Abstract
Background: Recent data suggest an increased risk of congenital anomalies with prenatal exposure to opioid analgesics. We sought to further quantify the risk of anomalies after opioid analgesic exposure during the first trimester in a population-based cohort study.
Methods: Using administrative health data from Ontario, we followed 599 579 gestational parent–infant pairs from singleton pregnancies without opioid use disorder. We identified opioid analgesics dispensed in the first trimester and congenital anomalies diagnosed during the first year of life. We estimated propensity score–adjusted risk ratios (RRs) between first trimester exposure (any opioid analgesic and specific agents) and congenital anomalies (any anomaly, organ system anomalies, major or minor anomalies and specific anomalies).
Results: The prevalence of congenital anomalies was 2.8% in exposed infants and 2.0% in unexposed infants. Relative to unexposed infants, we observed elevated risks among those who were exposed for some anomaly groups, including gastrointestinal anomalies (any opioid analgesic: adjusted RR 1.46, 95% confidence interval [CI] 1.15–1.85; codeine: adjusted RR 1.53, 95% CI 1.12–2.09; tramadol: adjusted RR 2.69, 95% CI 1.34–5.38) and several specific anomalies, including ankyloglossia (any opioid: adjusted RR 1.88, 95% CI 1.30–2.72; codeine: adjusted RR 2.14, 95% CI 1.35–3.40). These findings persisted in sensitivity analyses.
Interpretation: Although the absolute risk of congenital anomalies was low, our findings add to accumulating data that suggest a small increased risk of some organ system anomalies and specific anomalies with first trimester exposure to opioid analgesics. These findings further quantify the potential risks associated with prenatal exposure to opioid analgesics to inform treatment choices for pain in pregnancy.
Opioid use in pregnancy is a public health concern,1–5 with 2%–4% of pregnancies exposed to prescribed opioid analgesics.6–8 Opioid analgesics cross the placenta and have potential to cause fetal harm.9
Evidence concerning the safety of opioid analgesics for pain in pregnancy is discrepant and limited.10–13 Recent studies with Medicaid and private insurance health data from the United States suggested small increases in minor congenital anomalies and oral cleft anomalies with prenatal exposure to opioid analgesics.10,14 Some studies have reported an increased risk of any anomaly,15,16 heart anomalies,7,16–18 spina bifida,7,19,20 oral cleft anomalies,10,15,21 gastroschisis7 and clubfoot16,22,23 with exposure. Other studies, however, have found no association with any anomaly,22,24 major10,14,19,22,24,25 or minor14 anomalies, neural tube anomalies, 10,19,22,26 clubfoot10 or heart anomalies.8,15,19,22,27,28 Discrepant findings likely arise from different methodologies, including definitions of exposure and anomalies evaluated. Some studies included pregnant people with exposure to opioid agonist therapy and, therefore, opioid use disorder.20,22,23 Many studies captured exposure through maternal self-report.7,8,15–18,20–24,26,28,29 Others predominantly included births before 1990,8,15,17,18,21,25,27,28 preventing evaluation of current medications. Only 2 population-based cohort studies have been conducted.22,24
Therefore, we undertook a population-based cohort study to estimate associations between opioid analgesic exposure during the first trimester and congenital anomalies using health administrative data capturing all narcotic prescriptions during pregnancy.
Methods
Study design and data sources
We constructed a population-based cohort of deliveries using administrative health data of the single-payer health care system in Ontario, Canada, held at ICES. We identified parent–infant pair records for all live births and stillbirths at more than 20 weeks’ gestation delivered at Ontario hospitals with an estimated date of confinement between Apr. 7, 2013 (280 d from implementation of the Narcotic Monitoring System [NMS]), and Mar. 31, 2018, in MOMBABY, a validated database of linked parent–infant records. We used estimated date of confinement rather than delivery date to prevent overselection of preterm births and, thus, anomalies.30 To reduce confounding, we excluded pregnant people with opioid use disorder or opioid overdose within 2 years before delivery or those treated with methadone or buprenorphine for opioid use disorder.1,31,32 We also excluded pairs without a valid Ontario health card, pairs with unsuccessful record linkage, pregnant people aged 50 and older, those with multiple fetuses and pregnancies exposed to opioid analgesics during the second or third trimester, but not the first.
Exposure
We captured exposure using records of prescribed prenatal opioid analgesics in the NMS database. Established in July 2012 through the Ontario Narcotics Strategy, the NMS records information on controlled drug prescriptions issued to Ontario residents, regardless of payment method. The exposure of interest was to opioid analgesics during the first trimester, defined as a prescription fill date between the estimated date of conception (following a validated algorithm;33 Appendix 1, available at www.cmaj.ca/lookup/doi/10.1503/cmaj.211215/tab-related-content) and less than 14 weeks’ gestation. We classified exposure as any opioid analgesic and specific agents. The referent group was unexposed to any opioid analgesic during the index pregnancy period.
Outcomes
We identified congenital anomalies diagnosed within the first year of life using diagnosis codes from the Canadian version of the International Classification of Diseases, 10th Revision in the Hospital Discharge Abstract Database, the Same Day Surgery Database and the National Ambulatory Care Reporting System Database, which record mandatory hospital submissions (Appendix 1, Supplemental eTable 1). As in other studies,34–36 we classified anomalies using the algorithm from the Metropolitan Atlanta Congenital Defects Program, a surveillance system of the US Centers for Disease Control and Prevention (Appendix 1, Supplemental eTable 2).37, 38 We defined anomalies as major (of medical or surgical importance) or minor (associated with minor medical or cosmetic significance). We classified them as any anomaly, by organ system, major or minor37 and specific anomalies if at least 5 infants with the anomaly were exposed.
Confounders
We computed high-dimensional propensity scores (HDPS) for parent–infant pairs to ensure similarity of pairs by exposure. This method was developed for pharmacoepidemiologic administrative health data studies39 and has been used with ICES data.6,40,41 Briefly, using the HDPS algorithm, candidate covariates in the year before conception were identified empirically in the health care claims databases we selected (Appendix 1, Supplemental eTable 1). We prioritized covariates by their potential to control for confounding, and integrated them into an exposure propensity score.39 We forced the following a priori confounders into the HPDS: gestational parent age, parity, socioeconomic status quintile, Elixhauser comorbidity score, diabetes, obesity, hypertension, pain, other prescribed psychotropic medications (only data on benzodiazepines or barbiturates were available in the NMS), and year of delivery. The resultant HDPS (between 0 and 1) for each parent–infant pair represents their probability of being treated with opioid analgesics conditional on the confounders. We further stabilized the HDPS to account for extreme weights.42
Statistical analysis
Given the large study size, we compared gestational parent–infant characteristics by exposure with standardized differences; we considered differences greater than 0.10 statistically meaningful.43 We estimated risk ratios (RR) between opioid analgesic exposure during the first trimester and any congenital anomaly, organ system anomalies, major or minor anomalies, and specific anomalies using a log-binomial regression model. We estimated adjusted risk ratios using inverse probability of treatment weighting with HDPS.
Sensitivity analysis
Sensitivity analyses included redefining the exposure period to include the 4 weeks before conception to evaluate possible misclassification; excluding individuals with opioid analgesic use in the year before pregnancy for possible confounding; analyzing individuals with more than 1 pregnancy, for which at least 1 was exposed and 1 was unexposed, to evaluate confounding; excluding infants born earlier than 37 weeks’ gestation (for analysis of atrial septal defect, hypertrophic pyloric stenosis, ankyloglossia possibly related to prematurity); and stratifying by sex for effect measure modification (for hypertrophic pyloric stenosis). Additionally, we estimated adjusted risk differences to provide information on absolute effect size. Finally, we used 2 methods to explore the likely effect of potential unmeasured confounding. First, we determined the effect of an unmeasured confounder needed to fully account for our observed increased risk.44 Second, we used simple bias analysis45 to further remove possible unmeasured confounding arising from incomplete data on prenatal medications; we used estimates from previous studies (confounder–outcome association 1.5–3.0, and prevalence of psychotropic medication use by exposure group).46
Ethics approval
This study was approved by the Queen’s University Health Sciences Research Ethics Board.
Results
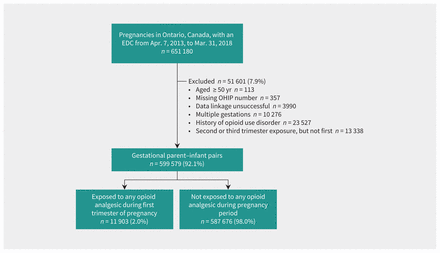
Of 651 180 births in Ontario during the study period, 599 579 (92.1%) were included in the study cohort (Figure 1). The 599 579 pregnancies occurred among 491 060 individuals, 111 055 (22.6%) of whom were nulliparous. Of the 599 579 pregnancies, 11 903 (2.0%) were exposed to opioid analgesics. Most pairs were exposed during the first trimester only (n = 9023, 75.8%) with 1195 (10.0%) exposed all 3 trimesters; the mean duration of exposure was 4.3 (standard deviation 9.2) weeks. Specific agents included codeine (n = 6524), oxycodone (n = 2885), hydromorphone (n = 1824), tramadol (n = 781), morphine (n = 670), fentanyl (n = 75), meperidine (n = 56) and others (n = 41). Standardized differences (Table 1) showed that exposed pairs were more likely to have been prescribed opioid analgesics in the year before pregnancy (47.6% v. 10.1%), to have a prescription for other psychotropics (8.4% v. 1.4%), and to have an Elixhauser comorbidity score of 1 or more (2.2% v. 0.7%) than those who were unexposed. Those exposed were also more likely to have a prior maternal diagnosis of pain (26.3% v. 9.8%), particularly lower back pain (20.9% v. 7.9%). Most covariates were balanced after HDPS weighting, except for history of opioid analgesic use and prescribed psychotropics.
Flow chart of cohort creation. Note: EDC = estimated date of conception, OHIP = Ontario Health Insurance Plan.
Characteristics of pregnancies exposed to opioid analgesics in the first trimester, compared with pregnancies unexposed during the pregnancy period in Ontario, Canada, 2013–2018
Overall, 12 260 (2.0%) infants received a congenital anomaly diagnosis in the first year of life; 329 (2.8%) of 11 903 exposed infants received a diagnosis of an anomaly, compared with 11 931 (2.0%) of 587 676 unexposed infants. The number of anomalies per infant was similar by exposure group; 230 (69.9%) exposed infants with anomalies had 1 anomaly, compared with 7973 (66.8%) of those unexposed (Appendix 1, Supplemental eTable 3).
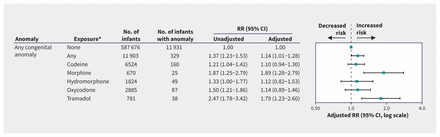
Figure 2 shows the unadjusted and adjusted RRs between exposure to opioid analgesics in the first trimester and any anomaly. After HDPS adjustment, infants exposed to any opioid analgesic had a small, elevated risk of any congenital anomaly (adjusted RR 1.14, 95% confidence interval [CI] 1.01–1.28) for an adjusted prevalence difference of 2.9 per 1000 infants (Appendix 1, Supplemental eTable 4). We observed an increased risk of any anomaly with exposure to morphine (adjusted RR 1.89, 95% CI 1.28–2.79) and tramadol (adjusted RR 1.79, 95% CI 1.23–2.60).
Forest plot of the risk of any congenital anomaly in pregnancies exposed to opioid analgesics in the first trimester compared with those not exposed during the pregnancy period, by specific agent. We adjusted estimates using inverse probability of treatment weighting with high-dimensional propensity scores. *We present data on specific opioid analgesic agents where at least 5 infants with the anomaly were exposed. Note: CI = confidence interval, RR = risk ratio.
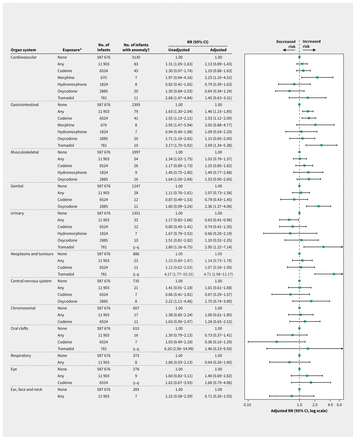
When classified by organ system and specific anomalies (Figure 3, Figure 4 and Figure 5), exposure was associated with increased risks of cardiovascular anomalies (with morphine), neoplasms and tumours (with tramadol), gastrointestinal (with any opioid analgesic and with codeine) and genital anomalies (with oxycodone). The risk of urinary anomalies was lower with any opioid analgesic exposure (adjusted RR 0.63, 95% CI 0.41–0.96) and higher with tramadol (adjusted RR 2.95, 95% CI 1.22–7.14).
Forest plot of the risk of congenital anomalies in pregnancies exposed to opioid analgesics in the first trimester compared with those not exposed during the pregnancy period, classified by organ system. We adjusted estimates using inverse probability of treatment weighting with high-dimensional propensity scores. *We present data on specific opioid analgesic agents where at least 5 infants with the anomaly were exposed. †Given ICES privacy restrictions, we report anomalies with 6 or fewer cases as 5–6. Note: CI = confidence interval, RR = risk ratio.
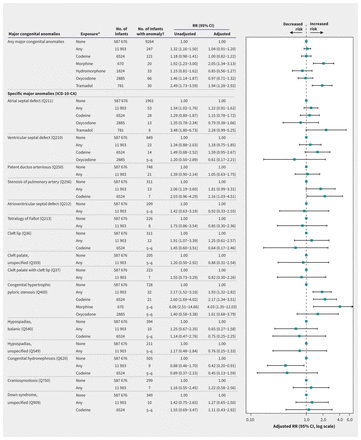
Forest plot of the risk of specific major congenital anomalies in pregnancies exposed to opioid analgesics in the first trimester compared with those not exposed during the pregnancy period, by specific agent. We adjusted estimates using inverse probability of treatment weighting with high-dimensional propensity scores. *We present data on specific opioid analgesic agents where at least 5 infants with the anomaly were exposed. †Given ICES privacy restrictions, we report anomalies with 6 or fewer cases as 5–6. Note: CI = confidence interval, ICD-10-CA = diagnostic code from the Canadian version of the International Classification of Diseases, 10th Revision, RR = risk ratio.
Forest plot of the risk of specific minor congenital anomalies in pregnancies exposed to opioid analgesics in the first trimester compared with those not exposed during the pregnancy period, by specific agent. We adjusted estimates using inverse probability of treatment weighting with high-dimensional propensity scores. *We present data on specific opioid analgesic agents where at least 5 infants with the anomaly were exposed. †Given ICES privacy restrictions, we report anomalies with 6 or fewer cases as 5–6. Note: CI = confidence interval, ICD-10-CA = diagnostic code from the Canadian version of the International Classification of Diseases, 10th Revision, RR = risk ratio.
The risk of major anomalies was elevated with exposure to morphine (adjusted RR 2.05, 95% 1.34–3.13, Figure 4) and tramadol (adjusted RR 1.94, 95% CI 1.28–2.92). Associations with specific major anomalies included atrial septal defect (with tramadol), ventricular septal defect (with codeine), pulmonary artery stenosis (with any opioid analgesic and with codeine) and hypertrophic pyloric stenosis (with any opioid analgesic, codeine and morphine). The risk of minor anomalies was elevated with exposure to any opioid analgesic, codeine, hydromorphone and oxycodone, and the risk of ankyloglossia was elevated with any opioid analgesic and codeine (Figure 5).
Specific agents associated with multiple anomalies included tramadol (with atrial septal defect, neoplasms and tumours, gastrointestinal anomalies and urinary anomalies), codeine (with gastrointestinal anomalies, ankyloglossia, hypertrophic pyloric stenosis, ventricular septal defect and pulmonary artery stenosis) and morphine (with cardiovascular anomalies and hypertrophic pyloric stenosis). Only oxycodone was associated with a single anomaly group (genital anomalies).
In sensitivity analyses (Table 2), when including exposure 4 weeks before conception or excluding individuals with exposure to opioid analgesics before pregnancy, results were unchanged from the primary analyses. Among 2980 individuals with at least 1 exposed and 1 unexposed pregnancy, exposure to any opioid analgesic was associated with an increased risk of any anomaly (adjusted RR 1.13, 95% CI 0.83–1.55). An increased risk of ankyloglossia and hypertrophic pyloric stenosis with exposure to any opioid analgesic remained when restricting to term infants. We observed a stronger association between any opioid analgesic exposure and hypertrophic pyloric stenosis in female infants (adjusted RR 3.97, 95% CI 2.11–7.50) than male infants (adjusted RR 1.49, 95% CI 0.92–2.41). For confounding to fully account for our observed increased risk, most observed associations required risk ratios greater than 3 between the possible unmeasured confounder and the exposure and anomalies (Appendix 1, Supplemental eTable 5). Most findings persisted after further adjustment for possible unmeasured confounding (Appendix 1, Supplemental eTable 6).
Results of sensitivity analyses
Interpretation
In this population-based cohort of 599 579 parent–infant pairs, the prevalence of congenital anomalies was 204.4 per 10 000 infants; this is lower than the prevalence of 430.5 per 10 000 infants in the 2014 general Canadian pregnancy population.47 Although the overall risk was low, we observed an increased risk of any congenital anomaly with tramadol,16 and a previously unreported risk with morphine. An association with oxycodone was observed only when including exposure 4 weeks before conception. Associations with any anomaly, however, are less informative: any anomaly is a crude classification and may not capture differences in etiology.48
Previous studies reported elevated risks of heart anomalies with first trimester exposure to any opioid analgesic,7 codeine7,17,18 and tramadol,16 but others reported no association with any opioid analgesic8,10,15,19,22 or codeine.10,19,22,27,28 A US case–control study reported increased risks of specific heart anomalies with periconceptional or first trimester exposure to opioid analgesics compared with acetaminophen.29 Like others,7,16–18 we observed associations with some cardiovascular anomalies; however, these did not persist in bias analysis.
Few studies have evaluated hypertrophic pyloric stenosis,15,16,22 a major stomach anomaly leading to gastric outlet obstruction that requires surgery;49 we observed an increased risk with any opioid analgesic, codeine and morphine. We observed a stronger association among female infants than male infants, which persisted in bias analysis. Hypertrophic pyloric stenosis is more common in male than female infants.50,51 It is unknown why the baseline risk differs by sex; similarly, we cannot explain why sex would modify associations with exposure to opioid analgesics. This observation may be hypothesis-generating rather than suggestive of opioid analgesic teratogenicity.
Others have observed associations with exposure to codeine in the second and third trimesters and to oxycodone in the third trimester.14 We observed an elevated risk of minor anomalies with any opioid analgesic, codeine, oxycodone and hydromorphone; associations were strongest for hydromorphone in bias analysis. We noted associations between any opioid analgesic and codeine with ankyloglossia, commonly known as tongue-tie, which is sometimes treated with release of the frenulum. We also observed other elevated risks: gastrointestinal anomalies with any opioid analgesic, codeine and tramadol; genital anomalies with oxycodone; and neoplasms and tumours and urinary anomalies with tramadol. We attribute the observed lower risk of urinary anomalies with any opioid analgesic to type I error. The difficulty of accurate exposure measurement is that etiology and time of organogenesis can vary within an organ system, which could reduce the specificity of estimated associations.48
Multiple sensitivity analyses suggested that misclassification of exposure or anomalies likely did not account for our results. Our HDPS adjustment attenuated some estimates, but elevated associations remained for some anomalies. Consistent with others,10 our quantitative bias analysis estimated that an unmeasured confounder would have to be strongly associated with exposure and anomalies to fully account for associations. To correct estimates for possible unmeasured confounding from psychotropic medications (only benzodiazepines and barbiturates are available in the NMS and were thus used as a proxy for other prenatal medications), we used psychotropic medication prevalence from a pregnant Medicaid population, 46 which was higher than what would be expected in our cohort. Most associations persisted, supporting the small increased risk observed by others.10,14 Previous studies of administrative health data did not observe confounding from selective serotonin reuptake inhibitors or benzodiazepines in a high-risk cohort of pregnant people.31
Our methods improve upon some limitations of previous studies. Rather than using maternal recall, we used data from a central database that captures all prescriptions of controlled substances to capture prescriptions of opioid analgesics during pregnancy;7,8,15–18,20,22–24,26,28,29,52 only a small proportion of NMS records (< 3%) were not linked. Our contemporary population-based study adds robust data to population-based studies from Sweden22 and Norway.24 Studies of Medicaid and private insurance beneficiaries often exclude many parent–infant pairs to ensure complete pregnancy coverage data;10,14 this is not a concern with our data set. Further, we systematically identified anomalies using the classification system from the Metropolitan Atlanta Congenital Defects Program.
Limitations
We identified cases using diagnostic codes for billing, which may not be entirely accurate; the diagnosis and documentation of minor anomalies and those with subtle medical significance could be vulnerable to exposure-dependent recording bias. A small number of exposed infants with certain anomalies reduced precision, which could have led to spurious associations and prevented evaluation of some previously reported associations. Deliveries before 20 weeks’ gestation were not recorded; therefore, early losses and terminations (potentially owing to anomalies) were not captured. Although we had extensive prescription data, we did not have data on over-the-counter pain medications that may have been used by pregnant people. Lastly, we determined exposure by the prescription fill date without further confirmation of use; this could have attenuated our associations. Future research investigating specific anomalies and agents is warranted, given the smaller number of exposed infants.
Conclusion
Although the absolute risk of anomalies was low, our study adds to those suggesting a small increased risk of congenital anomalies with exposure to opioid analgesics. Both the potential for harm or distress to the pregnant person as a consequence of forgoing treatment and the subsequent risk to the infant must be considered for effective treatment. These findings further quantify harms associated with prenatal exposure to opioid analgesics to inform treatment choices for pain in pregnancy.
Footnotes
Competing interests: Susan Brogly reports honoraria from the National Institutes of Health and a grant from the Canadian Institutes of Health Research, outside the submitted work. No other competing interests were declared.
This article has been peer reviewed.
Contributors: Martha Werler, Maria Velez, Astrid Guttmann and Susan Brogly contributed to the conception and design of the work. Wenbin Li, Astrid Guttmann and Susan Brogly contributed to the acquisition of the data. Alexa Bowie and Wenbin Li contributed to the analysis of the work. All authors contributed to the interpretation of data for the work. Alexa Bowie and Susan Brogly drafted the manuscript, and all authors revised critically for important intellectual content. All authors gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Funding: This study received funding from the Eunice Kennedy Shriver National Institutes of Child Health and Human Development under grant number 1R03HD099617-01.
Data sharing: The full list of diagnosis codes and anomalies is available by request to the authors. The data set from this study is held securely in coded form at ICES. Although data sharing agreements prohibit ICES from making the data set publicly available, access may be granted to those who meet prespecified criteria for confidential access, available at www.ices.on.ca/DAS. The full data set creation plan and underlying analytic code are available from the authors upon request, understanding that the programs may rely upon coding templates or macros that are unique to ICES.
Disclaimer: This study was supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health (MOH). Parts of this material are based on data and information compiled and provided by MOH, the Canadian Institute of Health Information (CIHI) and the Registrar General through Service Ontario. Parts of this material are based on data or information compiled and provided by CIHI. However, the analyses, conclusions, opinions and statements expressed in the material are those of the authors, and not necessarily those of CIHI. The analyses, conclusions, opinions and statements expressed herein are solely those of the authors and do not reflect those of the funding or data sources; no endorsement is intended nor should be inferred. The authors thank IQVIA Solutions Canada Inc. for use of their Drug Information File.
- Accepted December 16, 2021.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/