Abstract
BACKGROUND: Patients receiving in-centre hemodialysis are at high risk of exposure to SARS-CoV-2 and death if infected. One dose of the BNT162b2 SARS-CoV-2 vaccine is efficacious in the general population, but responses in patients receiving hemodialysis are uncertain.
METHODS: We obtained serial plasma from patients receiving hemodialysis and health care worker controls before and after vaccination with 1 dose of the BNT162b2 mRNA vaccine, as well as convalescent plasma from patients receiving hemodialysis who survived COVID-19. We measured anti–receptor binding domain (RBD) immunoglobulin G (IgG) levels and stratified groups by evidence of previous SARS-CoV-2 infection.
RESULTS: Our study included 154 patients receiving hemodialysis (135 without and 19 with previous SARS-CoV-2 infection), 40 controls (20 without and 20 with previous SARS-CoV-2 infection) and convalescent plasma from 16 patients. Among those without previous SARS-CoV-2 infection, anti-RBD IgG was undetectable at 4 weeks in 75 of 131 (57%, 95% confidence interval [CI] 47% to 65%) patients receiving hemodialysis, compared with 1 of 20 (5%, 95% CI 1% to 23%) controls (p < 0.001). No patient with nondetectable levels at 4 weeks developed anti-RBD IgG by 8 weeks. Results were similar in non-immunosuppressed and younger individuals. Three patients receiving hemodialysis developed severe COVID-19 after vaccination. Among those with previous SARS-CoV-2 infection, median anti-RBD IgG levels at 8 weeks in patients receiving hemodialysis were similar to controls at 3 weeks (p = 0.3) and to convalescent plasma (p = 0.8).
INTERPRETATION: A single dose of BNT162b2 vaccine failed to elicit a humoral immune response in most patients receiving hemodialysis without previous SARS-CoV-2 infection, even after prolonged observation. In those with previous SARS-CoV-2 infection, the antibody response was delayed. We advise that patients receiving hemodialysis be prioritized for a second BNT162b2 dose at the recommended 3-week interval.
Patients with end-stage kidney disease receiving incentre hemodialysis have been uniquely vulnerable during the COVID-19 pandemic. For these patients, unlike for most other people, self-isolation to avoid exposure to SARS-CoV-2 is impossible. Most patients receiving hemodialysis must leave their homes 3 times weekly to receive their life-saving treatments, often in shared spaces for hours at a time. COVID-19 case fatality rates are 20%–30% for patients receiving hemodialysis —10 times higher than in the general population.1,2 Advanced age, multiple comorbidities and blunted immune response likely all contribute to the high COVID-19 death rates in this population. Some hemodialysis centres have thus prioritized these patients for vaccination.
To facilitate wider vaccine distribution during current shortages, 3 the National Advisory Committee on Immunization of Canada has recommended delaying the second dose of the BNT162b2 vaccine from 3 to 16 weeks.4 In a randomized controlled trial (RCT), the clinical efficacy of the BNT162b2 was reported to be greater than 80% at 3 weeks after the first dose.5 However, no patients receiving hemodialysis were enrolled in this trial.5 Patients with end-stage kidney disease receiving hemodialysis often have impairments in both humoral and cellular immune responses6 and are noted to have lower antibody responses to other vaccines.7 Whether patients receiving hemodialysis develop robust immune responses after vaccination against SARS-CoV-2 remains uncertain.8 Data are required to better inform Canadian public health policy on whether second doses of vaccine can be safely delayed in this population.
Usually, once clinical trials are completed, antibody levels can be used as surrogate measures of vaccine efficacy, such as with hepatitis B9 and influenza.10 With respect to the novel coronavirus SARS-CoV-2, although there is increasing understanding of the antibodies that best correlate with viral neutralization and T-cell responses,11,12 assays vary from laboratory to laboratory and as yet there are no internationally accepted standards defining what antibody levels constitute immunity.13 The only way to evaluate vaccine efficacy using antibody levels, therefore, is through direct experimental comparison with controls who are known to reliably develop immunity after vaccination (i.e., healthy individuals similar to those enrolled in the RCT showing vaccine efficacy5) or who have developed immunity after natural infection (i.e., survivors of COVID-19).
We sought to determine whether short-term antibody responses after a single dose of the BNT162b2 mRNA vaccine are comparable between patients receiving hemodialysis and healthy individuals, and how this compares with antibody responses in patients receiving hemodialysis who survived natural infection with SARS-CoV-2.
Methods
Study participants
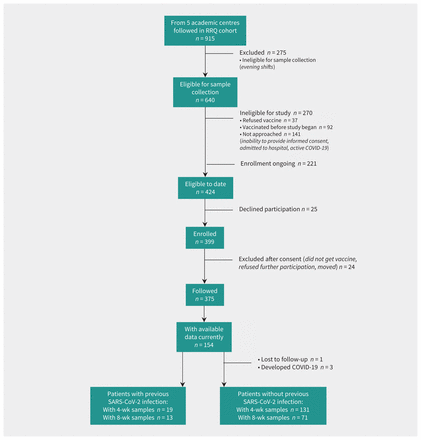
We have been prospectively following in-centre patients receiving hemodialysis in the Réseau Rénal Québécois/Quebec Renal Network (RRQ/QRN) COVID-19 study since March 2020, with participation of 54 of Quebec’s 55 hemodialysis units. For the present vaccine substudy, all patients dialyzing on daytime shifts in 5 academic centres who were able to understand the protocol and provide informed consent were approached for recruitment (Figure 1) according to a prespecified written protocol. We obtained clinical data and serial plasma samples before and every 4 weeks after vaccination with the BNT162b2 (Pfizer-BioNTech) vaccine from patients receiving hemodialysis. All patients completed a symptoms questionnaire 7 days after vaccination. The present study reports available 4- and 8-week data on patients who were vaccinated the weeks of Jan. 26, Feb. 26 and Mar. 4, 2021.
Study flow diagram for patients receiving hemodialysis. Note: RRQ = Réseau Rénal Québécois.
Health care workers without any comorbidities from the Centre hospitalier de l’Université de Montréal who received 1 dose of the BNT162b2 vaccine and volunteered for a healthy control study14 served as controls, with plasma obtained before and 3 weeks after vaccination. We also obtained convalescent plasma from patients receiving hemodialysis who had survived COVID-19, 4–16 weeks after the first positive polymerase chain reaction (PCR) test for SARS-CoV-2. We chose this control group on the presumption that patients receiving hemodialysis who had survived COVID-19 would have had an adequate immune response.
Antibody measurements
We assessed immunoglobulin G (IgG) and immunoglobulin M (IgM) antibodies against the receptor binding domain (RBD) of SARS-CoV-2 spike glycoprotein in plasma samples using an enzyme-linked immunosorbent assay (ELISA), as previously described.14–16 Receptor binding domain is required for SARS-CoV-2 to bind to the host angiotensin-converting enzyme 2 (ACE-2) receptor allowing viral entry and represents the target of the BNT162b2 vaccine.5 We chose anti-RBD antibodies as they have been shown to elicit viral neutralization better than anti-spike antibodies.17 We used bovine serum albumin and CR3022 monoclonal antibody (mAb) (a commercially available human monoclonal antibody specific to RBD18) as negative and positive controls for the ELISA assays, respectively. We reported anti-RBD IgG and IgM levels as relative light units (RLU) normalized to CR3022 mAb.14–16 We established the seropositive threshold as the mean RLU from COVID-19 negative plasma obtained from 10 volunteers prepandemic, plus 3 standard deviations above this mean.14,15 We have previously shown excellent reliability of this assay in detecting anti-RBD IgG levels in a cross-sectional cohort of individuals infected with SARS-CoV-2: anti-RBD IgG levels were detectable with this assay in 85% of infected individuals at 14 days, and in 100% by 53 days after symptom onset.14 Finally, we measured antinucleocapsid IgG (anti-N, ELISA, Abbott Architect 1200SR) to rule out new asymptomatic natural infection after vaccination.
Clinical outcomes
We are following patients longitudinally for development of COVID-19. Since March 2020, all patients have been assessed for COVID-19 using a systematic screening questionnaire and temperature measurement at each hemodialysis treatment session. Patients who fail screening undergo PCR testing of nasopharyngeal swabs for SARS-CoV-2 infection. Since April 2020, routine surveillance PCR testing has been performed during outbreaks and, in 1 hemodialysis unit, weekly. We record information on PCR results, clinical course, hospital admission and death in the study database.
Statistical analysis
Because we sought to include all data available at the time of analysis, we did not perform a formal sample size calculation. We stratified groups by past SARS-CoV-2 infection (positive PCR for SARS-CoV-2 infection or positive anti-RBD before vaccination). We compared median RLUs using the Mann–Whitney test. We compared the proportion with undetectable IgG (nonresponders) against the χ2 test in all patients, and in age and immunosuppression subgroups, and calculated 95% confidence intervals (CI) using the Wilson score method. We performed an exploratory multivariable linear regression model with a patient-level random intercept to assess the direction and strength of the association between receiving hemodialysis and log-transformed anti-RBD IgG levels, adjusting for age, sex and previous SARS-CoV-2 infection. Finally, we assessed correlation of anti-RBD level with age using Pearson’s correlation coefficient. We performed analyses in Stata (Statcorp 2019). Two-sided p values of < 0.01 were considered significant, considering multiple comparisons.
Ethics approval
The research ethics boards of Hôpital du Sacre-Coeur and McGill University Health Centre approved this study, and all patients provided written informed consent.
Results
Figure 1 shows how we arrived at our cohort of patients receiving hemodialysis. We enrolled 154 patients receiving hemodialysis (135 without and 19 with previous SARS-CoV-2 infection) who had data available at the time of analysis, of whom 1 was lost to follow-up and 66 had not yet reached the 8-week time point. We obtained complete baseline and 3-week follow-up data on 40 controls (20 without and 20 with previous SARS-CoV-2 infection) and convalescent plasma data from 16 patients. Most patients receiving hemodialysis were male and considerably older than controls, and most had diabetes mellitus (Table 1). No patients reported severe symptoms within 7 days after vaccination.
Baseline characteristics of patients and controls in the Réseau Rénal Québécois/Quebec Renal Network COVID-19 study
Symptomatic COVID-19 occurred in 4 patients, 18 to 46 days after vaccination. Of these patients, 3 were admitted to hospital for hypoxia; as of this writing, 1 remains in the intensive care unit, 1 has died, and 1 has been discharged. No patient was identified to have asymptomatic SARS-CoV-2 infection after vaccination, as defined by presence of new anti-N antibody or serial PCR.
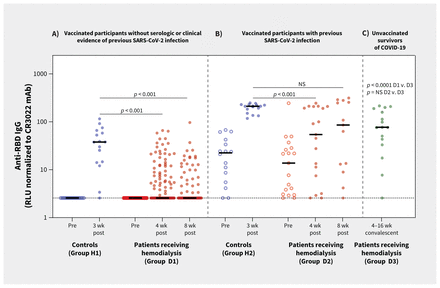
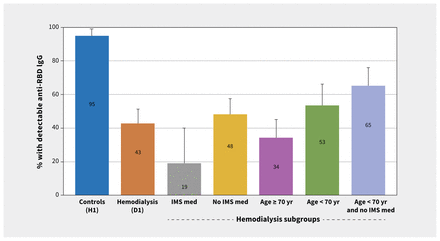
Among patients without previous SARS-CoV-2 infection, those receiving hemodialysis exhibited significantly lower median anti-RBD IgG levels at both 4 weeks (3 RLU, interquartile range [IQR] 3 to 8) and 8 weeks (3 RLU, IQR 3 to 9) after vaccination, compared with controls at 3 weeks (38 RLU, IQR 14 to 64, p < 0.001 for both comparisons), and compared with convalescent plasma from patients receiving hemodialysis who were survivors of COVID-19 (77 RLU, IQR 18 to 199, p < 0.001) (Table 2 and Figure 2). Among patients receiving hemodialysis, 75 of 131 (57%, 95% CI 47% to 65%) had anti-RBD IgG levels that were nondetectable, compared with 1 of 20 controls (5%, 95% CI 1% to 23%, p < 0.001). The proportion of responders (i.e., with detectable anti-RBD IgG) was significantly lower in all hemodialysis subgroups we examined, compared with controls (Figure 3). Importantly, none of the patients receiving hemodialysis with nondetectable antibodies at 4 weeks developed detectable anti-RBD by 8 weeks. In those with detectable levels at 4 weeks, anti-RBD IgG levels were still significantly lower in patients receiving hemodialysis than in controls, and these did not increase by 8 weeks.
Median anti–receptor binding domain immunoglobulin G levels in patients in the Réseau Rénal Québécois/Quebec Renal Network COVID-19 study
Anti–receptor binding domain (RBD) immunoglobulin G (IgG) levels in patients receiving hemodialysis and in controls. Note: mAb = monoclonal antibody, NS = not significant, RLU = relative light units. Postvaccine samples were taken 21 days (range 16–28 d) after a single dose of BNT162b2 mRNA vaccination in controls, and after 26 days (range 24–35 d) and 56 days (range 54–59 d) in patients receiving hemodialysis. Anti-RBD IgG levels were measured using enzyme-linked immunosorbent assay (ELISA), subtracting the signal obtained with bovine serum albumin, and normalized to the signal obtained with CR3022 mAb to SARS-CoV-2 in each plate. Solid horizontal lines represent medians. Dotted lines represent the “positive threshold for detection” of anti-RBD IgG, defined as the mean RLU plus 3 standard deviations from plasma samples obtained from 10 volunteers before the pandemic. See Table 2 for anti-RBD levels and p values. (A) Vaccinated patients receiving hemodialysis and who have no previous SARS-CoV-2 infection (n = 131) and vaccinated controls who have no previous SARS-CoV-2 infection (n = 20). (B) Vaccinated patients receiving hemodialysis (n = 19) and vaccinated controls (n = 20), each with evidence of previous SARS-CoV-2 infection. (C) Convalescent plasma obtained from patients receiving hemodialysis (n = 16) between 4 and 12 weeks after COVID-19.
Proportion of individuals without previous SARS-CoV-2 infection in various subgroups with detectable anti–receptor binding domain (RBD) immunoglobulin G (IgG) after vaccination. Note: IMS med = immunosuppressive medication. Error bars represent 95% confidence intervals by the Wilson score method. p values are given for each subgroup versus controls. Differences between controls and patients receiving hemodialysis or subgroups were all statistically significant (age < 70 yr with no immunosuppressive medication, p = 0.009; all other subgroups, p < 0.001).
The 19 patients receiving hemodialysis who had evidence of previous SARS-CoV-2 infection included 10 with both positive PCR and anti-RBD IgG, 2 with positive PCR only, and 7 with positive anti-RBD IgG only. In these patients, median anti-RBD IgG levels 4 weeks after vaccination were significantly lower than those of controls with previous SARS-CoV-2 infection 3 weeks after vaccination (54 RLU, IQR 8 to 208 v. 211 RLU, IQR 158 to 227, p < 0.001), but not significantly different from levels in convalescent plasma (77 RLU, IQR 18 to 199, p = 0.7) (Table 2 and Figure 2). By 8 weeks, anti-RBD IgG levels had increased to levels comparable with those in controls at 3 weeks (86 RLU, IQR 11 to 267, p = 0.3) (Table 2). There was no significant difference between patients and controls in the proportion of nonresponders at 4 weeks (3 of 19 [16%] v. 0 of 20 [0%], p = 0.06) (Table 2).
There was no difference in median anti-RBD IgM levels at 4 weeks between vaccinated controls and patients receiving hemodialysis, either in individuals without previous SARS-CoV-2 infection (4 RLU, IQR 4 to 4 v. 4 RLU, IQR 4 to 4, p = 0.96), or with previous SARS-CoV-2 infection (4 RLU, IQR 4 to 12 v. 4 RLU, IQR 4 to 6, p = 0.1).
In the multivariable linear regression model, log anti-RBD IgG levels were significantly associated with previous exposure to SARS-CoV-2 and with receiving hemodialysis but less strongly with age. We observed no significant association with sex (Table 3). When we repeated the model excluding those on immunosuppressive medication, we observed similar results. In patients without previous SARS-CoV-2 infection, age was weakly correlated with anti-RBD levels (R = −0.39, p < 0.001, Appendix 1, available at www.cmaj.ca/lookup/doi/10.1503/cmaj.210673/tab-related-content).
Predictors of log-transformed anti–receptor binding domain immunoglobulin G level
Interpretation
We found that patients receiving hemodialysis without previous SARS-CoV-2 infection showed lower anti-RBD IgG levels than healthy controls after a single dose of the BNT162b2 vaccine, with a majority failing to seroconvert at 4 weeks. Importantly, patients receiving hemodialysis who did not respond at 4 weeks remained nonresponders at 8 weeks, which is an argument against the possibility of a delayed response in these individuals. Older patients and those on immunosuppression had even lower seroconversion rates, but even younger patients not on immunosuppression had a significantly lower seropositivity rate than controls. Humoral response was somewhat better after singledose vaccination in patients who had been previously infected with SARS-CoV-2. In this group, most responded with anti-RBD IgG levels similar to healthy controls by 8 weeks and comparable with levels observed in convalescent plasma from patients receiving hemodialysis who survived COVID-19.
In an RCT, the BNT162b2 vaccine was shown to be greater than 80% clinically effective by 3 weeks after the first dose in healthy individuals.5 In the absence of internationally accepted surrogate measures of vaccine efficacy against SARS-CoV-2,13 our healthy control group serves as a benchmark representing the expected “optimal” antibody response. Indeed, 98% of our controls developed anti-RBD IgG within 3 weeks of a single dose of this vaccine, irrespective of previous SARS-CoV-2 infection, which is consistent with recent studies.19,20
Our results suggest that 1 dose of the BNT162b2 vaccine is insufficient to elicit a robust humoral immune response in most patients receiving hemodialysis who have never been previously exposed to SARS-CoV-2, casting doubt on whether a single dose is effective in this population. In contrast to healthy individuals in whom the second dose can probably be safely delayed,20 the same is likely not true for most patients receiving hemodialysis. We do not know if anti-RBD IgG confers immunity against SARS-CoV-2 infection, but it is likely a good surrogate, as anti-RBD IgG correlates strongly with viral neutralization,21 Fc-mediated effector functions and cellular responses.22 Further, in our study, all survivors of COVID-19 mounted an anti-RBD response 4–16 weeks after infection. We cannot rule out that vaccinated patients receiving hemodialysis with undetectable anti-RBD IgG may have developed protective cellular immune responses, but this is unlikely as, in previous studies, neutralization, Fc function and SARS-CoV-2–specific T-cell responses have been observed only in individuals who elicited RBD-specific antibodies.22,23 The most definitive proof of vaccine efficacy is the absence of the development of clinical COVID-19 after vaccination. Four patients in our cohort developed symptomatic COVID-19 18–46 days after vaccination; 3 of these had the usual variant, 1 of whom died. Given this small number and without a nonvaccinated comparison group, it is premature to draw conclusions as to whether these infections constitute true “vaccine failures”; further study is needed. However, it is highly likely that patients receiving hemodialysis require a second dose of vaccine to achieve efficacy. In our study, patients receiving hemodialysis who survived COVID-19 reliably developed anti-RBD IgG, as did those who received 1 dose of BNT162b2 if they had been previously infected. This suggests that patients receiving hemodialysis can develop humoral immune responses to SARS-CoV-2 if the antigen challenge is sufficient. This is consistent with previous studies showing that patients receiving hemodialysis may require higher or repeated doses of hepatitis B vaccine to elicit a response,7 and 1 recent study showing that 94% of patients receiving hemodialysis developed detectable anti-spike IgG antibodies 30 days after 2 doses of BNT162b2.24 To vaccinate Canada’s population of patients receiving hemodialysis with a second dose at the recommended 3-week interval would entail early allocation of about 25 000 doses, which in our view is modest, considering the high risk of exposure and COVID-19 case fatality rate of this population.
Longitudinal follow-up of our cohort is ongoing, and additional patients are being recruited for this study as vaccinations roll out across the province. Whether humoral responses improve and cellular immune responses develop in our cohort after a second dose, and at what interval, requires further study. Tracking the rate of symptomatic SARS-CoV-2 infection after vaccination in this and other studies will also aid greatly in our assessment of vaccine efficacy. If immune responses remain poor or variable after 2 doses, routine measurement of post-vaccination antibodies in the clinical setting may be required in this population, with use of a different vaccine platform in nonresponders, as suggested by Windpessi and colleagues.8
Our study has several strengths. We were able to obtain serial plasma samples before and after vaccination in all patients, and had 2 control groups against whom we could compare our surrogate measure of vaccine efficacy. Inclusion of vaccinated, healthy individuals allowed us to compare the antibody levels we observed in vaccinated patients receiving hemodialysis against the levels achieved in individuals expected to reliably develop acquired immunity to SARS-CoV-2,5 and inclusion of patients receiving hemodialysis who were survivors of COVID-19 allowed us to determine what levels are theoretically achievable in this population. We were reliably able to determine history of SARS-CoV-2 infection based on both prevaccination anti-RBD IgG and serial PCR data from regular surveillance testing, as anti-RBD IgG and anti-N have suboptimal sensitivity for detecting remote SARS-CoV-2 infection. Our cohort is similar in characteristics to the general population of patients receiving hemodialysis, as described in a recent population-based study from Ontario.2
Limitations
We did not measure T-cell responses and therefore may have underestimated the ability of vaccination to provide protective immunity. Follow-up is as yet short, and therefore we had very limited time to observe cases of COVID-19 after vaccination. We are unable to draw inferences about the causes of poor humoral response to vaccination in patients receiving hemodialysis in this study. Finally, the absence of a control group matched for age and comorbidities precludes us from evaluating hemodialysis as an independent risk factor for poor anti-SARS-CoV-2 antibody response.
Conclusion
The efficacy of the BNT162b2 SARS-CoV-2 vaccine in patients receiving hemodialysis remains uncertain. A single dose failed to elicit a humoral immune response in most patients receiving hemodialysis without previous SARS-CoV-2 infection even after prolonged observation, and in those with previous SARS-CoV-2 infection, the response was delayed. Whether patients receiving hemodialysis develop effective T-cell responses requires further study. Until then, we advise that the second dose of the BNT162b2 vaccine be administered to patients receiving hemodialysis at the recommended 3-week time interval, and that rigorous SARS-CoV-2 infection prevention and control measures be continued in hemodialysis units until vaccine efficacy is known.25
Acknowledgements
This work is submitted by the authors on behalf of the COVID-19 Study Team of the Réseau Rénal Québécois. The authors thank the participants for donating their plasma samples. We are grateful to our research coordinators, Ms. Norka Rios, Ms. Marie-Line Caron, and Ms. Guylaine Marcotte. We thank Dr. Nick Bertos and the Biobank Technology Platform of the Research Institute of the McGill University Health Centre for sample processing and storage.
Footnotes
Competing interests: None declared.
This article has been peer reviewed.
Contributors: Rita Suri, William Beaubien-Souligny and Annie-Claire Nadeau-Fredette contributed to the conception and design of the work. Rita Suri, Rémi Goupil, Annie-Claire Nadeau-Fredette, Lakshman Gunaratnam, Caroline Lamarche, Mehdi Benlarbi, Debashree Chatterjee, Guillaume Goyette and Andrés Finzi contributed to the acquisition and interpretation of the data. Rita Suri, Alexander Tom, Rémi Goupil and Annie-Claire Nadeau Fredette analyzed the data. Rémi Goupil and Rita Suri drafted the manuscript. Rita Suri, William Beaubien-Souligny, Annie-Claire Nadeau-Fredette, Andrés Finzi, Lakshman Gunaratnam and Caroline Lamarche revised it critically for important intellectual content. All of the authors gave final approval of the version to be published and agreed to be accountable for all aspects of the work. Mehdi Benlarbi is co-first author and Andrés Finzi is co-senior author.
Funding: Rita Suri, Rémi Goupil, Annie-Claire Nadeau-Fredette and William Beaubien-Souligny are supported by the Fonds de Recherche du Québec – Santé (FRQS) Chercheur-boursiers-clinicien (Clinician Researcher) Awards. Andrés Finzi is the recipient of Canada Research Chair on Retroviral Entry no. RCHS0235950-232424. This study was funded by a Canadian Institutes of Health Research (CIHR) Rapid Research COVID-19 funding opportunity. This work was also supported by le Ministère de l’Économie et de l’Innovation du Québec, Programme de soutien aux organismes de recherche et d’innovation to Andrés Finzi, by the Fondation du CHUM and by a CIHR Foundation Grant No. 352417 to Andrés Finzi.
Data sharing: De-identified data are available upon request to the lead author, Rita Suri (rita.suri{at}mcgill.ca). Further information and requests for reagents should be directed to and will be fulfilled by Andrés Finzi (andres.finzi{at}umontreal.ca).
- Accepted April 29, 2021.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/