Article Figures & Tables
Figures
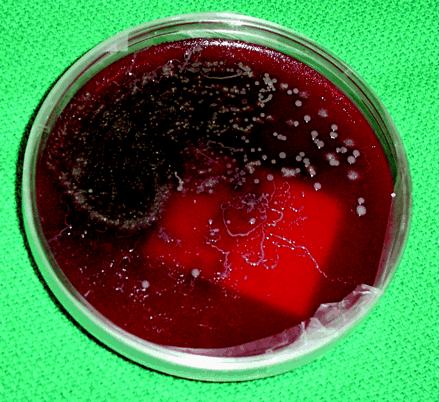
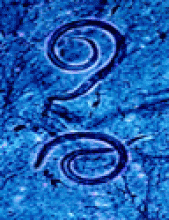
Fig. 1: Strongyloides stercoralis larva tracks on a blood agar plate from the bronchoalveolar lavage of a patient with disseminated strongyloidiasis.
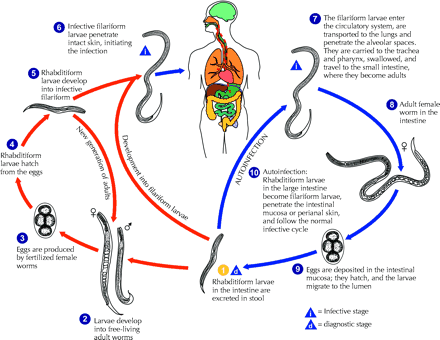
Fig. 2: The life cycle of Strongyloides stercoralis. The life cycle is more complex than that of most nematodes, with its alternation between free-living and parasitic cycles and its potential for autoinfection and multiplication within the host. In the free-living cycle, the rhabditiform larvae passed in the stool (1) can either moult twice and become infective filariform larvae (5) or moult 4 times and become free-living adult males or females (2) that mate and produce eggs (3), from which rhabditiform larvae hatch (4). The rhabditiform larvae (5) in turn can develop into either a new generation of free-living adults (2) or infective filariform larvae (6). The filariform larvae penetrate the human host skin to initiate the parasitic cycle (6). In the parasitic cycle, filariform larvae in contaminated soil penetrate the human skin (6) and are transported to the lungs, where they penetrate the alveolar spaces. They are carried through the bronchial tree to the pharynx, are swallowed and then reach the small intestine (7). In the small intestine they moult twice and become adult female worms (8). The females live threaded in the epithelium of the small intestine and, through parthenogenesis, produce eggs (9), which yield rhabditiform larvae. The rhabditiform larvae can either be passed in the stool (1) or cause autoinfection (10). In autoinfection, the rhabditiform larvae become infective filariform larvae, which can penetrate either the intestinal mucosa (internal autoinfection) or the skin of the perianal area (external autoinfection). In either case, the filariform larvae may follow the previously described route, being carried successively to the lungs, the bronchial tree, the pharynx and the small intestine, where they mature into adults, or they may disseminate widely in the body. To date, the occurrence of autoinfection in humans with helminthic infections is recognized only in S. stercoralis and Capillaria philippinensis infections. In the case of S. stercoralis, autoinfection may explain persistent infection in people who have not been in a disease-endemic area for many years and hyperinfection in immunodepressed patients. Source: Division of Parasitic Diseases, US Centers for Disease Control and Prevention, Atlanta. Photo: US Centers for Disease Control and Prevention / Alexander J. da Silva, PhD / Melanie Moser