See related commentary on page 1201 and at www.cmaj.ca/lookup/doi/10.1503/cmaj.141252
Prostate cancer is the most commonly diagnosed non–skin cancer in men and the third leading cause of cancer-related death among men in Canada.1 The current estimated lifetime risk of diagnosis is 14.3%, whereas the lifetime risk of death from prostate cancer is 3.6%.2 The prevalence of undiagnosed prostate cancer at autopsy is high and increases with age (> 40% among men aged 40–49 yr to > 70% among men aged 70–79 yr).3 Most cases of diagnosed prostate cancer have a good prognosis; the 10-year estimated relative survival ratio is now 95%, the highest among all cancers in men.1
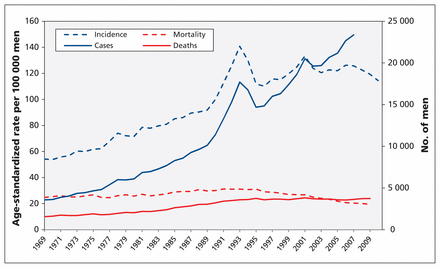
In Canada, the age-standardized rate of death from prostate cancer rose from 1969 to 1991, followed by a decline of 37.5% from 1992 to 2009, at an average rate of 2.6% per year (Figure 1). In 1990, the estimated age-standardized mortality was 30 cases per 100 000, and in 2010 it was just below 20 per 100 000.1 However, over the same period, the number of cases and the age-standardized incidence of prostate cancer both increased. Subsequent to the introduction and adoption of prostate-specific antigen (PSA) testing, the incidence of prostate cancer increased rapidly from 1990 to a peak in 1993 and a second, less-pronounced peak in 2001 (Figure 1). Much of the excess incidence represents overdiagnosis,4,5 that is, the detection of cancers that would not progress to cause symptoms or death.6
Cases of and deaths from prostate cancer, with associated age-standardized incidence and mortality (per 100 000 men), among Canadian men aged 45 years and older. Age was standardized to the 1991 Canadian population. Incidence data were not available for Quebec from 2008 to 2010; therefore, the population denominator for age-standardized incidence was adjusted and case counts for 2008–2010 were omitted. Mortality data were available only to 2009.
There is no conclusive evidence to determine what proportion of the decline in prostate cancer mortality is due to screening versus improved treatment, or other factors; it is likely that both screening and treatment have contributed.7 If PSA screening were the primary reason for the decrease in mortality, the steep increase in incidence due to early case detection associated with screening should have been followed by a sharp reduction in mortality. Instead, the reduction in prostate cancer mortality over time has been relatively steady and began too soon after the test’s introduction to be attributed mainly to PSA screening.7–10
This guideline provides recommendations on screening for prostate cancer using the PSA test with or without digital rectal examination in men in the general population. The guideline updates a prior guideline by the task force that was last published in 1994.11
Methods
The Canadian Task Force on Preventive Health Care is an independent panel of volunteer clinicians and methodologists that makes recommendations about clinical manoeuvres aimed at primary and secondary prevention (www.canadiantaskforce.ca). Work on each set of recommendations is led by a workgroup of two to six members of the task force. Each workgroup establishes the research questions and analytical framework for the guideline. More information about the task force’s methods can be found elsewhere12 and on the task force website (http://canadiantaskforce.ca/methods/methods-manual/).
The development of these recommendations was led by a workgroup of six members of the task force and scientific staff at the Public Health Agency of Canada. Guideline development was based on an analytical framework (Appendix 1, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.140703/-/DC1) that established the overall purpose and background of the guideline, framed the literature review and outlined the key and contextual research questions. The analytical framework was reviewed by all members of the task force and underwent external review by national and international stakeholders with expertise in prostate cancer before guideline development. The key and contextual questions in the analytical framework examined the benefits and harms of PSA screening with or without digital rectal examination and the benefits and harms of treatment of prostate cancer on decreasing prostate cancer mortality and all-cause mortality.
The Evidence Review and Synthesis Centre at McMaster University (Hamilton, Ont.) conducted a systematic review of the available evidence with the aid of a urologist, who served as an independent technical advisor, and scientific staff at the Public Health Agency of Canada.13 The systematic review was done according to the final, peer-reviewed protocol (http://canadiantaskforce.ca/perch/resources/ctfphc-prostate-cancer-screening-protocol.pdf) of the analytical framework and followed the methods described in the procedure manual.14 Because a previous search by the US Preventive Services Task Force was used to identify data published before 2007, the timeline to review the benefits of screening was extended from Jan. 1, 2007, to Nov. 30, 2013. The search for harms data went from 2003 to Nov. 30, 2013. The timeline for the treatment review was from Jan. 1, 2007, to Nov. 30, 2012. The search was updated Aug. 15, 2014, to include eligible trials that reported extended follow-up. Databases searched included PubMed, Ovid MEDLINE, MEDLINE, the Cochrane Central Register of Controlled Trials and the Cochrane Database of Systematic Reviews. The methods and findings of the systematic review13 can be obtained at www.canadiantaskforce.ca. The task force used the Grading of Recommendations Assessment, Development and Evaluation (GRADE) system to determine the quality of evidence and strength of recommendations (Box 1).15 The protocol, systematic review and guideline underwent external peer review by academic and clinical experts.
Grading of recommendations
Recommendations are graded according to the Grading of Recommendations Assessment, Development and Evaluation (GRADE) system.15 GRADE offers two strengths of recommendation: strong and weak. The strength of recommendations is based on the quality of supporting evidence, the degree of uncertainty about the balance between desirable and undesirable effects, the degree of uncertainty or variability in values and preferences, and the degree of uncertainty about whether the intervention represents a wise use of resources.
Strong recommendations are those for which the task force is confident that the desirable effects of an intervention outweigh its undesirable effects (strong recommendation for an intervention) or that the undesirable effects of an intervention outweigh its desirable effects (strong recommendation against an intervention). A strong recommendation implies that most individuals will be best served by the recommended course of action.
Weak recommendations are those for which the desirable effects probably outweigh the undesirable effects (weak recommendation for an intervention) or the undesirable effects probably outweigh the desirable effects (weak recommendation against an intervention) but appreciable uncertainty exists. A weak recommendation implies that most men would want the recommended course of action but that many would not. For clinicians, this means they must recognize that different choices will be appropriate for each person, and they must help each patient arrive at a management decision consistent with his values and preferences. Policy-making will require substantial debate and involvement of various stakeholders. Weak recommendations result when the balance between desirable and undesirable effects is small, the quality of evidence is lower, or there is more variability in the values and preferences of patients.
The quality of evidence is graded as high, moderate, low or very low, based on how likely further research is to change our confidence in the estimate of effect.
Benefits of PSA screening
A summary of the data described in this section can be found in Appendix 2 (available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.140703/-/DC1), with detailed tables available in the systematic review.13
The literature search for the systematic review identified six randomized controlled trials (RCTs).16–21 Because three of the trials16–18 were considered to have a high risk of bias, evidence from these trials was not considered for this guideline. None of the excluded studies showed benefit from prostate cancer screening. Findings from all sites of the European Randomized Study of Screening for Prostate Cancer (ERSPC),19 a multi-country study, were considered together when we formulated the recommendations.
A small absolute reduction in prostate cancer mortality was reported in the ERSPC study.19 The investigators found that the risk of death from prostate cancer was reduced in the screening group (relative risk [RR] 0.79, 95% confidence interval [CI] 0.69–0.91; absolute risk reduction 0.128%, or 13 lives saved per 10 000 men invited for screening), with a pretrial PSA testing rate of 20%.13,19 However, there was evidence of heterogeneity between study centres, with some finding that screening reduced prostate cancer mortality while others did not.
In contrast, the Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial (PLCO) found no effect of screening on prostate cancer mortality (RR 1.09, 95% CI 0.87–1.36; 0 deaths from prostate cancer prevented per 10 000 invited for screening).21 Participants in the PLCO trial had a high rate of pretrial PSA testing (52%), but sensitivity analysis found no effect of pretrial PSA testing on the results. High rates of opportunistic PSA screening were observed in the control group, however, which decreased the opportunity to show a beneficial effect of screening. Therefore, although the lack of benefit observed in the PLCO trial reduces confidence that PSA screening truly does reduce prostate cancer mortality, the task force placed relatively more weight on the findings of the ERSPC study.
There was no demonstrable effect of PSA screening on all-cause mortality in the PLCO or ERSPC studies (Table 1).13,15,19,21
Evidence of benefit of screening for prostate cancer with PSA testing
Digital rectal examination has been traditionally used to screen for prostate cancer, but no trials have examined the mortality benefit of this screening method when used alone.22 The PLCO study used both PSA and digital rectal examination for screening of all participants. In contrast, some of the ERSPC study centres used PSA as the primary screening test, followed by digital rectal examination depending on the PSA test result; other centres used both methods for initial screening but stopped using digital rectal examination in later screening rounds. The systematic review for the current guideline found no evidence that digital rectal examination reduces mortality when used in conjunction with the PSA test.13
Harms of screening
The harms of PSA screening relate to the investigation and treatment of prostate cancer in men with true- and false-positive test results and in men whose detected prostate cancer would not have caused them symptoms or death (i.e., over-diagnosis) (Table 2).13,15,19,21
Evidence of harms from screening for prostate cancer with PSA testing*
False-positive results
The rate of false-positive results varies depending on the PSA test threshold, ranging from 11.3% of all screened men at a threshold of 4.0 ng/mL to 19.8% at a threshold of 3.0 ng/mL.23,24 The PLCO study used a threshold of 4.0 ng/mL (annual screening), and most sites in the ERSPC study used a threshold of 3.0 ng/mL (screening every 4 yr). Lower thresholds can increase the number of cases of cancer detected, but they also increase the risk of a false-positive result and diagnosis of indolent tumours of no clinical significance. No level of PSA excludes prostate cancer.25,26 Other factors that increase the risk of a false-positive PSA test result include urinary tract infection and benign prostatic hypertrophy.
Harms of biopsy
Men who have a positive PSA test result are typically invited for further testing, which usually includes prostate biopsy. Depending on the chosen strategy for the PSA test threshold and the frequency of testing, men who undergo PSA screening could potentially have multiple biopsies over time, thereby increasing their risk of harms.
Harms of prostate biopsy include hematuria, infection, hospital admission and death. Observational studies showed that an estimated 31% of men (310 men per 1000) had hematuria up to 30 days after biopsy and that 0.9% of men (9 per 1000) had infection.13 A meta-analysis found that the more serious complications of hospital admission occurred in 21 men per 1000 (2.1%, 95% CI 1.6%–2.5%) and death in 2 per 1000 (0.2%, 95% CI 0.1%–0.2%) (Table 2).13
Overdiagnosis
Overdiagnosis occurs when cancer is detected correctly but would not cause symptoms or death. Estimates of the frequency of overdiagnosis can be influenced by the PSA test threshold used, the frequency of screening, the patient’s age, the PSA volume, the disease risk and the number of core samples taken during biopsy. Estimates can also vary depending on how they are calculated (i.e., whether the denominator is the number of patients screened, the number with a positive PSA test result or the number with a diagnosis of prostate cancer). In the ERSPC trial, the estimated prevalence of overdiagnosis ranged from 40% to 56% of men screened who received a diagnosis of prostate cancer.13
Benefits and harms of treatment
Two RCTs27,28 showed that radical prostatectomy reduced prostate cancer mortality among men with symptomatic early prostate cancer. Cohort studies reported decreased prostate cancer mortality29–34 and all-cause mortality29–36 associated with radical prostatectomy. There was no trial evidence to indicate that radiation therapy improves clinical outcomes in men with prostate cancer, although pooled analyses of observational data suggested that radiation therapy (alone or in combination with hormonal therapy) reduces prostate cancer mortality and all-cause mortality.30–37 No studies showed that hormonal therapy decreased all-cause or prostate cancer mortality, and no studies of the effect of cryotherapy or high-intensity focal ultrasonography on all-cause or prostate cancer mortality were identified.13
Radical prostatectomy, radiation therapy and androgen deprivation therapy are the most common treatments of prostate cancer and are associated with potential harms. In RCTs that compared radical prostatectomy with watchful waiting, men in both study arms had the potential to receive other treatments, which made it difficult to estimate the harms from a specific treatment. After 12.4 years of follow-up in the Scandinavian Prostate Cancer Group (SPCG-4) study, the cumulative prevalence of erectile dysfunction was 84% in the intervention group and 80% in the watchful-waiting group; the rates of urinary leakage were 41% and 11%, respectively.38 After 2 years of follow-up in the Prostate Cancer Intervention Versus Observation Trial (PIVOT), men who underwent radical prostatectomy had higher rates of urinary incontinence (17.1% v. 6.3%) and erectile dysfunction (81.1% v. 44.1%) than men in the observation arm.28 Cohort studies consistently reported an increased risk of erectile dysfunction associated with radical prostatectomy (RR 1.6, 95% CI 1.3–1.8, affecting 23.4% or 234 per 1000 of men) compared with no treatment.39–43 Both RCTs28,38 and cohort studies39–42 found that radical prostatectomy increased the risk of urinary incontinence compared with watchful waiting (meta-analysis of trial data: RR 3.2, 95% CI 2.3–4.6; 17.8% of men or 178 per 1000 affected13).
In addition, between 11% and 21% of men will experience short-term (< 30 d) postoperative complications, such as infection, additional surgery and blood transfusions.28,44–46 Trial data indicate that radiation therapy increases the risk of urinary incontinence47 (not supported by observational studies13) and erectile dysfunction.39–42 Combined radiation and hormonal therapy also increases the risk of bowel and erectile dysfunction.13 Hormonal therapy (androgen deprivation therapy) has been shown to increase the risk of erectile dysfunction (76% v. 33% in control group), but not urinary incontinence (9% v. 6% in control group).13
The studies included in the systematic review used various strategies for following control participants that often were not clearly defined.13 It was unclear which (if any) control groups were followed with active surveillance (continued monitoring with repeat PSA testing, and biopsy and treatment depending on the test results48) or with watchful waiting (no active treatment; treatment of symptoms only as they progress48).
Recommendations
A summary of the recommendations is shown in Box 2, and a clinical summary is provided in Appendix 3 (www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.140703/-/DC1). The task force based the recommendations on the overall balance between the possible benefits and harms of PSA screening (with or without digital rectal examination), weighing the possible benefits against potential harms of early diagnosis and treatment of prostate cancer. There is (a) no evidence that PSA screening reduces overall mortality among men of any age; (b) conflicting evidence suggesting a small and uncertain potential reduction in prostate cancer mortality among men aged 55–69 years; (c) no convincing evidence of a reduction in prostate cancer mortality in any other age group; and (d) consistent evidence that screening and active treatment lead to harm.
Summary of recommendations for clinicians and policy-makers
The recommendations apply to all men without a previous diagnosis of prostate cancer.
For men aged less than 55 years, we recommend not screening for prostate cancer with the prostate-specific antigen (PSA) test. (Strong recommendation; low-quality evidence.)
For men aged 55–69 years, we recommend not screening for prostate cancer with the PSA test. (Weak recommendation; moderate-quality evidence.)
For men 70 years of age and older, we recommend not screening for prostate cancer with the PSA test. (Strong recommendation; low-quality evidence.)
Recommendations apply to men in the general population. This includes men with lower urinary tract symptoms (e.g., nocturia, urgency, frequency and poor stream) and those with benign prostatic hyperplasia. About 25% of men in the screening trials had lower urinary tract symptoms, and benign prostatic hyperplasia is not a risk factor for prostate cancer.49
Groups at increased risk of prostate cancer and of dying from it include men of black race and men with a family history of prostate cancer. There are no trial data showing that the benefits or harms of screening differ in these populations, as compared with men in the general population. However, clinicians may wish to discuss the benefits and harms of screening with men at increased risk of prostate cancer, with explicit consideration of their values and preferences.
Recommendations do not apply to the use of the PSA test for surveillance after diagnosis or treatment of prostate cancer.
Men aged less than 55 years
For men aged less than 55 years of age, we recommend not screening for prostate cancer with the PSA test. (Strong recommendation; low-quality evidence.)
This recommendation is based on the low incidence of prostate cancer and prostate cancer mortality, and the lack of evidence for benefit of screening in this age group, as well as the evidence of harms.
Men aged 55–69 years
For men aged 55–69 years of age, we recommend not screening for prostate cancer with the PSA test. (Weak recommendation; moderate-quality evidence.)
This recommendation places a relatively low value on a small and uncertain potential reduction in prostate cancer mortality, and a relatively higher value on the risk of a false-positive result, unnecessary biopsies, overdiagnosis of prostate cancer and harms associated with unnecessary treatment. The risks and benefits of PSA screening and its potential consequences should be discussed with each patient in the context of his preferences. Men who place a high value on a small potential reduction in mortality and are less concerned with undesirable consequences may choose to be screened.
Men 70 years of age and older
For men 70 years of age and older, we recommend not screening for prostate cancer with the PSA test. (Strong recommendation; low-quality evidence.)
This recommendation reflects the lower life expectancy and the lack of evidence for benefit of screening in this age group as well as the evidence of harms.
For men aged less than 55 years and those 70 years and older, the strong recommendations against PSA screening reflect our certainty that the potential harms of screening outweigh the benefits given the current state of knowledge.
Considerations for implementation
Patient values and preferences
Because of recent efforts to encourage screening for prostate cancer, some men may be interested in PSA screening despite the current recommendations. Evidence suggests that a patient’s perceived vulnerability to the disease, as a result of family history or otherwise, and physician recommendation are both associated with patient request for screening with the PSA test.50 Although high-quality evidence on the best way to facilitate informed decision-making about prostate cancer screening is lacking, such discussions should aim to elicit the knowledge, preferences and values of patients who ask about PSA screening.51,52 Many men view screening positively but are unaware of the potential harms.53 In addition to a focus on the patient’s values and preferences, informed decision-making requires practitioners to distinguish between the benefits and harms of screening, subsequent investigations and treatment, including an overview of diagnostic and therapeutic options in the event that the PSA test result is abnormal.
The task force recognizes that some men may place greater value on the potential benefits of screening than on the harms and risks associated with diagnosis and treatment and may choose to be screened with the PSA test. To facilitate informed decision-making about screening for prostate cancer, the task force has developed decision aids and tools that are available at www.canadiantaskforce.ca.
Costs
The task force did not consider the costs of screening or treatment of prostate cancer when formulating these recommendations.
Suggested performance indicators
Suggested performance measures include rates of PSA testing and subsequent follow-up, and the degree to which men who request screening were accurately informed of the risks and benefits of screening (ideally using an evidence-based decision aid). Incidence and mortality data related to prostate cancer should continue to be monitored at the provincial, territorial and national levels.
Other guidelines
The screening recommendations from other national and international organizations are shown in Table 3.8,11,22,54–61 The current recommendations are consistent with the task force’s 1994 recommendations and with the latest guidelines from the US Preventive Services Task Force8 and Cancer Council Australia.61 The National Health Service in the United Kingdom has no organized screening program, but it suggests that men who are concerned about the risk of prostate cancer receive clear and balanced information about the advantages and disadvantages of the PSA test and prostate cancer treatments.57
Summary of recommendations for PSA screening for prostate cancer from Canada and elsewhere
Other screening tests
Several tests to supplement the PSA test in screening for prostate cancer have been developed in recent years. These tests are intended to detect cell activity in potentially malignant cells and are aimed at assessing risk during various stages, such as after a positive biopsy result,62 after a negative biopsy result63–65 and after surgery.66 These tests measure molecular markers, which typically indicate patterns of gene activity, such as expression of the PCA3 gene and other DNA mutations, which occur in the presence of a malignant tumour. We recommend that such tests not be used until sufficient evidence is available on their diagnostic performance, harms and benefits.
Gaps in knowledge
Future research may be best focused on finding alternatives to the PSA test for prostate cancer screening and reducing inappropriate PSA testing. It has been suggested that baseline PSA testing could help to assess the future risk of death or metastasis from prostate cancer (which could support risk-based screening67,68), but more work is needed before this approach could be recommended. Magnetic resonance imaging and clinical decision rules are also promising approaches to improving the risk:benefit ratio of screening, but they have not been tested in rigorous randomized trials. Future research should also develop methods to identify the subset of men with prostate cancer in whom clinically relevant disease would develop (in the absence of treatment). Altering PSA thresholds or screening intervals, as well as uncoupling screening from treatment (because some men may opt for watchful waiting or active surveillance instead of active treatment), may favourably change the risk:benefit ratio of PSA screening; however, more research is needed to test this hypothesis. Trials investigating the benefits of PSA screening among men with a family history of prostate cancer or men of black race would be helpful to determine whether screening in these high-risk populations is warranted. Finally, randomized trials should compare watchful waiting or active surveillance, or both, with treatment with a curative intent.
Conclusion
Available evidence does not conclusively show that PSA screening will reduce prostate cancer mortality, but it clearly shows an increased risk of harm. The task force recommends that the PSA test should not be used to screen for prostate cancer. The implication of the strong recommendations against screening men less than 55 years of age and those 70 years of age and older is that clinicians should not routinely discuss screening with men in these age groups unless the topic is raised by the patient. The implication of the weak recommendation against screening men aged 55–69 years is that clinicians who believe a patient places a high value on the small potential benefit of screening and is less concerned about the harms may wish to discuss the benefits and harms of screening with the patient so that he can make an informed decision about whether to be screened. Any use of PSA testing to screen for prostate cancer requires a thoughtful discussion between the clinician and the patient about the balance between unclear benefits and substantial harms.
Key pointsThe prevalence of undiagnosed prostate cancer at autopsy is high and increases with age (> 40% among men aged 40–49 yr to > 70% among men aged 70–79 yr).
Only a small proportion of men with prostate cancer have symptoms or die from the disease; most prostate cancers are slowly progressive and not life threatening.
Screening with the PSA test may lead to a small reduction in prostate cancer mortality but not a reduction in all-cause mortality.
Thresholds for PSA of 2.5 to 4.0 ng/mL are commonly used for screening; lower thresholds increase the probability of false-positive results, and no threshold completely excludes prostate cancer.
Harms associated with PSA screening (e.g., bleeding, infection, urinary incontinence, a false-positive result and overdiagnosis) are common.
The PSA test should not be used for screening without a detailed discussion with the patient, ideally with the use of decision aids to facilitate comprehension.
Acknowledgements
The authors acknowledge Ali Usman and Donna Fitzpatrick-Lewis, members of the Evidence Review and Synthesis Centre research team who conducted the systematic review upon which these recommendations were based; the staff at the Prevention Guidelines Division and the Surveillance Division of the Public Health Agency of Canada; and the reviewers whose comments helped to improve the quality of this manuscript (Kami Kandola, Council of Chief Medical Officers of Health liaison representative; Scott D. Smith, College of Family Physicians of Canada; Anthony B. Miller, Dalla Lana School of Public Health, University of Toronto; Chris Del Mar, Bond University; Robert Nuttall, Canadian Cancer Society; Barbara Foster, Health Canada; Amanda Shaw, Public Health Agency of Canada; and Alexandra Barratt, School of Public Health, University of Sydney).
Footnotes
Competing interests: None of the authors (members of the guideline writing group) have declared competing interests.
This article has been peer reviewed.
The complete list of current members of the Canadian Task Force on Preventive Health Care is available at http://canadiantaskforce.ca/about-us/members/
Contributors: Neil Bell, Sarah Connor Gorber, Amanda Shane, James Dickinson, Lesley Dunfield and Marcello Tonelli contributed substantially to the study concept and design, and the analysis and interpretation of the evidence, and drafted the manuscript. Michel Joffres contributed to the analysis and interpretation of data and revised the draft. Harminder Singh and Elizabeth Shaw contributed to the interpretation of data and revised the draft. All of the authors approved the final version of the manuscript and agree to act as guarantors of the work.
Funding: Funding for the Canadian Task Force on Preventive Health Care is provided by the Public Health Agency of Canada and the Canadian Institutes of Health Research. The views of the funding bodies have not influenced the content of the guideline; competing interests have been recorded and addressed. The views expressed in this article are those of the authors and do not represent those of the Public Health Agency of Canada.