast-enhanced axial abdomen–pelvis computed tomography scan, showing a bulging mass involving the inner bladder wall (arrow).
Following surgical removal of her colorectal cancer, the patient received adjuvant chemotherapy with orally administered capecitabine, and her bladder cancer was resected cystoscopically. The bladder cancer recurred locally 13 months later, and the patient had a second bladder resection procedure. Twenty-two months later, she remained recurrence-free.
The patient attended an outpatient rehabilitation program for her stroke, where lifestyle modifications regarding diet and exercise were recommended. We prescribed low-dose acetylsalicylic acid (80 mg/d) and an angiotensin-converting enzyme inhibitor (perindopril, 4 mg/d) for hypertension, and increased the dose of atorvastatin to 40 mg/d. The patient remained fully independent after 3 years of follow-up, without any further ischemic events or other identified causes of stroke.
Discussion
We present the case of a 77-year-old woman with an acute ischemic stroke, possibly related to 2 occult malignant lesions diagnosed shortly after her index event. About 4% of people who experience acute ischemic stroke will receive a new cancer diagnosis in the subsequent 2 years, with older age, a history of smoking and an elevated D-dimer level conferring substantial risk.1 In a prospective cohort study, Qureshi and colleagues2 estimated a 20% increase in the age-adjusted cancer rate for the year following an ischemic stroke. Men are at higher risk than women for a poststroke cancer diagnosis.2
The pathophysiology underpinning the association between acute ischemic stroke and cancer is poorly understood. However, cancer-associated hypercoagulability and inflammation may promote acute ischemic stroke through several mechanisms, such as paroxysmal embolism, nonbacterial thrombotic endocarditis and intravascular coagulation.3 Furthermore, adenocarcinomas may secrete mucin into the bloodstream, which facilitates the formation of platelet-rich microthrombi by interacting with cell adhesion molecules.3
When acute ischemic strokes are associated with occult cancer, they tend to recur more often and confer a worse prognosis than strokes that are not associated with cancer.4 An acute ischemic stroke may be regarded as an opportunity to search more actively for cancer in selected cases and to improve the prognosis related to both stroke and malignant disease.3 However, the optimal diagnostic approach to occult cancer in patients with acute ischemic stroke is unclear.3 For example, current Canadian Stroke Best Practice and American Heart Association/American Stroke Association recommendations on the secondary prevention of stroke do not provide guidance on when to suspect and investigate for occult malignant disease.5,6 Nonetheless, clinicians should think of occult cancer as part of a broader differential diagnosis for acute ischemic stroke.
In addition to reviewing the patient’s history of age- and sex-appropriate cancer screening tests (Box 1), clinicians may want to pay attention to risk factors (e.g., smoking, family history) and to symptoms (e.g., fever, night sweats, weight loss, rectorrhagia) and signs (e.g., cachexia, adenopathy) associated with cancer. An identified stroke cause does not exclude occult cancer. However, a higher proportion of patients with cryptogenic strokes than those with an identified cause of stroke have occult cancer,8 and most cancer-associated ischemic strokes are also associated with atrial fibrillation or large-vessel disease.9 Recommendations for cancer screening in the general population apply equally to survivors of acute ischemic stroke, and ensuring adequate screening may reduce cancer-related death in this group.7
Cancer screening recommendations as per the Canadian Task Force on Preventive Health Care guidelines7
Colorectal cancer
For people aged 50–74 yr, fecal occult blood test every 2 yr or flexible sigmoidoscopy every 10 yr
Lung cancer
For people aged 55–74 yr with a ≥ 30 pack-year smoking history who currently smoke or who quit < 15 years ago, low-dose computed tomography every year up to 3 consecutive times*
Routine screening with chest radiography is not recommended
Breast cancer
For women aged 50–69 yr, mammography every 2–3 yr
For women aged 70–74 yr, mammography every 2–3 yr; the decision to undergo screening is conditional on the relative value that a woman places on possible benefits and harms from screening
Cervical cancer
For women aged 25–69 yr, Papanicolaou test every 3 yr
For women aged ≥ 70 yr who have not been adequately screened, continued screening until 3 negative Papanicolaou test results have been obtained
Prostate cancer
Routine screening with prostate-specific antigen test is not recommended
↵* Screening should be carried out only in health care settings with expertise in early diagnosis and treatment of lung cancer.
Several markers of systemic inflammation (e.g., high C-reactive protein and fibrinogen levels, low albumin and hemoglobin levels) and upregulated coagulation activity (e.g., high D-dimer levels) are associated with occult cancer in acute ischemic stroke (Box 2). C-reactive protein levels greater than 20 mg/L, when associated with acute ischemic stroke of undetermined cause, are predictive of occult cancer.11 Fibrinogen levels greater than 600 mg/dL have also been associated with a diagnosis of malignant disease following acute ischemic stroke.11 Multiple ischemic lesions (acute or chronic) involving more than 1 vascular territory on brain imaging are also suggestive of occult cancer.12 A paroxysmal embolism may be suspected in people with a substantial patent foramen ovale on echocardiography, with or without associated deep vein thrombosis on lower extremity venous ultrasonography. Transesophageal echocardiography may be warranted to identify nonbacterial thrombotic endocarditis, as a transthoracic approach has limited sensitivity.3
Risks for occult cancer in acute ischemic stroke
Clinical features
Older age2
Male sex2
Lower body mass index8
History of smoking10
History of heavy drinking10
History of ischemic stroke (before current event)1
History of cancer (past/nonactive)9
Stroke features
Undetermined stroke etiology (after initial investigation)11
Multiple vascular territories involved (on brain imaging)12
Laboratory features
The choice of antithrombotic therapy in cancer-associated stroke is individualized. Anticoagulation is indicated for people with atrial fibrillation, venous thromboembolism and nonbacterial thrombotic endocarditis.3,4 Treatment with low-molecular-weight heparin or a direct oral anticoagulant such as edoxaban may reduce paraneoplastic hypercoagulability.4
Several features in our case led us to suspect occult cancer. Our patient was older, had a first-degree family history of cancer, had not been screened for colorectal cancer and had few known modifiable cerebrovascular risk factors. Her normocytic anemia and slightly elevated C-reactive protein level were compatible with low-grade systemic inflammation.
Our case shows the importance of considering occult cancer in ischemic stroke survivors. It highlights factors that may prompt additional investigations as part of a comprehensive approach to ischemic stroke.
Footnotes
Competing interests: Mark Keezer has received speaker and advisory fees for Eisai Co., Elsevier, Sunovion, Novartis and UCB, and unrestricted educational grants and research grants from UCB and Eisai Co. Laura Gioia has received speaker fees and advisory board honoraria from Bayer, BMS-Pfizer and Servier, and investigator-initiated funding from Servier. No other competing interests were declared.
This article has been peer reviewed.
The authors have obtained patient consent.
Contributors: All of the authors contributed to the design and writing of the manuscript, approved the final version to be published and agreed to be accountable for all aspects of the work.