“Medicine used to be simple, ineffective and relatively safe ... Now it is complex, effective and potentially dangerous” — C. Chantler
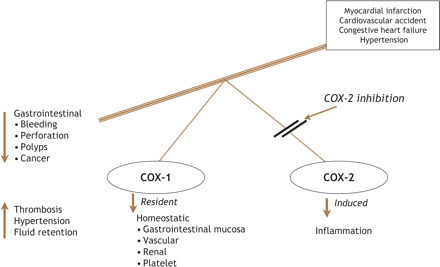
Few issues associated with new drug development have generated as much attention and controversy as the introduction and then withdrawal from general use of rofecoxib (Vioxx).1 Rofecoxib is one of a family of selective cyclooxygenase (COX)-2 inhibitors that were introduced to practitioners in 1999 as anti-inflammatory agents that apparently avoided the complications of gastrointestinal ulceration, perforation and bleeding associated with conventional NSAIDs such as ibuprofen, naproxen and indomethacin.2 Conventional NSAIDs inhibit both the COX-1 and COX-2 enzymes in a relatively balanced way, whereas the selective inhibitors were aimed at avoiding interference with the COX-1 enzyme, which is constitutive and protective, in the gastrointestinal tract (Fig. 1). As previously reviewed in the Journal, however, COX-2 inhibitors, unlike ASA, permit unopposed thromboxane A2 production by platelets, and thereby potentiate platelet aggregation, thrombosis and vasoconstriction.3 In addition, an imbalance between unopposed COX-1 activity and COX-2 inhibition may remove the protective effects of endogenous prostanoids such as prostacyclin and thereby promote hypertension, salt and water retention, and aggravation of congestive heart failure.
Fig. 1: Impact of COX-2 inhibition on physiologic functions and potential clinical implications.
About one year ago Lévesque and colleagues, using Quebec health insurance data and vital statistics for patients 66 years of age or older, reported an increased frequency of myocardial infarction associated with the use of rofecoxib, especially when the drug was taken in high doses.4 Concomitant use of ASA appeared to attenuate the risk at rofecoxib doses less than 25 mg. No increased risk was seen with traditional NSAIDs such as naproxen, nor with celecoxib (Celebrex), irrespective of dosage. These results confirmed those of an earlier nested case–control study by Graham and colleagues.5
In this issue, using the same Quebec cohort, Lévesque and colleagues demonstrate that the risk associated with rofecoxib in elderly patients without prior myocardial infarction appears to be greatest among first-time users and, interestingly, occurs in the early phase of exposure (i.e., median 9 [6.0–13.0] days after their first prescription).6 Close examination of the proportion of the days exposed to the drug indicates, however, that there is substantial overlap on these temporal estimates during at least the first 20 days. Although the overall rate of myocardial infarction was low (baseline 10.6/1000, increasing to 12.8 and 18.3 respectively for low and high doses of rofecoxib), the common use of these agents for arthritis and related problems in an elderly population at risk of heart disease represents an untoward and unnecessary public health hazard of major proportion that has been assertively addressed elsewhere.8 Indeed, the success of an aggressive marketing campaign, which resulted in over 100 million prescriptions for rofecoxib being dispensed over the 5 years after it was introduced in the United States, has given rise to estimates of between 88 000 and 140 000 excess cases of serious heart disease attributable to rofecoxib use.5 Recently an expert advisory panel on the safety of COX-2 selective NSAIDs convened by Health Canada (on which one of the study authors participated) voted 12–1 that rofecoxib be allowed back on the market in Canada and unanimously supported continuing the availability of celecoxib.7 The panel concluded that “the appropriate use of these agents be influenced by individual circumstances of each patient and that a broad selective of anti-inflammatory drugs is desirable given that effectiveness of one agent may diminish over the time and that the same patient may respond differently to different agents.”
It would have been helpful to know in the current study what doses of ASA were used and to what extent this might have played a role in reducing the risk of myocardial infarction. It seems likely that the overall risk is underestimated in the general population using these anti-inflammatory agents since patients with prior myocardial infarction were excluded and since silent myocardial infarction as well as sudden death before admission to hospital — which accounts for as many as one-half of deaths from this event — were not surveyed. The authors wisely highlight that they cannot rule out the possibility that risk increases with long-term use. Indeed, both dose and duration of rofecoxib use have been associated in other data with excess risk, and there is likely a sound pathophysiologic basis for temporal dispersion of the risk.8,9 Grosser and colleagues suggest that in a population with exaggerated endogenous hemostasis, cardiovascular risk may emerge rapidly.9 If, however, excess blood pressure, growth of vascular smooth muscle and genetic predisposition are at play, a longer time and dose–response relation for risk may unfold.
In the spirit of Chantler's admonition at the outset of this commentary, what are the lessons to be learned from the this remarkable and unfortunate story of rofecoxib use? First, there is clear heterogeneity across the NSAIDs in the intensity of their effect: rofecoxib's potency as an inhibitor is at least 9 times that of celecoxib and more than 200 times that of ibuprofen and naproxen.3 It follows that, if a COX-2 inhibitor is to be used at all, then celecoxib is preferred and a low dose is most desirable. Second, it has been estimated that only a small proportion of the millions of prescriptions for COX-2 inhibitors provided over the past 5 years were for patients who clearly warranted such a strategy to avoid excess gastrointestinal hazard. The current recommendation of the US Food and Drug Administration is to use nonselective NSAIDs in conjunction with a protein pump inhibitor, which seems a sensible alternative strategy.10 Given other evidence suggesting ibuprofen may interfere with the protective effects of ASA, naproxen would seem the logical nonselective NSAID of choice in patients at risk of vascular disease.11 Third, much of the morbidity and mortality associated with COX-2 inhibitors could have been avoided had the proper trials involving patients at risk been performed with full and comprehensive reporting of the risk.5,8 Finally, uncommon side effects of new drugs used for common diseases should be carefully evaluated, both during the period when such agents are approved for general use and after. Careful surveillance of phase IV trials and regulatory supervision needs to be intensified in this country and elsewhere. Understanding how to use COX-2 inhibitors while weighing the risks and potential benefits for individual patients is a high priority for all of us who are entrusted with the care of those with multiple comorbidities who require symptomatic relief for arthritic and other pain.
@ See related article page 1563
Footnotes
-
This article has been peer reviewed.
Competing interests: None declared.